Advanced Catalytic Route for High-Purity R-Glycerol Acetonide Intermediates
Introduction to Novel Chiral Intermediate Synthesis
The pharmaceutical industry continuously demands higher purity chiral building blocks to ensure the safety and efficacy of active pharmaceutical ingredients (APIs). A significant technological advancement in this domain is detailed in patent CN114195754B, which discloses a robust preparation method for the R-glycerol acetonide intermediate, specifically identified as S-(-)-4-chloromethyl-2,2-dimethyl-1,3-dioxolane. This compound serves as a pivotal chiral precursor in the synthesis of landiolol hydrochloride, an ultra-short-acting beta-1 receptor blocker essential for emergency cardiac care. The disclosed methodology addresses long-standing challenges in maintaining stereochemical integrity during the ring-opening condensation of S-epichlorohydrin. By leveraging specific Lewis acid catalysts, the process achieves exceptional enantiomeric excess values exceeding 96%, coupled with yields surpassing 85%. For global procurement teams and R&D directors, this represents a critical opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering materials that meet the stringent regulatory standards required for cardiovascular drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral glycerol formal derivatives has relied heavily on traditional Lewis acids such as boron trifluoride diethyl etherate (BF3·Et2O). While effective to a degree, these conventional catalytic systems often struggle to maintain optimal chiral purity under standard reaction conditions. Data indicates that processes utilizing boron-based catalysts frequently result in enantiomeric excess values hovering around 95%, which may be insufficient for next-generation APIs requiring ultra-high optical purity. Furthermore, the handling of boron trifluoride complexes can present operational hazards and complicate the downstream purification process due to the formation of stable complexes that are difficult to separate. These limitations not only impact the final quality of the API but also introduce variability in batch-to-batch consistency, posing significant risks for supply chain reliability in high-stakes therapeutic areas like cardiology.
The Novel Approach
The innovative strategy outlined in the patent data introduces a paradigm shift by employing alternative Lewis acid catalysts, specifically highlighting the efficacy of titanium tetraisopropoxide (Ti(OiPr)4) and iron(III) chloride (FeCl3). This novel approach fundamentally alters the reaction landscape, enabling the ring-opening condensation to proceed with superior stereocontrol. Experimental results demonstrate that substituting traditional boron catalysts with titanium-based systems can elevate the e.e. value to an impressive 99%, while simultaneously boosting chemical yields to over 92%. This improvement is not merely incremental; it represents a substantial leap in process efficiency that directly translates to reduced waste and lower raw material consumption. For manufacturers, this means a streamlined pathway to high-purity intermediates that simplifies the overall synthetic route for complex cardiovascular medications, thereby enhancing the economic viability of the production process.
Mechanistic Insights into Lewis Acid Catalyzed Ring-Opening Condensation
The core of this technological breakthrough lies in the precise mechanistic interaction between the chiral epoxide substrate and the selected Lewis acid catalyst within an acetone medium. The reaction involves the activation of the epoxide ring of S-epichlorohydrin by the metal center of the catalyst, which increases the electrophilicity of the carbon atoms and facilitates nucleophilic attack by the oxygen atom of the acetone solvent. This ring-opening condensation is highly sensitive to the steric and electronic properties of the catalyst. Titanium tetraisopropoxide, being a bulky and moderately strong Lewis acid, likely creates a specific coordination environment that shields one face of the epoxide, thereby preventing racemization and ensuring that the chiral information from the starting material is faithfully transferred to the dioxolane product. This mechanistic precision is crucial for avoiding the formation of unwanted enantiomers that would otherwise require costly and yield-reducing resolution steps later in the synthesis.
Furthermore, the choice of catalyst significantly influences the impurity profile of the final product. In conventional methods, side reactions such as polymerization of the epoxide or over-reaction with the solvent can lead to complex impurity spectra that are difficult to purge. The novel catalytic system described minimizes these side pathways by operating under milder temperature conditions, typically ranging from -70°C to 40°C, with a preferred window around 0°C. This thermal control, combined with the specific reactivity of catalysts like FeCl3 or Ti(OiPr)4, ensures that the reaction proceeds cleanly to the desired 1,3-dioxolane structure. The ability to quench the reaction simply with aqueous bases like sodium bicarbonate or organic amines like triethylamine further underscores the robustness of the mechanism, allowing for easy separation of the catalyst residues and resulting in a crude product that requires minimal purification to achieve pharmaceutical grade specifications.
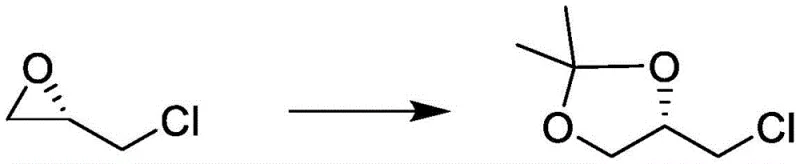
How to Synthesize S-(-)-4-chloromethyl-2,2-dimethyl-1,3-dioxolane Efficiently
Implementing this advanced synthesis route requires careful attention to reaction parameters to maximize the benefits of the novel catalytic system. The process begins with the dissolution of the chosen catalyst in acetone, followed by the controlled addition of S-epichlorohydrin. Maintaining the stoichiometric ratio of substrate to catalyst between 1:0.01 and 1:0.5 is critical for balancing reaction rate and cost efficiency. The reaction mixture is then stirred for a duration of 0.5 to 7 hours, depending on the specific catalyst activity and temperature profile selected. Upon completion, confirmed by gas chromatography (GC), the reaction is terminated via a neutralization step. This straightforward workup procedure eliminates the need for complex chromatographic separations often associated with chiral synthesis, making the protocol highly attractive for industrial adoption. Detailed standardized operating procedures for scaling this reaction are provided in the technical guide below.
- Dissolve the selected Lewis acid catalyst (such as Ti(OiPr)4 or FeCl3) in acetone solvent within a reaction vessel equipped with temperature control.
- Cool the reaction mixture to a temperature between -70°C and 40°C, preferably around 0°C, and slowly add S-epichlorohydrin dropwise while maintaining the internal temperature.
- After the reaction is complete (monitored by GC), quench the mixture with a base such as sodium bicarbonate or triethylamine, extract with dichloromethane, and purify to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel catalytic process offers compelling strategic advantages beyond mere technical performance. The primary benefit lies in the substantial cost reduction in pharmaceutical intermediate manufacturing driven by the elimination of expensive and hazardous reagents. By replacing boron trifluoride complexes with more abundant and manageable metal salts like iron chloride or titanium alkoxides, the raw material costs are significantly lowered. Additionally, the simplified workup procedure reduces the consumption of solvents and energy required for purification, leading to a leaner and more cost-effective production cycle. These efficiencies accumulate to provide a competitive pricing structure for the final intermediate, allowing downstream API manufacturers to optimize their own cost of goods sold without compromising on quality standards.
- Cost Reduction in Manufacturing: The adoption of this catalytic system removes the dependency on high-cost specialty reagents and minimizes the generation of hazardous waste streams. The use of inexpensive solvents like acetone and dichloromethane, coupled with the ability to recover and recycle unreacted starting materials, drives down the overall variable cost per kilogram. Furthermore, the high yield achieved (over 90% in optimized examples) means that less raw material is wasted, directly improving the material balance and reducing the environmental burden associated with waste disposal. This economic efficiency is vital for maintaining margins in the highly competitive generic and branded pharmaceutical markets.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is a cornerstone of supply chain stability. Because the process tolerates a relatively wide range of temperatures and uses readily available industrial chemicals, the risk of production delays due to reagent shortages or equipment failures is drastically minimized. Suppliers utilizing this method can guarantee shorter lead times for high-purity pharmaceutical intermediates, as the simplified purification steps allow for faster turnaround from reactor to shipping. This reliability is particularly crucial for emergency medicines like landiolol, where uninterrupted supply is a matter of patient safety.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns well with modern green chemistry principles. The avoidance of corrosive boron species reduces the risk of equipment corrosion and operator exposure, facilitating safer commercial scale-up of complex pharmaceutical intermediates. The aqueous quenching step generates benign salt byproducts that are easier to treat in standard wastewater facilities compared to fluorine-containing waste streams. This compliance advantage simplifies the regulatory approval process for new manufacturing sites and ensures long-term operational continuity in regions with strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this chiral intermediate. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this intermediate into their existing supply chains and development pipelines. The answers reflect the consensus on best practices for handling and sourcing this specific class of chiral building blocks.
Q: What is the primary advantage of using Ti(OiPr)4 over BF3·Et2O in this synthesis?
A: According to patent data, using Ti(OiPr)4 as the catalyst yields significantly higher chiral purity (e.e. value up to 99%) compared to BF3·Et2O (e.e. value around 95%), which is critical for downstream pharmaceutical applications requiring strict stereochemical control.
Q: Can this process be scaled for industrial production of cardiovascular drug intermediates?
A: Yes, the process utilizes common solvents like acetone and dichloromethane with simple quenching procedures using bases like sodium bicarbonate, making it highly amenable to large-scale commercial manufacturing without complex equipment requirements.
Q: What represents the key quality metric for this R-glycerol acetonide intermediate?
A: The enantiomeric excess (e.e. value) is the most critical quality attribute, with the novel method achieving values greater than 96%, ensuring the intermediate is suitable for synthesizing high-value chiral drugs like Landiolol hydrochloride.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-(-)-4-chloromethyl-2,2-dimethyl-1,3-dioxolane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving cardiovascular drugs depends on the availability of high-quality chiral precursors. Our technical team has extensively analyzed the catalytic routes described in recent patents, including the superior titanium-catalyzed condensation method, and possesses the expertise to implement these processes at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of S-(-)-4-chloromethyl-2,2-dimethyl-1,3-dioxolane meets the exacting e.e. value requirements necessary for GMP-grade API synthesis.
We invite global partners to collaborate with us to leverage these advanced manufacturing capabilities for their next-generation drug projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your development timeline and production economics. Let us be your trusted partner in delivering high-purity pharmaceutical intermediates that power the future of healthcare.
