Advanced Electrochemical Manufacturing of High-Purity Phosphorothioate Intermediates for Global Pharma
Advanced Electrochemical Manufacturing of High-Purity Phosphorothioate Intermediates for Global Pharma
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the urgent need for greener, more sustainable manufacturing protocols. Patent CN112921345B introduces a groundbreaking direct electrochemical synthesis method for phosphorothioate compounds, a class of molecules critical to the development of modern pharmaceuticals and agrochemicals. This technology replaces traditional stoichiometric oxidants with clean electric energy, fundamentally altering the economic and environmental footprint of producing these high-value intermediates. By utilizing a precise three-electrode system with platinum electrodes, the process achieves efficient coupling of phosphite triesters and thiols under mild constant voltage conditions. For R&D directors and procurement specialists seeking a reliable phosphorothioate supplier, this patent represents a pivotal shift towards electrified organic synthesis that promises enhanced purity profiles and reduced operational complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of phosphorothioate compounds has relied heavily on the reaction between halophosphoric acid esters and thiols, a pathway fraught with significant logistical and safety challenges. The synthesis of the requisite halophosphoric acid ester precursors is inherently complex, often involving multiple steps that generate substantial amounts of corrosive halogenated waste, thereby increasing the burden on environmental compliance teams. Alternative cross-dehydrogenative coupling (CDC) reactions using hydrogen phosphite diesters have been explored, yet these methods frequently suffer from narrow substrate scope, particularly failing to accommodate aliphatic thiols which are essential for diverse drug candidates. Furthermore, methods employing chemical promoters like trichloroisocyanuric acid (TCCA) necessitate the use of stoichiometric oxidants, leading to atom inefficiency and complicated downstream purification processes to remove chlorinated byproducts. These traditional limitations result in higher production costs, longer lead times, and a larger environmental footprint, creating bottlenecks for the commercial scale-up of complex sulfur-containing intermediates.
The Novel Approach
In stark contrast to these legacy methods, the electrochemical strategy detailed in the patent offers a streamlined, catalyst-free pathway that leverages electrons as the primary reagent. This approach utilizes readily available phosphite triesters and thiols, reacting them directly in an acetonitrile solution containing a supporting electrolyte like tetrabutylammonium tetrafluoroborate. The reaction proceeds smoothly at temperatures ranging from 25 to 55°C under a controlled constant voltage of 0.6 to 1.4V, eliminating the need for harsh chemical oxidants entirely. This methodology not only simplifies the reaction setup but also dramatically improves the universality of the substrate, successfully accommodating a wide array of aromatic, heteroaromatic, and aliphatic thiols. 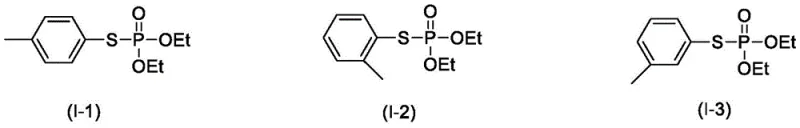
Mechanistic Insights into Direct Electrochemical Oxidative Coupling
The core of this innovation lies in the anodic oxidation mechanism where the thiol substrate is activated at the platinum anode surface to generate reactive thiyl radical species or cationic intermediates. These electro-generated species subsequently undergo nucleophilic attack by the phosphorus center of the phosphite triester, forming the P-S bond with high selectivity. The use of a three-electrode system with a silver/silver nitrate reference electrode ensures precise control over the oxidation potential, preventing over-oxidation of the sensitive sulfur or phosphorus moieties which often leads to impurity formation in batch chemical oxidations. This precise potential control is critical for maintaining high product integrity, as it allows the reaction to proceed through the desired kinetic pathway while suppressing side reactions that typically plague thermal methods. 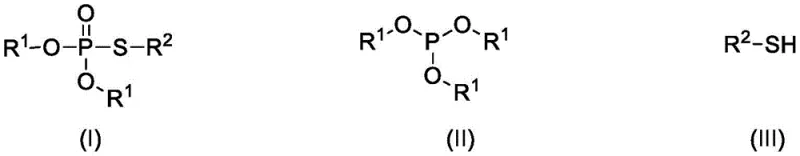
From an impurity control perspective, the absence of external chemical oxidants means there are no residual oxidizing agents or their reduced byproducts (such as chloride ions from TCCA) to contaminate the final product stream. The primary byproduct of this electrochemical transformation is hydrogen gas evolved at the cathode, which harmlessly vents from the system, leaving the reaction mixture remarkably clean. This cleanliness translates directly into simplified workup procedures, often requiring only solvent evaporation and standard column chromatography to achieve high-purity specifications. For quality assurance teams, this implies a more consistent impurity profile across batches, reducing the risk of unexpected toxicological alerts during regulatory filings. The ability to tune the reaction by adjusting voltage and current density provides an additional layer of process control that is unavailable in conventional thermal batch reactors.
How to Synthesize O,O-Diethyl-S-p-tolyl Phosphorothioate Efficiently
The synthesis of specific targets like O,O-diethyl-S-p-tolyl phosphorothioate serves as a benchmark for validating the efficiency of this electrochemical platform. The protocol involves dissolving the phosphite triester and the corresponding thiol in an acetonitrile solution of Bu4NBF4, followed by electrolysis under optimized conditions of 1.0V and 40°C. This specific set of parameters has been empirically determined to balance reaction rate with selectivity, ensuring that the conversion proceeds to completion within a practical timeframe of roughly 4 hours. While the general principles are straightforward, precise control of the electrode surface area and stirring rate is essential to maintain mass transfer and prevent concentration polarization at the electrode interface. Detailed standardized synthesis steps see the guide below.
- Prepare the electrolytic cell with Platinum (Pt) cathode and anode, using 0.1 mol/L silver nitrate acetonitrile solution as the reference electrode.
- Dissolve phosphite triester and thiol in 0.05-0.15 mol/L Bu4NBF4 acetonitrile solution with a molar ratio of 100: 160-200.
- Apply a constant voltage of 0.9-1.1V at 35-45°C for 3-5 hours, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this electrochemical methodology offers compelling strategic advantages that extend beyond mere technical novelty. The elimination of expensive and hazardous chemical oxidants directly correlates to a reduction in raw material costs and a simplification of the supply chain, as there is no longer a dependency on volatile oxidant markets. Furthermore, the mild reaction conditions reduce the energy load associated with heating and cooling large-scale reactors, contributing to lower overall utility expenses and a smaller carbon footprint for the manufacturing site. The robustness of the method across various substrates ensures that the same equipment setup can be utilized for a diverse portfolio of phosphorothioate intermediates, maximizing asset utilization and flexibility in production scheduling.
- Cost Reduction in Manufacturing: The removal of stoichiometric oxidants like TCCA eliminates a significant line item in the bill of materials, while simultaneously reducing the costs associated with waste disposal and neutralization of halogenated byproducts. The simplified purification process reduces solvent consumption and labor hours required for chromatography or crystallization steps, driving down the cost of goods sold significantly. Additionally, the use of electricity as a reagent allows for precise dosing of energy, minimizing waste and improving the overall atom economy of the transformation compared to traditional chemical oxidation routes.
- Enhanced Supply Chain Reliability: By relying on electricity rather than specialized chemical reagents that may face supply disruptions, manufacturers can insulate their production lines from upstream volatility in the chemical market. The starting materials, phosphite triesters and thiols, are commodity chemicals with stable global supply chains, ensuring consistent availability for long-term production contracts. This stability is crucial for meeting the rigorous delivery schedules demanded by multinational pharmaceutical clients who require uninterrupted supply of critical intermediates for their clinical and commercial programs.
- Scalability and Environmental Compliance: Electrochemical reactors are inherently scalable, with flow chemistry adaptations allowing for continuous processing that further enhances safety and throughput. The generation of hydrogen gas as the only stoichiometric byproduct aligns perfectly with increasingly stringent environmental regulations regarding hazardous waste discharge. This green chemistry profile facilitates faster regulatory approvals and strengthens the sustainability credentials of the final drug product, a key differentiator in modern pharmaceutical procurement.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity on process feasibility. Understanding these details is essential for evaluating the integration of this method into existing manufacturing workflows.
Q: What are the advantages of electrochemical synthesis over traditional chemical oxidants?
A: Electrochemical synthesis eliminates the need for stoichiometric chemical oxidants like TCCA or halogenated reagents, significantly reducing hazardous waste generation and simplifying the post-reaction purification process.
Q: Is this method suitable for aliphatic thiols?
A: Yes, unlike some traditional CDC reactions that are limited to aryl thiols, this electrochemical protocol demonstrates good universality and successfully reacts with both aromatic and aliphatic thiols such as cyclohexanethiol.
Q: What are the typical reaction conditions for scaling this process?
A: The process operates under mild conditions, typically between 25-55°C and 0.6-1.4V, using acetonitrile as the solvent, which facilitates easier thermal management during commercial scale-up compared to exothermic chemical oxidations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphorothioate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in delivering high-value intermediates with superior purity and sustainability. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch meets the exacting standards required for pharmaceutical applications. We are committed to leveraging cutting-edge technologies to provide our partners with a competitive edge in the global market.
We invite you to collaborate with us to explore how this electrochemical route can optimize your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your target molecule. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can support your development timeline and commercial goals effectively.
