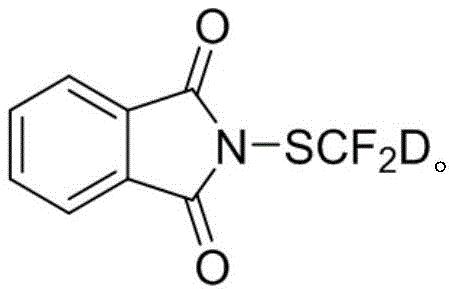
Advanced Deuterated Difluoromethylthio Reagent for High-Purity Drug Synthesis
The pharmaceutical and agrochemical industries are increasingly recognizing the strategic value of deuterated drugs, driven by the potential to enhance metabolic stability and prolong half-life without compromising potency. Patent CN114044751A, published in early 2022, introduces a groundbreaking methodology for the preparation of a deuterated difluoromethylthio reagent, specifically N-(difluoromethyl-d)thio)phthalimide (PhthSCF2D). This innovation addresses a critical gap in the availability of stable, high-purity deuterated building blocks capable of introducing the lipophilic and metabolically robust SCF2D moiety into complex molecular architectures. The patent details a robust three-step synthesis that avoids the harsh cryogenic conditions typical of prior art, thereby unlocking new possibilities for the commercial scale-up of complex pharmaceutical intermediates. By leveraging a unique deuterium exchange strategy on benzyl mercaptan, the inventors have achieved a deuterated purity of 97% D, setting a new benchmark for isotopic enrichment in fluorinated sulfur reagents.
The development of reliable deuterated reagents is paramount for modern drug discovery, particularly when aiming to block metabolic soft spots. The C-D bond is significantly stronger than the C-H bond, offering a kinetic isotope effect that can slow down oxidative metabolism by cytochrome P450 enzymes. However, the practical application of this principle has often been hindered by the scarcity of versatile reagents that can introduce deuterated fluoroalkyl groups under mild conditions. The technology disclosed in CN114044751A provides a comprehensive solution, offering a shelf-stable solid reagent that can be stored in dry environments and utilized across a diverse range of substrate classes. This advancement not only supports the synthesis of novel deuterated APIs but also facilitates mechanistic studies and the development of next-generation agrochemical intermediates with improved environmental profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of difluoromethylthio reagents, such as the non-deuterated PhthSCF2H, relied on methodologies that were operationally cumbersome and difficult to scale. Literature references, such as those found in J. Org. Process Res. Dev. 2015 and 2017, describe routes that often necessitate extremely low reaction temperatures, typically around -30°C, to control selectivity and prevent decomposition. These cryogenic requirements impose significant energy burdens and engineering challenges on manufacturing facilities, effectively limiting production to small laboratory scales. Furthermore, existing methods struggled to achieve high deuterium incorporation when attempting to synthesize deuterated analogs, often resulting in products with mixed isotopic compositions that compromised their utility in metabolic tracing or therapeutic applications. The inability to efficiently replace hydrogen with deuterium in the difluoromethylthio group represented a major bottleneck for researchers aiming to exploit the benefits of deuteration in fluorinated drug candidates.
The Novel Approach
The novel approach detailed in patent CN114044751A fundamentally reimagines the synthetic pathway to overcome these historical limitations. Instead of relying on direct deuteration of the final product or using expensive deuterated starting materials that lead to scrambling, the inventors developed a stepwise protocol starting with benzyl mercaptan. By utilizing sodium hydride to deprotonate the thiol and subsequently introducing alkaline heavy water (D2O/NaOH), the method ensures a clean exchange of the thiol proton for deuterium before the difluorocarbene insertion occurs. This strategic sequencing prevents hydrogen contamination from the solvent or reagents, securing the high 97% D purity. Moreover, the reaction conditions are remarkably mild, proceeding efficiently from 0-5°C up to room temperature, which drastically simplifies the equipment requirements and enhances process safety. This shift from cryogenic to near-ambient processing is a pivotal improvement for cost reduction in pharmaceutical intermediate manufacturing, enabling seamless transition from gram-scale discovery to multi-kilogram production.
Mechanistic Insights into Deuterated Difluoromethylthio Reagent Synthesis
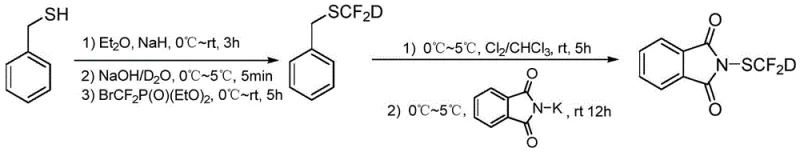
The core mechanistic innovation lies in the precise control of the deuterium source during the formation of the carbon-sulfur bond. In the first step, sodium hydride reacts with benzyl mercaptan to generate the corresponding thiolate anion while evolving hydrogen gas, which serves as a driving force and removes potential proton sources. The subsequent addition of alkaline heavy water ensures that the sulfur center is fully deuterated (S-D) prior to the introduction of the difluorocarbene species generated from reagents like bromodifluoromethyl diethyl phosphate. This pre-deuteration is critical; if the difluorocarbene were introduced first, subsequent exchange attempts might fail due to the stability of the C-S bond or lead to side reactions. The difluorocarbene then inserts into the S-D bond to form the intermediate BnSCF2D. In the second step, chlorination with Cl2 in chloroform converts the benzyl thioether into the highly reactive chlorodifluoromethanesulfenyl chloride (ClSCF2D). Finally, nucleophilic displacement by potassium phthalimide yields the target reagent. This mechanism ensures that the deuterium atom remains intact throughout the sequence, avoiding the isotopic dilution common in other labeling strategies.
Impurity control is another area where this mechanism offers distinct advantages. The use of benzyl mercaptan as a transient carrier for the deuterium allows for easy purification of the intermediate BnSCF2D via extraction, removing inorganic salts and unreacted starting materials before the sensitive chlorination step. The chlorination is performed in solution, minimizing the handling of volatile and toxic ClSCF2D. The final substitution with potassium phthalimide is highly selective, producing the phthalimide-protected reagent which is a stable crystalline solid. This stability is crucial for supply chain reliability, as it allows the reagent to be manufactured, quality controlled, and shipped without the degradation risks associated with liquid or unstable sulfenyl chlorides. The rigorous exclusion of moisture and the use of anhydrous solvents further ensure that hydrolysis byproducts are minimized, resulting in a high-purity product suitable for sensitive downstream applications in API synthesis.
How to Synthesize PhthSCF2D Efficiently
The synthesis of this high-value deuterated reagent follows a logical three-step sequence that balances yield, purity, and operational simplicity. The process begins with the generation of the deuterated benzyl thiol intermediate, followed by chlorination to activate the sulfur center, and concludes with the installation of the phthalimide protecting group. Each step has been optimized to maximize deuterium retention and overall yield, making it an attractive route for contract development and manufacturing organizations. For detailed procedural parameters, stoichiometry, and workup instructions, please refer to the standardized guide below which outlines the critical process controls identified in the patent documentation.
- Preparation of intermediate BnSCF2D via reaction of benzyl mercaptan with sodium hydride, alkaline heavy water, and a difluorocarbene reagent.
- Chlorination of the intermediate BnSCF2D using chlorine gas in chloroform to generate ClSCF2D solution.
- Reaction of the chloroform solution with potassium phthalimide followed by filtration, concentration, and recrystallization to obtain pure PhthSCF2D.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this new synthetic route offers tangible benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the manufacturing process. By eliminating the need for specialized cryogenic reactors capable of maintaining -30°C, facilities can utilize standard jacketed reactors, significantly reducing capital expenditure and energy consumption. This operational flexibility translates directly into cost reduction in fine chemical intermediates manufacturing, as the process becomes less dependent on expensive cooling infrastructure and more amenable to batch processing in existing multipurpose plants. Furthermore, the stability of the final PhthSCF2D product as a solid reduces the logistical complexities associated with shipping hazardous liquids or unstable intermediates, enhancing overall supply chain resilience.
- Cost Reduction in Manufacturing: The elimination of extreme low-temperature requirements removes a major cost driver associated with energy-intensive cooling systems and specialized equipment maintenance. Additionally, the use of commercially available and relatively inexpensive starting materials like benzyl mercaptan and sodium hydride ensures that raw material costs remain predictable and manageable. The high yield and purity reported in the patent examples suggest that downstream purification costs, such as extensive chromatography, can be minimized in favor of simpler crystallization or filtration techniques, further optimizing the cost of goods sold for this critical reagent.
- Enhanced Supply Chain Reliability: The robustness of the synthesis against minor variations in temperature (operating at 0-5°C rather than -30°C) makes the process more reproducible on a large scale, reducing the risk of batch failures. The ability to produce a stable, shelf-stable solid reagent means that inventory can be held safely for longer periods, allowing suppliers to buffer against demand fluctuations and ensure continuous availability for their clients. This reliability is crucial for pharmaceutical companies managing tight development timelines, as it reduces the lead time for high-purity pharmaceutical intermediates and mitigates the risk of project delays due to reagent shortages.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common organic solvents like diethyl ether, chloroform, and dichloromethane, which are well-understood in terms of waste management and recovery. The avoidance of exotic or highly toxic reagents simplifies the environmental health and safety (EHS) profile of the manufacturing site. As the industry moves towards greener chemistry, the efficiency of this route—characterized by high atom economy in the key steps and minimal byproduct formation—aligns well with sustainability goals, facilitating regulatory approval and reducing the environmental footprint of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this deuterated reagent technology. These answers are derived directly from the experimental data and claims presented in patent CN114044751A, providing a factual basis for evaluating its suitability for your specific R&D or production needs. Understanding these details is essential for integrating this reagent into your existing workflows effectively.
Q: What is the deuterium purity of the PhthSCF2D reagent described in patent CN114044751A?
A: The patent specifies that the prepared deuterated difluoromethylthio reagent achieves a deuterated purity of 97% D, ensuring high isotopic enrichment for metabolic studies.
Q: How does this new method improve upon previous synthesis routes for difluoromethylthio reagents?
A: Unlike prior art methods requiring cryogenic conditions (-30°C) and complex operations, this novel approach operates at mild temperatures (0-5°C to room temperature), facilitating easier mass production and operational safety.
Q: What types of drug substrates can be modified using this reagent?
A: The reagent demonstrates broad applicability, successfully introducing the SCF2D group into olefins, aldehydes, heteroaromatics, boronic acids, amines, alkynes, thiols, and beta-ketoesters via photocatalytic or thermal methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable PhthSCF2D Supplier
The technological breakthroughs described in patent CN114044751A represent a significant leap forward in the field of deuterated fluorinated chemistry, offering a practical pathway to access high-value intermediates that were previously difficult to source. NINGBO INNO PHARMCHEM stands at the forefront of translating such innovative academic and patent literature into commercial reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which are equipped to verify isotopic purity and chemical integrity, guaranteeing that every batch of PhthSCF2D meets the exacting standards required for global pharmaceutical applications.
We invite you to explore how this advanced reagent can accelerate your drug discovery programs or optimize your manufacturing processes. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive pricing structures. By partnering with us, you gain access not just to a chemical product, but to a comprehensive support system designed to navigate the complexities of modern chemical supply chains and deliver value at every stage of your project lifecycle.
