Scalable Synthesis of Baricitinib Intermediate via Novel Azetidine Route
Scalable Synthesis of Baricitinib Intermediate via Novel Azetidine Route
The pharmaceutical industry continuously seeks robust and economically viable pathways for synthesizing complex heterocyclic intermediates, particularly those serving as critical building blocks for kinase inhibitors. Patent CN114380727A, published in April 2022, introduces a groundbreaking preparation method for 2-[1-(ethylsulfonyl)-3-azetidine]acetonitrile, a pivotal intermediate in the synthesis of baricitinib, a potent oral JAK1 and JAK2 inhibitor. This technical disclosure addresses the longstanding challenges associated with the instability of the four-membered azetidine ring and the prohibitive costs of traditional synthetic routes. By shifting the starting material strategy from expensive N-Boc protected precursors to readily available 3-oxo-azetidine hydrochloride, the inventors have established a pathway that is not only chemically elegant but also industrially superior. For R&D directors and procurement managers evaluating supply chain resilience, this patent represents a significant opportunity to optimize the manufacturing of high-value rheumatoid arthritis therapeutics through a streamlined, four-step process involving protection, sulfonylation, condensation, and elimination.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in US2019/135807 and US2009/233903, rely heavily on N-Boc-3-oxoazetidine as the foundational starting material. This reliance creates a significant economic bottleneck, as N-Boc protected azetidines are inherently more expensive due to the additional synthetic steps required for their own preparation. Furthermore, these conventional routes often necessitate the use of costly reagents such as diethyl (2-cyanoethyl) phosphonate and strong bases like potassium tert-butoxide to effect C-hydroxylation or Horner-Wadsworth-Emmons type reactions. The cumulative effect of these expensive inputs, combined with relatively low overall yields hovering around 35% to 35.7%, renders these methods suboptimal for large-scale commercial production. The use of tetrahydrofuran as a primary solvent in critical steps further complicates solvent recovery and increases the environmental footprint, making these legacy processes less attractive for modern green chemistry initiatives.
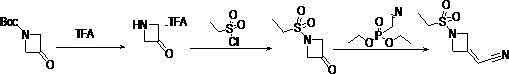
The Novel Approach
In stark contrast to the limitations of the prior art, the novel approach detailed in CN114380727A fundamentally reimagines the synthetic logic by utilizing 3-oxo-azetidine hydrochloride directly. This strategic shift eliminates the need for the expensive Boc protecting group at the outset, immediately lowering the raw material cost basis. The process ingeniously employs ethylene glycol to protect the ketone functionality as a spiro-ketal prior to sulfonylation, thereby stabilizing the sensitive four-membered ring against potential degradation under acidic or basic conditions. This protection strategy allows for the use of milder and more cost-effective sulfonylation conditions using ethylsulfonyl chloride. Subsequent condensation with ethyl cyanoacetate followed by a catalytic elimination step delivers the target nitrile with exceptional efficiency. This route not only simplifies the operational complexity but also significantly enhances the atom economy, making it a highly desirable candidate for industrial scale-up.
Mechanistic Insights into the Four-Step Cascade Synthesis
The core of this technological advancement lies in the precise orchestration of four distinct chemical transformations that maximize yield while minimizing impurity formation. The initial step involves the acid-catalyzed condensation of 3-oxo-azetidine hydrochloride with ethylene glycol in a high-boiling solvent like toluene or xylene at temperatures ranging from 110°C to 140°C. This step is critical as it forms a stable spiro-[azetidine-3,2'-[1,3]dioxolane] intermediate, effectively masking the reactive ketone. Following this, the sulfonylation is conducted in dichloromethane at controlled low temperatures between -10°C and 30°C using triethylamine or DIPEA as a base. This ensures selective N-sulfonylation without compromising the integrity of the azetidine ring or the ketal protection. The third step utilizes ethyl cyanoacetate in a base-mediated condensation, likely proceeding through a Knoevenagel-type mechanism or similar nucleophilic attack, to introduce the two-carbon nitrile precursor. Finally, the elimination step, catalyzed by lithium chloride or sodium hydroxide in polar aprotic solvents like DMSO at 80°C to 120°C, facilitates the dehydration and aromatization of the side chain to yield the final exocyclic double bond of the acetonitrile group.
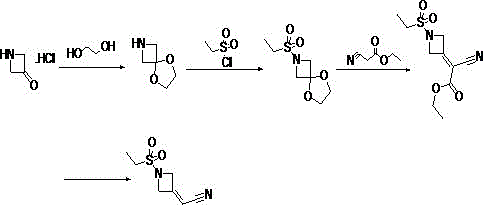
Impurity control is meticulously managed throughout this cascade, particularly regarding the stability of the azetidine ring. The use of the ethylene glycol ketal serves a dual purpose: it prevents self-condensation of the ketone and shields the ring from nucleophilic attack by the sulfonyl chloride. Furthermore, the choice of mild bases like sodium acetate or potassium carbonate in the condensation step, rather than aggressive alkoxides, minimizes ring-opening side reactions. The final purification involves activated carbon decolorization and recrystallization from ethyl acetate, which effectively removes trace metal salts and organic byproducts. This rigorous attention to reaction conditions ensures that the final product meets stringent purity specifications required for pharmaceutical intermediates, with the patent reporting isolated yields as high as 81.1% in the final step and over 90% in the intermediate stages.
How to Synthesize 2-[1-(ethylsulfonyl)-3-azetidine]acetonitrile Efficiently
The synthesis of this critical JAK inhibitor intermediate requires precise adherence to the patented protocol to ensure reproducibility and high purity. The process begins with the formation of the protected ketal, followed by sequential functionalization and elimination. Operators must maintain strict temperature controls, particularly during the exothermic sulfonylation and the high-temperature elimination phases. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures, are outlined below to guide process chemists in replicating this efficient route.
- Protect the ketone group of 3-oxo-azetidine hydrochloride using ethylene glycol and an acid catalyst in toluene or xylene at 110-140°C to form the spiro-ketal intermediate.
- Perform sulfonylation on the protected intermediate using ethylsulfonyl chloride and an organic base in dichloromethane at low temperatures (-10 to 30°C).
- Conduct a condensation reaction between the sulfonylated intermediate and ethyl cyanoacetate using a base in THF or toluene at 50-110°C.
- Execute the final elimination step using a catalyst like lithium chloride in DMSO/water at 80-120°C to yield the target acetonitrile derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers transformative advantages in terms of cost structure and supply reliability. The primary driver of cost reduction is the substitution of the expensive N-Boc-3-oxoazetidine starting material with the much more economical 3-oxo-azetidine hydrochloride. This change alone significantly lowers the entry cost for the synthesis. Additionally, the elimination of high-cost reagents such as diethyl (2-cyanoethyl) phosphonate and potassium tert-butoxide further compresses the bill of materials. The process avoids the use of cryogenic conditions often required for organolithium or Grignard reagents, instead operating at manageable temperatures that reduce energy consumption and infrastructure requirements. These factors collectively contribute to a substantially reduced manufacturing cost per kilogram, enhancing the margin potential for downstream API production.
- Cost Reduction in Manufacturing: The economic benefits of this route are derived from the strategic selection of commodity-grade raw materials and the avoidance of specialized, high-cost reagents. By utilizing ethyl cyanoacetate instead of phosphonate esters for the carbon chain extension, the process leverages widely available chemicals with stable pricing. Furthermore, the high yields achieved in each step—often exceeding 90% for intermediates—mean that less raw material is wasted, directly improving the overall material throughput. The simplified workup procedures, which rely on standard extraction and crystallization techniques rather than complex chromatography, also reduce labor and solvent costs, leading to significant operational savings.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of robust, non-proprietary starting materials that are sourced from multiple global suppliers. Unlike specialized Boc-protected azetidines which may have limited vendors, 3-oxo-azetidine hydrochloride is a more common building block with a broader supply base. This diversification reduces the risk of supply disruptions and provides greater negotiating leverage for procurement teams. The stability of the intermediates, particularly the spiro-ketal protected species, allows for potential storage and batch accumulation, enabling flexible production scheduling and better inventory management to meet fluctuating market demands for baricitinib.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing solvents like toluene, dichloromethane, and ethyl acetate which are standard in large-scale pharmaceutical manufacturing and have well-established recovery protocols. The reduction in the number of steps and the avoidance of heavy metal catalysts or hazardous reagents simplify the waste stream, facilitating easier treatment and disposal. This alignment with green chemistry principles not only reduces environmental compliance costs but also enhances the sustainability profile of the supply chain, a key metric for modern pharmaceutical companies aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and technical disclosures within patent CN114380727A, providing clarity on yield expectations, safety considerations, and scalability factors for potential licensees or manufacturing partners.
Q: Why is the new synthesis route more cost-effective than conventional methods?
A: The new route utilizes 3-oxo-azetidine hydrochloride, which is significantly cheaper than the N-Boc protected starting materials used in prior art. Additionally, it eliminates the need for expensive reagents like diethyl (2-cyanoethyl) phosphonate and potassium tert-butoxide, drastically reducing raw material costs.
Q: What are the yield improvements in this patented process?
A: While conventional methods reported in US patents often achieve total yields around 35%, this novel four-step process demonstrates individual step yields exceeding 90% for protection and sulfonylation, with a final isolation yield of over 80%, representing a substantial improvement in overall efficiency.
Q: How does this method address the instability of the four-membered ring?
A: The process employs a strategic protection-deprotection sequence using ethylene glycol to form a stable spiro-ketal intermediate. This stabilizes the reactive ketone functionality during the harsh sulfonylation conditions, preventing ring degradation and ensuring high purity of the final azetidine product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-[1-(ethylsulfonyl)-3-azetidine]acetonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient intermediate synthesis in the race to deliver life-saving medications to patients. Our technical team has thoroughly analyzed the pathway described in CN114380727A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life. We are equipped with rigorous QC labs and state-of-the-art pilot plants capable of handling the specific thermal and solvent requirements of this azetidine synthesis, ensuring that every batch meets stringent purity specifications. Our commitment to quality assurance means that we can reliably supply this complex heterocyclic intermediate with the consistency demanded by global regulatory standards.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for baricitinib and related JAK inhibitors. By leveraging our expertise in process optimization, we can help you achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most robust and economically viable chemical strategy available in the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
