Advanced Organocatalytic Strategy For High-Purity Chiral Beta-Aminoketone Derivatives And Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies for constructing chiral building blocks, particularly those containing nitrogen functionalities which are ubiquitous in bioactive molecules. Patent CN109748841B introduces a groundbreaking approach for the catalytic asymmetric synthesis of chiral beta-aminoketone derivatives, addressing critical limitations in existing synthetic routes. This technology utilizes a novel chiral binaphthol derivative catalyst to facilitate the asymmetric 1,4-addition of potassium organotrifluoroborate salts to beta-phthalimide acryl ketone compounds. The significance of this invention lies in its ability to generate valuable chiral intermediates with high enantioselectivity and yield under mild, metal-free conditions. These chiral beta-aminoketone structures are pivotal precursors for numerous high-value applications, ranging from the side chains of anticancer drugs like paclitaxel to immune response modifiers and protein phosphatase inhibitors. By shifting away from traditional transition metal catalysis, this method offers a greener, more sustainable pathway that aligns perfectly with the rigorous quality and environmental standards demanded by modern regulatory bodies.
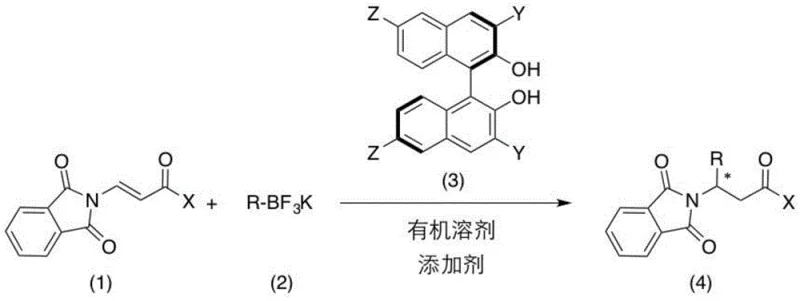
For R&D directors evaluating process feasibility, the shift from metal-catalyzed hydrogenation or addition reactions to organocatalysis represents a significant strategic advantage. Conventional methods for synthesizing chiral beta-aminoketones often rely on expensive noble metals such as rhodium, ruthenium, or palladium, which not only drive up raw material costs but also introduce severe purification challenges. The removal of trace metal residues to meet parts-per-million (ppm) specifications in active pharmaceutical ingredients (APIs) typically requires additional processing steps involving specialized scavengers or complex chromatography, which can drastically reduce overall throughput. Furthermore, metal catalysts often exhibit sensitivity to air and moisture, necessitating stringent operational controls that complicate scale-up efforts. In contrast, the novel approach detailed in the patent employs a polyfluorinated chiral binaphthol skeleton that operates effectively without any transition metals. This organocatalytic system leverages the inherent stability of potassium organotrifluoroborates, which are superior to traditional boronic acids in terms of shelf-life and resistance to protodeboronation. The result is a reaction profile that is not only highly stereoselective, achieving ee values exceeding 99% in optimized examples, but also operationally simple, requiring standard laboratory equipment rather than specialized high-pressure hydrogenation reactors.
Mechanistic Insights into Polyfluorinated Binaphthol Catalyzed Asymmetric 1,4-Addition
The core of this technological breakthrough resides in the unique design of the chiral catalyst and the synergistic effect of the additive system. The patent highlights a series of binaphthol derivatives (L1-L6), with catalyst L6 featuring a 3,3'-bis(2,3,5,6-tetrafluoro-4-(trifluoromethyl)phenyl) substitution pattern proving to be exceptionally effective. The presence of multiple electron-withdrawing fluorine atoms significantly enhances the acidity of the hydroxyl groups on the binaphthol backbone, thereby strengthening the hydrogen-bonding interactions with the substrate. This activation is crucial for inducing high facial selectivity during the nucleophilic attack. The mechanism likely involves the formation of a tight ion pair or a coordinated complex between the chiral catalyst, the electrophilic beta-phthalimide acryl ketone, and the activated boron species. The additives play a non-trivial role in this catalytic cycle; specifically, the combination of lithium bromide and boron trifluoride diethyl etherate acts to activate the relatively inert potassium trifluoroborate salt, facilitating the transmetallation or direct nucleophilic addition step. Additionally, the inclusion of 4 Angstrom molecular sieves serves to sequester trace water, preventing the hydrolysis of the boron reagent and maintaining the integrity of the Lewis acidic environment required for high turnover.
From an impurity control perspective, this organocatalytic route offers a cleaner profile compared to metal-mediated processes. Without the risk of metal-induced side reactions such as over-reduction or isomerization, the primary impurities are typically limited to unreacted starting materials or minor diastereomers, which are easily separated. The high enantiomeric excess observed across a broad range of substrates, including sterically hindered heterocycles and alkenyl chains, suggests a robust transition state that is resistant to minor fluctuations in reaction parameters. This mechanistic robustness is essential for technology transfer, as it ensures that the stereochemical outcome remains consistent whether the reaction is performed on a gram scale in a research lab or a multi-kilogram scale in a production facility. The ability to tolerate diverse functional groups on both the electrophile and the nucleophile further underscores the versatility of this catalytic system, making it a powerful tool for the late-stage functionalization of complex molecular architectures.
How to Synthesize Chiral Beta-Aminoketone Derivatives Efficiently
The practical implementation of this synthesis protocol is designed for scalability and ease of execution, minimizing the need for exotic reagents or extreme conditions. The general procedure involves charging a reaction vessel with the beta-phthalimide acryl ketone substrate, the specific potassium organotrifluoroborate salt, and the chiral binaphthol catalyst L6 in anhydrous trifluorotoluene. Crucially, the activation system comprising 4 Angstrom molecular sieves, lithium bromide, and boron trifluoride diethyl etherate is added to initiate the transformation. The mixture is then heated to a moderate temperature, typically around 60 degrees Celsius, and stirred until conversion is complete as monitored by thin-layer chromatography. Workup is straightforward, involving filtration through celite to remove the solid additives and catalyst residues, followed by concentration and purification via standard silica gel column chromatography.
- Prepare the reaction system by adding chiral binaphthol catalyst L6, beta-phthalimide acryl ketone substrate, and potassium organotrifluoroborate salt into anhydrous trifluorotoluene under nitrogen protection.
- Introduce specific additives including 4 Angstrom molecular sieves, lithium bromide, and boron trifluoride diethyl etherate to activate the nucleophile and enhance stereoselectivity.
- Heat the sealed reaction mixture to 60 degrees Celsius, monitor progress via TLC, and upon completion, purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free organocatalytic process translates into tangible economic and logistical benefits that extend far beyond the laboratory bench. The most immediate impact is seen in the reduction of raw material costs associated with catalyst procurement. Noble metals like rhodium and ruthenium are subject to volatile market pricing and geopolitical supply risks, whereas the organic ligands and additives used in this process are derived from abundant feedstocks and can be synthesized at a fraction of the cost. Moreover, the elimination of heavy metals from the process stream removes the necessity for costly metal scavenging resins and the associated waste disposal fees, leading to a significantly leaner cost structure for the final intermediate. This cost efficiency is compounded by the operational simplicity of the reaction, which does not require high-pressure equipment or cryogenic conditions, thereby reducing energy consumption and capital expenditure on specialized reactor infrastructure.
- Cost Reduction in Manufacturing: The transition to a non-metal catalytic system fundamentally alters the cost equation for producing high-purity pharmaceutical intermediates. By avoiding the use of expensive transition metal catalysts, manufacturers can bypass the substantial expenses linked to metal recovery and the rigorous analytical testing required to certify low residual metal levels in the final product. This streamlined approach reduces the number of unit operations in the production line, directly lowering labor and utility costs while increasing the overall batch throughput. Furthermore, the stability of the potassium trifluoroborate reagents allows for bulk purchasing and long-term storage without degradation, enabling better inventory management and hedging against raw material price fluctuations.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of chemically stable and commercially available reagents. Unlike sensitive organometallic complexes that may have long lead times or require cold-chain logistics, the components of this catalytic system are robust and can be sourced from multiple global suppliers. This diversification of the supply base mitigates the risk of production stoppages due to single-source failures. Additionally, the mild reaction conditions and tolerance to various functional groups mean that the process is less prone to batch failures caused by minor variations in raw material quality, ensuring a consistent and reliable flow of critical intermediates to downstream API manufacturing sites.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden bottlenecks related to heat transfer and safety, particularly when dealing with exothermic metal-catalyzed reactions. This organocatalytic method operates under mild thermal conditions with manageable exotherms, making it inherently safer and easier to scale from pilot plant to commercial production volumes. From an environmental standpoint, the absence of toxic heavy metals simplifies wastewater treatment and waste solid disposal, helping facilities maintain compliance with increasingly stringent environmental regulations. The use of recyclable molecular sieves and the potential for solvent recovery further enhance the green chemistry profile of the process, aligning with corporate sustainability goals and reducing the carbon footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the applicability of this technology, we have compiled answers to common inquiries regarding the reaction scope, catalyst loading, and purification requirements. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, providing a realistic overview of what can be achieved in a production setting. Understanding these nuances is critical for project planning and risk assessment when integrating new synthetic routes into existing manufacturing portfolios.
Q: What are the primary advantages of using potassium organotrifluoroborates over traditional boronic acids in this synthesis?
A: Potassium organotrifluoroborates offer superior stability against oxidation and hydrolysis compared to boronic acids, allowing for broader substrate compatibility and easier handling in large-scale pharmaceutical intermediate manufacturing without stringent inert atmosphere requirements during storage.
Q: How does the metal-free nature of this process impact downstream purification costs?
A: By eliminating transition metal catalysts such as rhodium or ruthenium, the process removes the need for expensive and complex heavy metal scavenging steps, significantly simplifying the workup procedure and ensuring the final active pharmaceutical ingredient meets strict residual metal limits.
Q: Can this catalytic system accommodate heterocyclic substrates commonly found in drug discovery?
A: Yes, the optimized catalyst system utilizing polyfluorinated binaphthol derivatives demonstrates excellent tolerance for various heterocycles including thiophenes, furans, and indoles, making it highly suitable for synthesizing complex scaffolds required in modern medicinal chemistry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Beta-Aminoketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalytic technologies in accelerating drug development timelines. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN109748841B can be seamlessly translated into robust industrial processes. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of complex chiral building blocks or reliable supply of key intermediates for your pipeline, our infrastructure is designed to support your growth from clinical trials to market launch.
We invite you to engage with our technical procurement team to discuss how this metal-free synthesis route can optimize your supply chain and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your project. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver high-quality chiral beta-aminoketone derivatives that adhere to the highest industry standards.
