Scalable Production of Stable D3-Flualprazolam for High-Precision Forensic Analysis
Scalable Production of Stable D3-Flualprazolam for High-Precision Forensic Analysis
The rapid emergence of novel psychoactive substances, often referred to as designer drugs, presents a formidable challenge to global forensic toxicology laboratories, necessitating the development of robust and reliable analytical standards. Addressing this critical gap, the Chinese patent CN115232133A, published on October 25, 2022, discloses a groundbreaking preparation method for D3-Flualprazolam, a deuterated analog essential for precise quantitative analysis. This innovation specifically targets the instability issues plaguing previous deuterated benzodiazepines by strategically positioning deuterium atoms on the triazole methyl group rather than the labile methylene bridge. For procurement specialists and R&D directors in the forensic and pharmaceutical sectors, this patent represents a significant leap forward in securing a stable supply of high-purity reference materials that are crucial for legal evidence and regulatory compliance.
As a leading manufacturer in the fine chemical industry, we recognize that the ability to synthesize such complex isotopically labeled compounds with high fidelity is a key differentiator in the market. The methodology outlined in this patent not only solves the scientific problem of deuterium loss but also offers a streamlined synthetic pathway that is amenable to industrial scale-up. By leveraging this intellectual property, we can offer our global clientele a superior product that ensures accuracy in mass spectrometry and NMR-based detection methods, thereby supporting the broader mission of public safety and drug control enforcement.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of deuterated benzodiazepines often suffered from significant structural vulnerabilities that compromised their utility as internal standards in rigorous analytical environments. Specifically, earlier patents, including those filed by the same applicants around 2013, typically located the deuterium label on the methylene group adjacent to the triazole ring. This positioning proved chemically precarious because the hydrogen-deuterium exchange at this specific site occurs readily in the presence of moisture or protic solvents, leading to the gradual loss of the isotopic label over time. Such instability renders the reference material unreliable for long-term storage and repeated use, forcing laboratories to frequently recalibrate or source fresh batches, which drives up operational costs and introduces variability into forensic data.
Furthermore, conventional detection methods for flualprazolam often rely on complex separation and purification protocols prior to structural confirmation using techniques like GC-MS or LC-QTOF. These traditional workflows are not only time-consuming and labor-intensive but also require sophisticated instrumentation that may not be universally available in all regional testing facilities. The lack of a stable, commercially viable deuterated internal standard has historically hindered the standardization of flualprazolam detection, creating a bottleneck in the efficient identification and quantification of this controlled substance in biological samples and seized materials.
The Novel Approach
The novel approach detailed in patent CN115232133A fundamentally reengineers the molecular architecture of the deuterated standard to ensure maximum stability and analytical performance. By shifting the deuterium labeling to the methyl group attached to the triazole ring, the inventors have effectively eliminated the risk of isotopic exchange with environmental water, thereby guaranteeing the chemical integrity of the standard throughout its shelf life. This strategic modification is achieved through a concise two-step synthetic route that begins with a benzodiazepine precursor and proceeds through a thionation followed by a condensation cyclization with deuterated acetyl hydrazide. This route is not only chemically elegant but also operationally simple, avoiding the need for harsh conditions or exotic reagents that often complicate manufacturing processes.
From a commercial perspective, this new methodology drastically simplifies the production workflow, making it highly attractive for cost reduction in pharmaceutical intermediate manufacturing. The process yields a product with high chemical purity and stable properties, which can be conveniently used for the preparation of analytical standard solutions without the fear of degradation. For supply chain managers, this translates to a more reliable inventory of critical reagents, reducing the risk of stockouts and ensuring continuity in forensic testing operations. The robustness of this synthesis allows for consistent batch-to-batch quality, which is paramount when dealing with legal evidence where precision is non-negotiable.
Mechanistic Insights into Oxygen-Sulfur Exchange and Cyclization
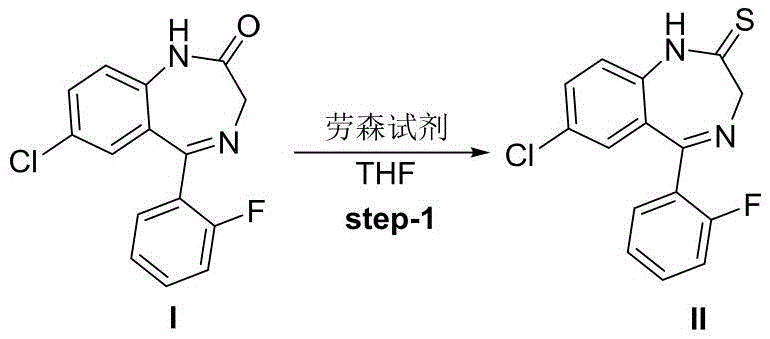
The core of this synthetic breakthrough lies in the precise execution of an oxygen-sulfur exchange reaction followed by a targeted condensation cyclization. In the first step, the carbonyl oxygen of the starting benzodiazepine (Compound I) is replaced by a sulfur atom using a specialized thionating agent. This transformation activates the molecule for the subsequent nucleophilic attack by the deuterated hydrazine derivative. The choice of reagent here is critical; while phosphorus pentasulfide or Japansese reagent could theoretically be used, the patent highlights Lawesson's reagent as the preferred catalyst due to its superior yield and selectivity profile. This thionation step generates the key thiobenzodiazepine intermediate (Compound II), which possesses the necessary electronic configuration to facilitate ring closure in the next stage.
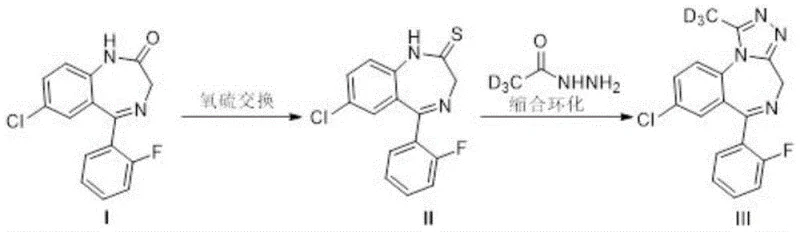
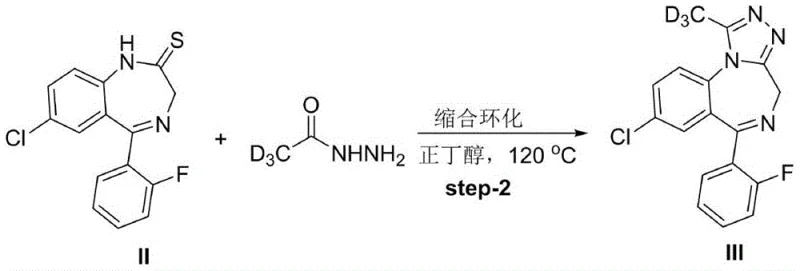
Following the formation of the thio-intermediate, the second step involves a condensation cyclization reaction with deuterated acetyl hydrazide. This reaction is conducted in a high-boiling solvent like n-butanol at elevated temperatures, typically around 120°C. Under these thermal conditions, the hydrazine moiety attacks the thiocarbonyl carbon, leading to the elimination of hydrogen sulfide and the formation of the triazole ring fused to the benzodiazepine core. Crucially, the deuterium atoms contained within the acetyl group of the hydrazine reagent are incorporated directly into the methyl position of the newly formed triazole ring. This specific mechanistic pathway ensures that the deuterium label is locked into a chemically inert environment, shielded from exchange reactions that would otherwise compromise the isotopic purity of the final D3-Flualprazolam product.
Impurity control is inherently managed through the specificity of these reaction conditions. The use of Lawesson's reagent minimizes side reactions that could lead to over-thionation or decomposition of the sensitive benzodiazepine scaffold. Furthermore, the cyclization step is monitored via TLC to ensure complete conversion, preventing the carryover of unreacted intermediates which could interfere with downstream analytical applications. The final purification via column chromatography effectively removes any residual solvent or minor byproducts, resulting in a final active pharmaceutical ingredient (API) intermediate that meets the stringent purity specifications required for forensic reference standards.
How to Synthesize D3-Flualprazolam Efficiently
To replicate the high yields and purity described in the patent, strict adherence to the optimized reaction parameters is essential. The process begins with the preparation of the thiobenzodiazepine intermediate, where the molar ratio of the starting benzodiazepine to Lawesson's reagent is carefully controlled at approximately 1:1.5. This stoichiometry ensures complete conversion while minimizing waste of the relatively expensive thionating agent. The reaction is typically carried out in tetrahydrofuran (THF) at room temperature after an initial cooling phase, providing a balance between reaction rate and safety. Following the thionation, the crude intermediate is carried forward without extensive purification, streamlining the workflow and reducing material loss.
- Perform oxygen-sulfur exchange on benzodiazepine I using Lawesson's reagent in THF to obtain thiobenzodiazepine II.
- React thiobenzodiazepine II with deuterated acetyl hydrazide in n-butanol at 120°C to induce condensation cyclization.
- Purify the crude reaction mixture via column chromatography to isolate high-purity D3-Flualprazolam III.
The subsequent cyclization step requires precise thermal management to drive the formation of the triazole ring. The crude thiobenzodiazepine is reacted with deuterated acetyl hydrazide in n-butanol, with the system heated to 120°C for approximately 8 hours. This specific solvent and temperature combination was identified as optimal, providing higher yields compared to alternatives like tert-butanol or lower temperatures. The use of n-butanol also facilitates the easy recovery of the solvent via reduced pressure distillation after the reaction is complete. The final isolation involves standard column chromatography, a scalable technique that ensures the removal of any trace impurities, delivering a product ready for certification as a reference standard.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible benefits that extend beyond mere technical feasibility. The primary advantage lies in the substantial cost savings achieved through the simplification of the manufacturing process. By eliminating the need for complex multi-step sequences or unstable intermediates that require cryogenic storage, the overall production cost is significantly reduced. The reagents employed, such as Lawesson's reagent and n-butanol, are commodity chemicals available from multiple global suppliers, which mitigates the risk of supply chain disruptions caused by single-source dependencies. This accessibility ensures a steady flow of raw materials, allowing for consistent production scheduling and reliable delivery timelines to end-users.
- Cost Reduction in Manufacturing: The streamlined two-step process eliminates the need for expensive transition metal catalysts or specialized equipment often required in alternative deuteration strategies. This reduction in capital expenditure and operational complexity translates directly into a more competitive pricing structure for the final deuterated standard. Furthermore, the high yield reported in the patent examples means that less raw material is wasted, improving the overall atom economy of the process. These efficiencies allow manufacturers to pass on savings to customers, making high-quality forensic standards more accessible to laboratories operating under tight budgetary constraints.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures that production can be scaled up rapidly to meet surges in demand without compromising quality. Since the process does not rely on fragile intermediates or extremely sensitive reaction conditions, it is less prone to batch failures, which are a common cause of supply delays in fine chemical manufacturing. The stability of the final D3-Flualprazolam product also reduces the logistical burden associated with cold-chain shipping and storage, allowing for more flexible distribution networks. This reliability is crucial for maintaining the continuity of forensic testing services, where the absence of a standard can halt critical investigations.
- Scalability and Environmental Compliance: The synthesis is designed with scalability in mind, utilizing standard batch reactors and workup procedures that are easily transferable from pilot scale to commercial production. The solvents used, such as THF and n-butanol, are well-understood in terms of waste management and can be efficiently recovered and recycled, aligning with modern green chemistry principles. This environmental compatibility reduces the regulatory burden on manufacturing facilities and minimizes the ecological footprint of the production process. For corporate buyers focused on sustainability goals, sourcing from a manufacturer utilizing this efficient and cleaner technology demonstrates a commitment to responsible chemical stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of D3-Flualprazolam based on the patented methodology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details helps stakeholders make informed decisions regarding the integration of this material into their analytical workflows and supply chains.
Q: Why is the deuterium position in this D3-Flualprazolam synthesis more stable than previous methods?
A: Unlike prior art where deuterium was placed on a methylene group prone to exchange with water, this patent places deuterium on the triazole methyl group, ensuring exceptional chemical stability during storage and analysis.
Q: What are the critical reaction conditions for the cyclization step?
A: The cyclization requires heating the thiobenzodiazepine intermediate with deuterated acetyl hydrazide in n-butanol at an optimized temperature of 120°C to maximize yield and minimize side reactions.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process utilizes readily available reagents like Lawesson's reagent and standard solvents like THF and n-butanol, avoiding expensive transition metal catalysts, which facilitates easy scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D3-Flualprazolam Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure required to bring this advanced synthesis route from the laboratory bench to full-scale commercial production. Our team of experienced chemists is adept at optimizing complex organic transformations, ensuring that every batch of D3-Flualprazolam meets the highest standards of isotopic purity and chemical stability. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to deliver consistent quality. Our rigorous QC labs employ advanced analytical techniques to verify the structural integrity of the deuterated label, guaranteeing that our products perform flawlessly in your forensic applications.
We invite you to collaborate with us to secure a stable and cost-effective supply of this critical reference material. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our manufacturing capabilities can support your operational goals. Let us help you enhance the precision and reliability of your forensic toxicology programs with our premium grade D3-Flualprazolam.
