Advanced Synthesis of 4-Heteroatom Cyclohexenyl Halides for Pharmaceutical Intermediates
Introduction to Next-Generation Intermediate Synthesis
The synthesis of 4-heteroatom substituted cyclohexenyl halides represents a critical bottleneck in the production of advanced pharmaceutical intermediates and fine chemicals. These structural motifs are indispensable precursors for coupling reactions that yield pharmaceutically active compounds, yet their preparation has historically been plagued by operational complexity. Patent CN102070586A introduces a groundbreaking methodology that fundamentally reshapes the manufacturing landscape for these valuable building blocks. By shifting away from cryogenic dependency towards a mild, hydrazone-based oxidative pathway, this technology offers a robust solution for cost reduction in pharmaceutical intermediates manufacturing. The process leverages accessible starting materials and standard reactor configurations, ensuring that high-purity cyclohexenyl halides can be produced with exceptional consistency. For R&D teams and supply chain leaders, this innovation translates directly into enhanced process reliability and reduced capital expenditure on specialized low-temperature infrastructure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the cyclohexenyl halide scaffold has relied heavily on harsh organometallic strategies that impose severe constraints on industrial scalability. The most prevalent literature methods utilize strong, non-nucleophilic bases such as Lithium Hexamethyldisilazide (LiHMDS) or Lithium Diisopropylamide (LDA) to generate enolates, which are subsequently trapped with electrophiles like PhNTf2. As illustrated in the reaction scheme below, these transformations mandate ultra-low temperature conditions, typically around -78 °C, to control regioselectivity and prevent decomposition.
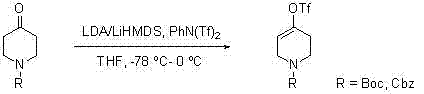
Maintaining such cryogenic environments on a multi-kilogram or tonnage scale requires significant energy input and specialized refrigeration equipment, drastically inflating operational costs. Furthermore, these routes invariably necessitate purification via silica gel column chromatography to isolate the desired product from side reactions and unreacted starting materials. Column chromatography is notoriously difficult to scale, often becoming the rate-limiting step in commercial production due to solvent consumption and throughput limitations. Alternative approaches involving phosphites and halogen gases also suffer from similar drawbacks, requiring temperatures as low as -60 °C and handling hazardous gaseous reagents, which introduces substantial safety risks and regulatory hurdles for commercial scale-up of complex heterocyclic intermediates.
The Novel Approach
In stark contrast to the legacy technologies, the patented process described in CN102070586A utilizes a温和 (mild) three-step sequence that operates within a manageable temperature window of -20 °C to 40 °C. This novel strategy initiates with the formation of a hydrazone intermediate from the corresponding 4-heteroatom substituted cyclohexanone, a transformation that proceeds efficiently under standard reflux or mild cooling conditions. The subsequent oxidative halogenation employs copper(II) salts (CuX2) in an alcoholic solvent, avoiding the need for super-strong bases or cryogenic cooling. This shift not only simplifies the reaction engineering but also enhances safety profiles by eliminating pyrophoric reagents. The final elimination step utilizes organic bases to drive the formation of the double bond, yielding the target halide with high fidelity. This approach effectively bypasses the need for column chromatography, relying instead on crystallization, distillation, or simple aqueous workups, thereby establishing a new benchmark for efficiency in pharmaceutical intermediates synthesis.
Mechanistic Insights into Hydrazone-Mediated Oxidative Halogenation
The core innovation of this synthesis lies in the copper-mediated oxidative decomposition of the hydrazone intermediate, which serves as a versatile precursor for introducing the halogen functionality. In the second step of the process, the hydrazone reacts with a mixture of CuX2 and a base in an alcohol solvent. Mechanistically, the copper species facilitates the oxidation of the hydrazone nitrogen, leading to the extrusion of nitrogen gas and the generation of a reactive carbocation or radical species that is immediately trapped by the halide anion. This step is highly tunable; by selecting specific copper salts such as CuCl2 or CuBr2, manufacturers can selectively introduce chlorine or bromine atoms at the 4-position. The reaction temperature is carefully controlled between -20 °C and 40 °C to balance reaction rate with selectivity, preventing over-halogenation or degradation of the sensitive heteroatom ring system.
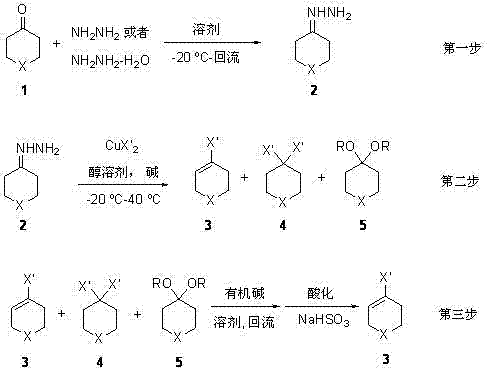
Following the oxidative step, the reaction mixture contains a complex array of species, including the desired mono-halide, di-halide byproducts, and trace amounts of ketals formed from the alcohol solvent. The brilliance of the third step lies in the selective elimination and hydrolysis protocol. By treating the crude mixture with an organic base under reflux, the di-halide species undergo dehydrohalogenation to converge towards the desired alkene structure, while the ketal impurities are hydrolyzed back to the ketone upon acidic workup. The addition of saturated sodium bisulfite (NaHSO3) in the final stage acts as a scavenger for residual oxidants and helps decompose remaining impurities, ensuring that the final isolated product achieves a purity exceeding 97%. This rigorous impurity control mechanism is vital for meeting the stringent specifications required by reliable pharmaceutical intermediates suppliers serving the global API market.
How to Synthesize 4-Heteroatom Cyclohexenyl Halides Efficiently
The implementation of this synthesis route is designed for seamless integration into existing chemical manufacturing facilities. The process begins with the condensation of the ketone and hydrazine, followed by the critical copper-mediated oxidation, and concludes with the base-promoted elimination. Each step utilizes common solvents such as methanol, ethanol, or toluene, and reagents that are commercially available in bulk quantities. The operational simplicity allows for direct scale-up from laboratory benchtop to pilot plant without the need for specialized cryogenic reactors. For detailed procedural parameters, stoichiometry, and specific workup instructions tailored to your specific substrate, please refer to the standardized synthesis guide provided below.
- React 4-heteroatom substituted cyclohexanone with hydrazine hydrate in solvent at -20°C to reflux to form the corresponding hydrazone intermediate.
- Oxidize the hydrazone using a CuX2/alkali mixture in alcohol solvent at -20°C to 40°C to generate a mixture of halide products.
- Treat the mixture with organic base under reflux, followed by acidification and NaHSO3 washing to isolate the pure 4-heteroatom cyclohexenyl halide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers profound strategic advantages beyond mere technical feasibility. The elimination of cryogenic requirements (-78 °C) translates directly into substantial reductions in utility costs and capital investment, as standard jacketed reactors can be utilized instead of specialized low-temperature vessels. Furthermore, the avoidance of column chromatography removes a major bottleneck in production throughput, allowing for continuous or large-batch processing that significantly shortens manufacturing cycles. This efficiency gain is crucial for reducing lead time for high-purity pharmaceutical intermediates, enabling faster response to market demands and clinical trial timelines.
- Cost Reduction in Manufacturing: The replacement of expensive, moisture-sensitive bases like LiHMDS with stable copper salts and hydrazine results in a drastic decrease in raw material costs. Additionally, the simplified purification workflow eliminates the massive solvent volumes associated with flash chromatography, leading to lower waste disposal fees and reduced solvent recovery costs. The overall process economics are optimized by maximizing atom economy and minimizing unit operations, delivering a highly competitive cost structure for bulk production.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as hydrazine hydrate, copper halides, and common alcohols ensures a robust and resilient supply chain. Unlike specialized organolithium reagents which may have long lead times or supply volatility, the inputs for this process are widely available from multiple global vendors. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages, ensuring consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, avoiding the safety hazards associated with handling large quantities of pyrophoric reagents or toxic halogen gases. The waste stream is easier to manage, primarily consisting of aqueous salt solutions and recoverable organic solvents, facilitating compliance with increasingly strict environmental regulations. The ability to produce >97% purity material via distillation or slurry rather than chromatography makes this route ideal for green chemistry initiatives and sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on yield expectations, purification methods, and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their current production portfolios.
Q: How does this new process improve upon traditional lithiation methods?
A: Traditional methods often require super-low temperatures (-78°C) and expensive bases like LiHMDS. This patented process operates between -20°C and 40°C using readily available copper salts, significantly reducing energy consumption and equipment costs.
Q: Is column chromatography required for purification?
A: No. A key advantage of this technology is the elimination of column chromatography. The final product is purified via distillation or slurry washing, achieving purity levels greater than 97%, which is critical for industrial scalability.
Q: What represents the overall yield and purity of the final halide?
A: According to the patent data, the three-step reaction sequence achieves a total yield ranging from 50% to 80%, with the final product consistently demonstrating a purity of over 97% as confirmed by GC analysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Heteroatom Cyclohexenyl Halide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN102070586A can be fully realized in a GMP-compliant environment. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of 4-heteroatom cyclohexenyl halide meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value and efficiency in your supply chain.
