Advanced Synthesis of Zofenopril Calcium: A Scalable Route for Global Pharmaceutical Manufacturing
Advanced Synthesis of Zofenopril Calcium: A Scalable Route for Global Pharmaceutical Manufacturing
The pharmaceutical landscape for cardiovascular therapeutics continues to demand robust, cost-effective, and high-purity manufacturing processes for critical active pharmaceutical ingredients (APIs). Zofenopril Calcium, a potent angiotensin-converting enzyme (ACE) inhibitor, stands out for its unique sulfhydryl group which offers distinct therapeutic benefits in treating hypertension and reducing mortality rates following acute myocardial infarction. However, the commercial viability of such complex molecules often hinges on the efficiency of their synthetic routes. A pivotal advancement in this domain is detailed in patent CN101012192A, which discloses a novel preparation method that addresses historical bottlenecks in yield and operational complexity. This technical insight report analyzes the proprietary methodology outlined in the patent, focusing on its transition from N-acetyl-L-hydroxyproline to the final calcium salt. By leveraging a streamlined sequence of esterification, tosylation, and strategic salt metathesis, this route promises to redefine the supply chain dynamics for this essential cardiovascular medication.
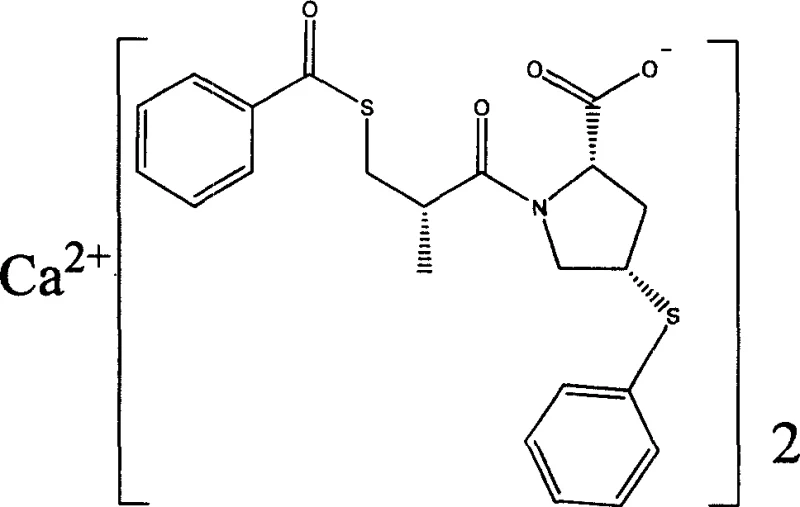
The structural integrity and stereochemical purity of Zofenopril Calcium are paramount for its biological efficacy, as depicted in the molecular visualization above. The patent emphasizes a process that not only secures the correct cis-configuration of the proline ring but also ensures the stability of the thioester linkage. For global procurement teams and R&D directors, understanding the nuances of this synthesis is critical. The method avoids the use of exotic reagents, relying instead on standard industrial solvents like ethanol, methanol, and ethyl acetate, which facilitates easier regulatory approval and supply chain continuity. Furthermore, the reported purity levels exceeding 99.0% suggest a significant reduction in downstream purification burdens, directly impacting the cost of goods sold (COGS) and making this route highly attractive for commercial scale-up of complex cardiovascular intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, including United States Patent 4316906 and Chinese Patent ZL99810517.1, has long established the baseline for Zofenopril synthesis, yet these methods suffer from inherent inefficiencies that hinder optimal industrial application. The background technology section of the subject patent explicitly highlights that existing schemes involve complicated operation procedures, particularly during the critical condensation reaction steps where the side chain is attached to the proline nucleus. These traditional routes often necessitate harsh reaction conditions or expensive protecting group strategies that result in lower overall yields and higher raw material costs. Additionally, the purification of the final product in older methods can be arduous, often requiring multiple chromatographic steps or difficult crystallizations to remove closely related impurities and diastereomers. For a procurement manager, these inefficiencies translate into volatile pricing and potential supply disruptions, as the margin for error in manufacturing is slim. The reliance on less accessible starting materials in some legacy processes further exacerbates the risk profile, making the supply chain fragile in the face of raw material shortages.
The Novel Approach
In stark contrast, the novel approach presented in CN101012192A introduces a rational design that prioritizes operational simplicity and raw material availability. The process initiates with N-acetyl-L-hydroxyproline, a commercially accessible chiral pool building block, thereby securing the stereochemistry from the outset. The innovation lies in the specific sequence of activating the hydroxyl group via tosylation followed by a clean nucleophilic substitution with thiophenol. This sequence effectively installs the phenylthio group with high fidelity. Crucially, the patent describes a unique salt formation strategy where the free acid is first converted to a potassium salt, purified via recrystallization, and subsequently converted to the calcium salt. This two-step salt formation acts as a powerful purification engine, scrubbing away impurities that might co-precipitate in a direct calcium salt formation. By breaking the synthesis down into manageable, high-yielding steps with clear work-up procedures involving standard extractions and filtrations, this method significantly lowers the barrier to entry for manufacturers seeking cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Chiral Proline Functionalization and Salt Metathesis
From a mechanistic perspective, the success of this synthesis relies heavily on the preservation of chirality and the controlled reactivity of the sulfur-containing intermediates. The initial esterification of N-acetyl-L-hydroxyproline serves a dual purpose: it protects the carboxylic acid from unwanted side reactions during the subsequent tosylation and activates the molecule for the nucleophilic displacement. The tosylation step converts the secondary hydroxyl group into a superior leaving group (tosylate), which is then displaced by the thiophenolate anion generated in situ from thiophenol and sodium ethoxide. This SN2-type substitution is critical as it proceeds with inversion of configuration if the center were chiral, but here it establishes the cis-4-substitution pattern relative to the nitrogen, which is thermodynamically favored and kinetically accessible under the described conditions. The subsequent hydrolysis of the ester and the N-acetyl group must be carefully controlled to prevent racemization, a risk mitigated by the mild alkaline conditions and controlled temperatures specified in the patent embodiments.
The final stages of the synthesis demonstrate sophisticated process chemistry designed to maximize purity. The condensation of the cis-4-phenylthio-L-proline hydrochloride with (S)-3-(benzoylthio)-2-methylpropionyl chloride is performed under Schotten-Baumann-like conditions, maintaining a pH of 7.5 to 8.5 to ensure the amine is nucleophilic enough to react while preventing hydrolysis of the acid chloride. The true brilliance, however, is observed in the salt exchange mechanism. By first forming the potassium salt in anhydrous alcohol and recrystallizing it, the process isolates the API in a highly pure crystalline form. The subsequent reaction with calcium chloride in hot water (90°C to 100°C) drives the metathesis equilibrium towards the insoluble calcium salt. This temperature-dependent solubility switch allows for the precipitation of the final product as a white crystalline powder, effectively excluding soluble inorganic salts and organic impurities. This mechanistic understanding assures R&D directors that the process is robust and capable of consistently delivering high-purity zofenopril calcium suitable for stringent regulatory filings.
How to Synthesize Zofenopril Calcium Efficiently
The synthesis of Zofenopril Calcium via this patented route involves a logical progression of functional group transformations that are well-suited for standard reactor setups. The process begins with the activation of the proline scaffold and concludes with a precise ion-exchange crystallization. Operators must pay close attention to pH control during the acylation step and temperature management during the final calcium salt precipitation to ensure the correct polymorph and particle size distribution. While the general workflow is straightforward, the specific stoichiometry and solvent choices detailed in the patent are critical for replicating the high yields reported in the examples. For a comprehensive understanding of the exact parameters required for GMP manufacturing, please refer to the standardized synthesis steps provided in the technical guide below.
- Esterify N-acetyl-L-hydroxyproline with methanol and perform tosylation to activate the hydroxyl group for substitution.
- Substitute the tosyl group with thiophenol under alkaline conditions to introduce the phenylthio moiety, followed by hydrolysis.
- Condense the resulting proline derivative with (S)-3-(benzoylthio)-2-methylpropionyl chloride and perform potassium-to-calcium salt exchange.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and supply continuity, the implications of adopting this synthesis route are profound. The shift towards a process that utilizes widely available starting materials like N-acetyl-L-hydroxyproline and common solvents significantly de-risks the supply chain. Unlike processes that rely on bespoke catalysts or rare reagents, this method leverages commodity chemicals that are readily sourced from multiple global suppliers, ensuring that production schedules are not held hostage by single-source bottlenecks. Furthermore, the operational simplicity—characterized by standard filtration, extraction, and distillation units—means that the technology can be transferred to existing manufacturing facilities with minimal capital expenditure on new equipment. This adaptability is a key driver for reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the elimination of complex purification steps and the improvement in overall yield. By avoiding the need for expensive chromatographic separations or cryogenic conditions often found in alternative syntheses, the operational expenditure (OPEX) is substantially lowered. The ability to recover and recycle solvents like ethanol and ethyl acetate, as implied by the standard work-up procedures, further contributes to waste minimization and cost efficiency. Additionally, the high yield in the final salt formation step ensures that valuable intermediates are not lost, maximizing the throughput of the production line and driving down the unit cost of the final API without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the chemical transformations described. The reactions are conducted at moderate temperatures (ranging from 0°C to 100°C) and atmospheric pressure, which reduces the safety risks associated with high-pressure hydrogenation or extreme thermal conditions. This safety profile translates to fewer unplanned shutdowns and maintenance issues. Moreover, the use of stable intermediates, such as the N-acetyl protected species, allows for potential campaign manufacturing where intermediates can be stockpiled if necessary, providing a buffer against upstream supply disruptions. This reliability makes the manufacturer a reliable zofenopril calcium supplier capable of meeting long-term contractual obligations with multinational pharmaceutical partners.
- Scalability and Environmental Compliance: The patent data explicitly demonstrates the scalability of the process through embodiments that range from small laboratory scales to near-kilogram batches, indicating a clear path to ton-scale production. The aqueous work-ups and the absence of heavy metal catalysts simplify the wastewater treatment protocols, aligning the process with increasingly strict environmental regulations. The final product is isolated as a stable solid, which simplifies packaging, storage, and transportation logistics. This ease of scale-up ensures that as demand for cardiovascular therapies grows, the manufacturing capacity can be expanded seamlessly, supporting the commercial scale-up of complex cardiovascular intermediates without the typical teething problems associated with process intensification.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Zofenopril Calcium using this advanced methodology. These answers are derived directly from the experimental data and claims within the patent literature, providing a transparent view of the process capabilities. Understanding these details helps potential partners assess the feasibility of integrating this supply source into their global procurement strategy.
Q: What is the primary advantage of this new Zofenopril Calcium synthesis route?
A: The primary advantage is the significant simplification of the condensation reaction and the use of widely available raw materials like N-acetyl-L-hydroxyproline, which drastically reduces production costs compared to older patented methods.
Q: How is high purity (>99%) achieved in this process?
A: High purity is achieved through a specific intermediate purification strategy involving the formation of a potassium salt followed by recrystallization with isopropanol, before the final conversion to the calcium salt.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability with embodiments ranging from gram to nearly kilogram scales, utilizing standard solvents and moderate temperature conditions that are easily adaptable to reactor vessels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Zofenopril Calcium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires more than just chemical knowledge; it demands engineering excellence and unwavering quality commitment. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles described in CN101012192A are realized in our manufacturing plants. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch of Zofenopril Calcium against the highest international pharmacopoeia standards. Our commitment to quality ensures that the material you receive is not only chemically pure but also consistent in its physical properties, which is vital for downstream formulation.
We invite global pharmaceutical companies and procurement leaders to engage with us to explore how this optimized synthesis route can benefit your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this manufacturing method. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your volume requirements. Let us collaborate to secure a sustainable and cost-effective supply of this critical cardiovascular medication for patients worldwide.
