Advanced Synthesis of Benzoisofuran-Based NON Ligands for Asymmetric Catalysis
Advanced Synthesis of Benzoisofuran-Based NON Ligands for Asymmetric Catalysis
The landscape of asymmetric catalysis is continuously evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. A significant breakthrough in this domain is documented in Chinese patent CN111233852A, which introduces a novel class of NON-type chiral bisoxazoline ligands featuring a unique benzoisofuran scaffold. This innovation addresses critical limitations found in existing ligand families by providing a robust, scalable, and highly selective platform for asymmetric transformations. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, understanding the structural advantages and synthetic accessibility of these ligands is paramount. The patent details a comprehensive methodology that transforms simple o-iodobenzoic acid derivatives into sophisticated chiral catalysts capable of inducing high stereocontrol in challenging fluorination reactions.
The strategic value of this technology lies not only in its catalytic performance but also in its manufacturability. Unlike many specialized ligands that require exotic starting materials or cryogenic conditions throughout the entire sequence, this process utilizes commercially accessible reagents and operates under relatively mild conditions. This balance between structural complexity and synthetic practicality makes it an attractive candidate for integration into existing supply chains focused on cost reduction in fine chemical manufacturing. By leveraging this intellectual property, manufacturers can potentially access new chemical space for drug discovery while maintaining rigorous control over production costs and timelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of NON-type oxazoline ligands has been dominated by scaffolds such as DBFOX and XABOX, which, while effective, present distinct challenges for widespread industrial adoption. These traditional ligands often suffer from a narrow substrate scope, meaning they are highly specialized for specific reaction types but fail to deliver consistent results across diverse chemical transformations. Furthermore, the synthesis of these conventional frameworks frequently involves harsh reaction conditions, including extreme temperatures or the use of hazardous reagents that complicate waste management and safety protocols. From a supply chain perspective, the reliance on expensive or difficult-to-source precursors for these legacy ligands creates bottlenecks that can delay project timelines and inflate the cost of goods sold for downstream API manufacturers.
The Novel Approach
The methodology outlined in CN111233852A represents a paradigm shift by introducing a benzoisofuran-based backbone that combines rigidity with tunable steric properties. This novel approach utilizes a modular synthetic strategy where the core skeleton is constructed through a series of efficient coupling and cyclization reactions. The resulting ligands are not only easier to synthesize but also demonstrate superior stability, resisting degradation upon exposure to air, which is a common failure point for many sensitive chiral phosphines or amines. This durability translates directly into operational efficiency, reducing the need for inert atmosphere handling during storage and formulation. For companies aiming for the commercial scale-up of complex catalysts, this robustness is a decisive factor that lowers the barrier to entry for implementing asymmetric fluorination technologies in their processes.
Mechanistic Insights into Zn-Catalyzed Asymmetric Fluorination
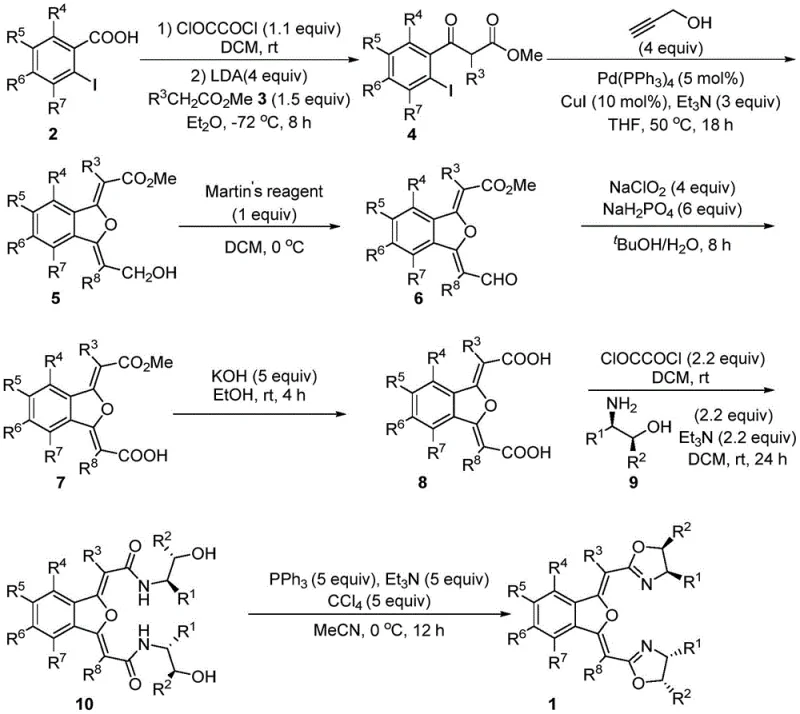
The core utility of these NON-type ligands is exemplified in their application for the asymmetric fluorination of β-ketoesters, a transformation of high value in medicinal chemistry for introducing fluorine atoms into bioactive molecules. The mechanism involves the formation of a chiral Lewis acid complex, typically utilizing Zinc bis(trifluoromethanesulfonyl)imide [Zn(NTf2)2] as the metal center. Upon coordination with the bisoxazoline ligand, the zinc ion creates a chiral environment that activates the β-ketoester substrate towards nucleophilic attack by the fluorinating agent, N-fluorobenzenesulfonimide (NFSI). The rigid benzoisofuran scaffold plays a crucial role in defining the chiral pocket, ensuring that the fluorine atom is delivered to only one face of the enolate intermediate with high fidelity.
Experimental data from the patent indicates that this catalytic system achieves remarkable enantiomeric excess (ee) values, reaching up to 92% for certain substrates under optimized conditions. The reaction proceeds smoothly at room temperature in solvents like dichloromethane, avoiding the energy-intensive cooling or heating cycles often required by less active catalysts. This mechanistic efficiency is critical for high-purity pharmaceutical intermediates where impurity profiles must be tightly controlled. The ability to achieve high conversion and selectivity simultaneously minimizes the need for costly downstream purification steps, such as chiral chromatography, thereby streamlining the overall production workflow and enhancing the economic viability of the process.
How to Synthesize NON-Type Chiral Bisoxazoline Ligand Efficiently
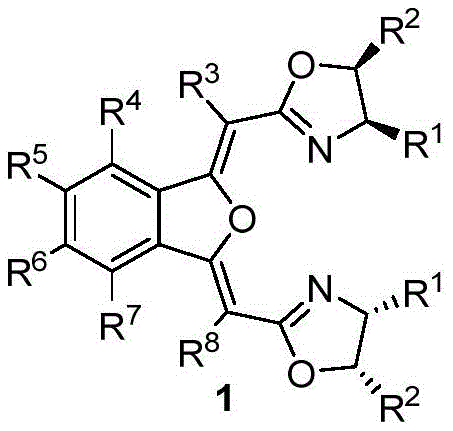
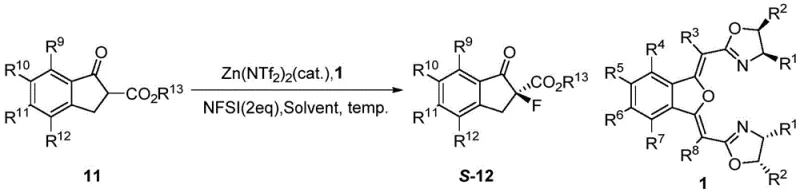
The synthesis of these advanced ligands is a testament to modern organic process chemistry, balancing step economy with high yield. The route begins with the activation of o-iodobenzoic acid followed by a low-temperature Claisen condensation to establish the carbon framework. Subsequent palladium-copper catalyzed cyclization constructs the benzoisofuran core, which is then functionalized through oxidation and hydrolysis steps to reveal the dicarboxylic acid motif. The final stages involve amidation with chiral amino alcohols and a cyclodehydration step to close the oxazoline rings. This logical progression allows for quality control checkpoints at each intermediate stage, ensuring that the final ligand meets stringent purity specifications required for catalytic applications. Detailed standardized synthesis steps are provided in the guide below.
- React o-iodobenzoic acid with oxalyl chloride to form acid chloride, then perform Claisen condensation with ester 3 using LDA at -72°C to yield intermediate 4.
- Execute Pd/Cu-catalyzed cyclization of intermediate 4 with propargyl alcohol to form the benzoisofuran core (intermediate 5), followed by oxidation to aldehyde 6.
- Oxidize aldehyde 6 to carboxylic acid 7, hydrolyze to diacid 8, couple with chiral amino alcohol 9 to form diamide 10, and finally cyclodehydrate to obtain ligand 1.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel ligand technology offers tangible benefits beyond mere technical performance. The synthesis route relies on commodity chemicals such as o-iodobenzoic acid, methyl acetate, and propargyl alcohol, which are readily available in the global market at stable prices. This reliance on bulk chemicals rather than bespoke fine chemicals significantly de-risks the supply chain against volatility. Furthermore, the reaction conditions described in the patent, such as ambient temperature cyclizations and standard workup procedures, are compatible with existing multipurpose reactor infrastructure, eliminating the need for capital-intensive equipment upgrades. These factors collectively contribute to a more resilient and cost-effective sourcing strategy for chiral catalysts.
- Cost Reduction in Manufacturing: The elimination of expensive transition metals in the ligand structure itself, combined with the use of earth-abundant zinc for catalysis, drastically reduces the raw material cost profile. Additionally, the high stability of the ligand means reduced waste due to degradation during storage and transport. The synthetic route avoids cryogenic conditions for most steps, leading to substantial energy savings compared to processes requiring sustained ultra-low temperatures. These efficiencies compound over large production volumes, offering a competitive pricing structure for end-users seeking to optimize their bill of materials without sacrificing quality.
- Enhanced Supply Chain Reliability: The robustness of the benzoisofuran scaffold ensures that the ligand maintains its integrity over extended periods, simplifying inventory management and reducing the frequency of re-ordering. Since the starting materials are common industrial chemicals, the risk of supply disruption due to raw material scarcity is minimized. This reliability is crucial for maintaining continuous production schedules in API manufacturing, where downtime can result in significant financial losses. The ability to source these ligands from a reliable pharmaceutical intermediate supplier with a proven synthesis track record further secures the supply chain against external shocks.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard organic solvents like dichloromethane, ethyl acetate, and ethanol that are easily recovered and recycled in industrial settings. The avoidance of heavy metal catalysts in the ligand synthesis phase simplifies waste treatment and regulatory compliance regarding metal residues in the final product. This environmental compatibility aligns with modern green chemistry principles and corporate sustainability goals. The high yields reported in the patent examples suggest that the process is efficient, minimizing the generation of byproduct waste and maximizing atom economy, which is a key metric for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ligand technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps R&D and procurement teams evaluate the fit of this technology within their specific project requirements. It clarifies the operational parameters and potential limitations, ensuring realistic expectations for performance and integration.
Q: What are the stability characteristics of the novel NON-type ligand?
A: According to patent CN111233852A, the synthesized ligand exhibits excellent stability in air and does not easily deteriorate, allowing for long-term storage without significant loss of catalytic activity.
Q: What is the primary catalytic application of this ligand?
A: The ligand is specifically designed for the asymmetric fluorination of β-ketoesters, demonstrating high enantioselectivity (up to 92% ee) when complexed with Zinc salts.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the patent highlights that the method uses cheap and readily available raw materials with mild reaction conditions and high yields, making it highly suitable for large-scale commercial synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable NON-Type Chiral Bisoxazoline Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced chiral ligands play in accelerating drug discovery and process development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ligand meets the high standards required for asymmetric catalysis. Our commitment to quality assurance means that our clients can rely on consistent performance, batch after batch, minimizing the risk of failed reactions due to reagent variability.
We invite you to engage with our technical procurement team to discuss how this novel ligand technology can be integrated into your specific synthesis campaigns. We offer a Customized Cost-Saving Analysis to help you quantify the potential economic benefits of switching to this more efficient catalytic system. Please contact us to request specific COA data and route feasibility assessments tailored to your target molecules. By partnering with us, you gain access to cutting-edge chemical technology backed by a supply chain dedicated to reliability, speed, and technical excellence.
