Advanced C4-Functionalized Pybox Ligands for Asymmetric Catalysis and Commercial Scale-Up
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the innovations detailed in patent CN110305122B, which introduces a novel class of pyridine ring C4-position sulfonyl and phosphoroxy functionalized Pybox ligands. This technological breakthrough addresses long-standing limitations in chiral ligand design by expanding the electronic and steric tuning capabilities available to process chemists. Historically, the modification of Pybox ligands was largely confined to halogen, oxygen, or nitrogen substituents at the C4 position of the pyridine core, which restricted the fine-tuning of catalytic activity for complex transformations. The disclosed invention leverages sulfonyl and phosphorus-oxygen functional groups to create ligands with superior stability and catalytic performance, offering a robust solution for the synthesis of high-value pharmaceutical intermediates. By utilizing simple and easily obtainable raw materials like 4-bromo-2,6-dimethyl pyridine carboxylate, this method streamlines the production workflow while maintaining rigorous standards for enantioselectivity. For R&D directors and procurement specialists, this represents a pivotal shift towards more efficient and cost-effective chiral catalyst sourcing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing structurally diverse Pybox ligands have predominantly relied on modifying the oxazole ring or introducing simple halogen substituents at the C4 position of the pyridine ring. While these methods established the foundation for asymmetric hydrosilylation and cyclopropanation reactions, they often suffer from restricted electronic tunability and limited scope in accommodating diverse substrate profiles. The reliance on halogenated intermediates frequently necessitates harsh reaction conditions and complex purification steps to remove metal residues, which can be detrimental to sensitive downstream processes in API manufacturing. Furthermore, the existing library of C4-modified ligands lacks the electronic diversity required to optimize reactions involving sterically demanding substrates, leading to suboptimal yields and enantiomeric excess in many industrial applications. This stagnation in ligand development has created a bottleneck for process chemists seeking to enhance the efficiency of chiral synthesis routes without incurring prohibitive costs or supply chain risks associated with exotic reagents.
The Novel Approach
The innovative methodology presented in the patent data overcomes these historical constraints by introducing sulfonyl and phosphoroxy functional groups directly onto the C4 position of the pyridine ring. This strategic modification significantly alters the electron-withdrawing properties of the ligand backbone, thereby enhancing the Lewis acidity of the coordinated metal center and improving asymmetric induction. The synthesis route is remarkably straightforward, proceeding through a three-step sequence that avoids the use of expensive transition metal catalysts in the initial functionalization stage when creating sulfonyl variants. By employing readily available sodium sulfonate salts or phosphorus-hydrogen compounds, the process ensures a stable supply of critical raw materials while simplifying the operational complexity typically associated with ligand synthesis. 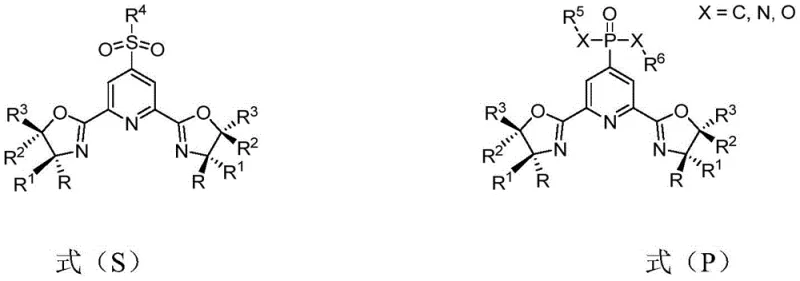
Mechanistic Insights into Sulfonyl and Phosphoroxy Functionalized Pybox Catalysis
The enhanced performance of these novel ligands stems from the precise coordination geometry established between the tridentate Pybox framework and the central metal ion, such as copper or rhodium. The lone pair electrons on the nitrogen atoms of the pyridine and oxazoline rings form a rigid chelate that locks the metal into a specific chiral environment, crucial for high enantioselectivity. The introduction of the sulfonyl or phosphoroxy group at the C4 position exerts a strong electronic influence that modulates the electrophilicity of the metal center, facilitating faster reaction rates and tighter transition state control. This electronic tuning is particularly beneficial for asymmetric CuAAC reactions, where the ligand must balance reactivity with stereocontrol to prevent the formation of racemic byproducts. The steric bulk provided by the substituents on the oxazoline rings further shields the active site, ensuring that substrate approach occurs only from the desired trajectory. 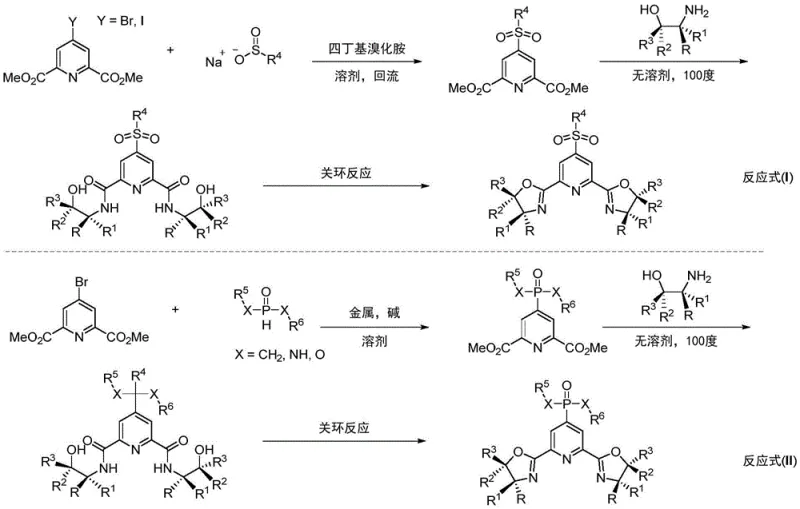
Impurity control is inherently built into the design of this synthesis pathway, as the mild reaction conditions minimize the degradation of sensitive functional groups. The use of DMF as a solvent in the initial sulfonylation step significantly improves yield compared to traditional acetonitrile-based systems, reducing the burden on downstream purification units. The subsequent ammonolysis and cyclization steps are conducted under solvent-free or low-temperature conditions, which suppresses side reactions such as epimerization or hydrolysis that could compromise the optical purity of the final product. This focus on process cleanliness ensures that the resulting ligands meet the stringent quality specifications required for GMP-compliant pharmaceutical manufacturing. For supply chain managers, this translates to reduced waste generation and lower environmental compliance costs, aligning with modern sustainability goals in fine chemical production.
How to Synthesize C4-Sulfonyl Pybox Ligand Efficiently
The synthesis of these high-performance ligands follows a logical three-step progression designed for scalability and reproducibility in an industrial setting. The process begins with the nucleophilic substitution of a halogenated pyridine ester, followed by amidation with chiral amino alcohols, and concludes with a dehydration cyclization to form the oxazoline rings. Detailed standard operating procedures regarding stoichiometry, temperature gradients, and workup protocols are essential for maintaining batch-to-batch consistency.
- React 4-bromo-2,6-dimethyl pyridine carboxylate with sodium sulfonate in DMF at 80°C to form the sulfonyl intermediate.
- Perform solvent-free ammonolysis with chiral amino alcohol at 100°C to generate the diamide precursor.
- Execute ring closure using DAST reagent at -20°C to finalize the chiral Pybox ligand structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers substantial advantages in terms of cost structure and supply chain resilience for manufacturers of chiral intermediates. The reliance on commodity chemicals like 4-bromo-2,6-dimethyl pyridine carboxylate eliminates the dependency on bespoke starting materials that often suffer from long lead times and price volatility. The elimination of expensive transition metal catalysts in the initial functionalization step for sulfonyl ligands directly reduces the raw material cost base, allowing for more competitive pricing models in the global market. Additionally, the high yields reported in the patent examples indicate a robust process that maximizes material throughput, minimizing the financial impact of lost batches or reprocessing requirements. 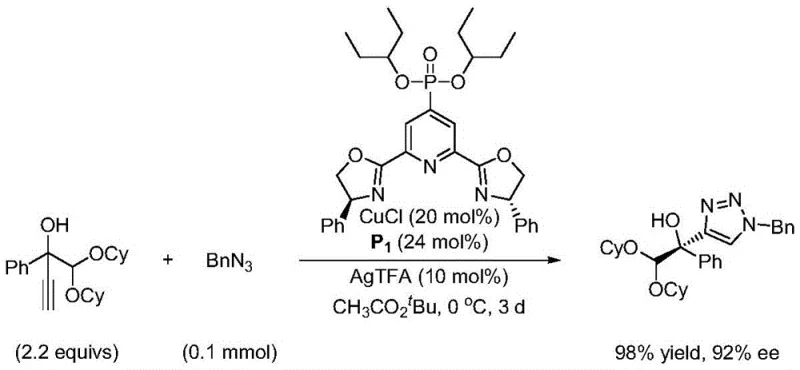
- Cost Reduction in Manufacturing: The streamlined three-step synthesis significantly lowers operational expenditures by reducing solvent consumption and energy requirements associated with prolonged heating or cryogenic cooling. By avoiding the use of precious metal catalysts in the primary bond-forming steps, the process removes the need for costly metal scavenging operations, which are often a major expense in fine chemical manufacturing. The high atom economy of the reaction ensures that a greater proportion of input materials are converted into valuable product, thereby reducing waste disposal costs and improving overall process efficiency. These cumulative savings contribute to a more favorable cost of goods sold, enabling manufacturers to maintain healthy margins even in competitive bidding scenarios.
- Enhanced Supply Chain Reliability: The utilization of widely available industrial reagents ensures that production schedules are not disrupted by shortages of specialized chemicals. The robustness of the reaction conditions allows for flexibility in sourcing, as alternative suppliers for common solvents and bases can be qualified without compromising product quality. This diversification of the supply base mitigates the risk of single-source dependency, which is critical for maintaining continuous operations in the face of global logistical challenges. Furthermore, the stability of the intermediate compounds allows for potential stockpiling strategies, providing a buffer against unexpected demand surges or transportation delays.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of hazardous reagents in key steps facilitate a smoother transition from laboratory scale to commercial production volumes. The process generates minimal hazardous waste, simplifying compliance with increasingly stringent environmental regulations and reducing the administrative burden associated with waste management. The ability to operate under solvent-free conditions during the ammonolysis step further enhances the green chemistry profile of the manufacturing process. These factors collectively support sustainable growth and enable companies to meet corporate social responsibility targets while delivering high-quality chiral building blocks to the market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel ligand technology in industrial processes. The answers are derived directly from the experimental data and beneficial effects described in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this chemistry into existing production lines.
Q: What distinguishes this Pybox ligand from conventional C4-modified variants?
A: Unlike traditional ligands limited to halogen or oxygen substitution at the C4 position, this patent introduces sulfonyl and phosphoroxy functional groups, significantly altering electronic properties and enhancing asymmetric induction capabilities.
Q: Are the starting materials for this synthesis readily available for large-scale production?
A: Yes, the process utilizes 4-bromo-2,6-dimethyl pyridine carboxylate, a commercially accessible raw material, avoiding complex multi-step precursor synthesis and facilitating easier supply chain management.
Q: How does the new synthesis method impact impurity profiles?
A: The optimized solvent systems and mild reaction conditions minimize side reactions, resulting in cleaner crude products that require less intensive purification, thereby improving overall process efficiency and yield consistency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pybox Ligand Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chiral technology commercialization, leveraging deep expertise to translate complex academic innovations like patent CN110305122B into reliable industrial realities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Pybox ligand meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance means that clients can trust our materials to perform consistently in their critical asymmetric synthesis campaigns.
We invite forward-thinking organizations to collaborate with us on optimizing their chiral synthesis routes using these advanced ligand systems. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our solutions can enhance your operational efficiency. Let us help you secure a competitive advantage through superior catalyst technology and supply chain reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
