Advanced Manufacturing of Diazaspiro Lactam Intermediates for NMDA Modulators
The pharmaceutical industry continuously seeks robust and scalable pathways for complex molecular scaffolds, particularly those targeting central nervous system disorders. Patent CN114269338A introduces a significant advancement in the preparation of diazaspiro lactam compounds, which serve as critical intermediates for NMDA receptor modulators. These modulators are pivotal in addressing synaptic plasticity issues related to memory acquisition and pain perception. The disclosed technology provides a novel process that overcomes the economic and technical hurdles often associated with constructing spirocyclic lactam cores. By leveraging specific coupling strategies, this method ensures higher efficiency and purity, addressing the growing demand for reliable pharmaceutical intermediate supplier capabilities in the neurology sector. The innovation lies not just in the final molecule but in the streamlined connectivity of the building blocks.
Traditional synthetic routes for spirocyclic structures often suffer from excessive step counts, harsh reaction conditions, and difficult purification profiles that hinder commercial viability. Conventional methods may rely on multiple protection and deprotection sequences that introduce unnecessary waste and extend lead times significantly. Furthermore, older methodologies might struggle with stereochemical control, leading to impurity profiles that are unacceptable for modern regulatory standards. In contrast, the novel approach detailed in the patent utilizes a direct coupling strategy that minimizes intermediate isolation steps. This reduction in operational complexity translates directly into cost reduction in pharma manufacturing by lowering solvent consumption and labor hours. The new pathway avoids the pitfalls of low-yielding cyclization steps that have historically plagued this chemical class.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of diazaspiro frameworks has required convoluted sequences involving unstable intermediates and sensitive reagents. Many legacy processes depend on transition metal catalysts that require extensive removal protocols to meet heavy metal specifications for drug substances. These additional purification stages increase the overall cost of goods and complicate the supply chain logistics for procurement teams. Moreover, the use of cryogenic conditions in some traditional routes imposes significant energy burdens and equipment constraints on manufacturing facilities. The accumulation of byproducts in multi-step syntheses often necessitates chromatographic purification, which is notoriously difficult to scale beyond kilogram quantities. These factors collectively create bottlenecks that delay the availability of high-purity pharmaceutical intermediates for clinical and commercial programs.
The Novel Approach
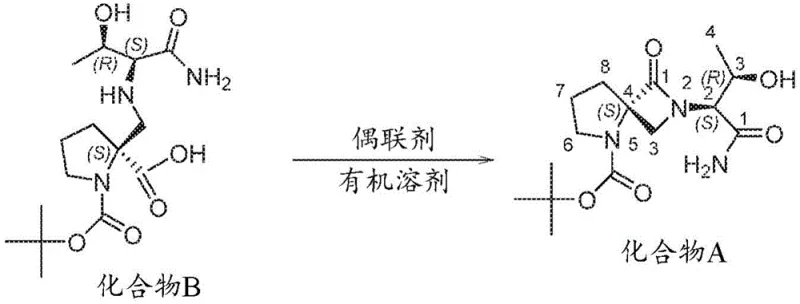
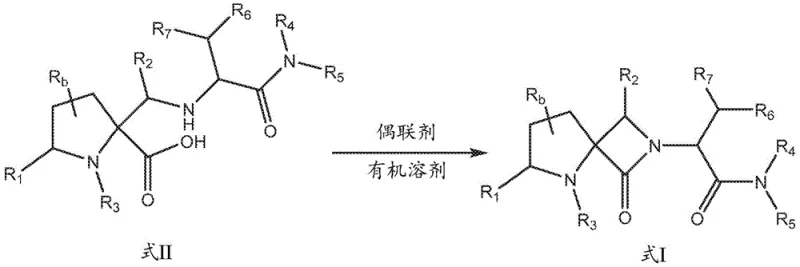
The patented process revolutionizes this landscape by employing a highly efficient coupling agent-mediated cyclization to form the core lactam structure. By contacting a compound of Formula II with a coupling agent in an organic solvent, the reaction proceeds smoothly to obtain the target compound of Formula I. This methodology allows for the use of widely available reagents such as carbodiimides or phosphonium salts, which are familiar to process chemists and easily sourced globally. The reaction conditions are mild enough to preserve sensitive functional groups while driving the equilibrium toward the desired product. This strategic simplification of the synthetic route enhances supply chain reliability by reducing dependency on exotic or single-source reagents. Consequently, manufacturers can achieve consistent quality and throughput without compromising on safety or environmental compliance standards.
Mechanistic Insights into Coupling-Mediated Cyclization
The core of this technological breakthrough involves the activation of the carboxylic acid moiety within the precursor to facilitate intramolecular or intermolecular amide bond formation. The coupling agent acts as a dehydrating species that generates a reactive intermediate, such as an O-acylisourea or active ester, which is then attacked by the amine nucleophile. This mechanism is carefully tuned to prevent racemization at chiral centers, ensuring the optical purity required for biological activity. The selection of the solvent plays a crucial role in stabilizing the transition state and solubilizing the reactants without interfering with the coupling efficiency. Common solvents like THF or DCM are preferred due to their ability to dissolve both organic substrates and coupling reagents effectively. Understanding this mechanistic nuance is essential for R&D directors aiming to replicate or adapt this chemistry for analogous structures.
Impurity control is inherently built into the design of this reaction pathway through the careful selection of reagents and stoichiometry. The use of specific bases, such as triethylamine or DIPEA, helps to scavenge acid byproducts generated during the coupling process, preventing side reactions like epimerization. Furthermore, the process allows for the formation of the spirocyclic ring with high diastereoselectivity, minimizing the formation of hard-to-remove stereoisomers. The downstream workup procedures described involve standard aqueous extractions and crystallizations, which are highly effective at purging residual reagents and soluble impurities. This focus on impurity management ensures that the final isolated compound meets stringent purity specifications without requiring resource-intensive chromatography. For quality assurance teams, this predictability in the impurity profile is a major advantage for regulatory filings and validation batches.
How to Synthesize Diazaspiro Lactam Efficiently
The synthesis of these complex intermediates follows a logical progression starting from readily available proline derivatives or similar cyclic amino acids. The initial steps involve the formation of key precursors like Formula IX through condensation with aldehydes, followed by functionalization to generate the coupling substrate. Detailed standardized synthesis steps see the guide below which outlines the precise order of addition and temperature controls necessary for success. Maintaining strict temperature profiles during the deprotonation and coupling phases is critical to maximizing yield and minimizing degradation. Operators must ensure that moisture levels are controlled, as water can hydrolyze the activated intermediates and reduce overall efficiency. Adherence to these procedural details guarantees the reproducibility needed for technology transfer between laboratory and plant scales.
- Prepare the precursor compound of Formula II by protecting the nitrogen on the five-membered ring using a dicarbonate or alkylating agent.
- Contact the compound of Formula II with a suitable coupling agent in an organic solvent to facilitate amide bond formation.
- Isolate the final compound of Formula I through standard workup procedures including extraction and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this process offers substantial benefits that resonate deeply with procurement managers and supply chain heads looking to optimize their vendor networks. The elimination of complex catalytic systems and the reliance on commodity chemicals significantly lowers the barrier to entry for manufacturing partners. This accessibility fosters a more competitive supply market, ensuring that buyers are not locked into single-source dependencies for critical materials. The robustness of the chemistry means that production schedules are less likely to be disrupted by batch failures or yield fluctuations. Such stability is paramount for maintaining continuous supply lines for downstream API production and meeting patient demand without interruption. Ultimately, the process design prioritizes operational efficiency and risk mitigation over theoretical elegance.
- Cost Reduction in Manufacturing: The streamlined nature of the synthetic route eliminates several unit operations that traditionally drive up processing costs. By avoiding the need for expensive transition metal catalysts and the associated scavenging steps, the overall material cost is significantly reduced. The high atom economy of the coupling reaction ensures that raw materials are converted into product rather than waste, further enhancing economic efficiency. Additionally, the ability to use standard glass-lined or stainless steel reactors without special coatings reduces capital expenditure requirements for manufacturers. These cumulative savings allow for more competitive pricing models without sacrificing margin or quality standards.
- Enhanced Supply Chain Reliability: The reagents and solvents specified in this protocol are globally available commodities with mature supply chains. This ubiquity means that procurement teams can source materials from multiple vendors, mitigating the risk of shortages or price spikes. The simplicity of the process also reduces the lead time for high-purity pharmaceutical intermediates by shortening the total cycle time from raw material to finished good. Manufacturers can respond more agilely to changes in demand forecasts, ensuring that inventory levels are optimized throughout the value chain. This resilience is a critical factor for long-term supply agreements and strategic partnerships in the pharmaceutical sector.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, utilizing reaction conditions that are easily manageable in large-scale vessels. The absence of hazardous reagents and the use of recyclable solvents align with modern green chemistry principles and environmental regulations. Waste streams are simpler to treat, reducing the burden on environmental health and safety departments and lowering disposal costs. The high yields achieved at each step minimize the volume of waste generated per kilogram of product, contributing to a lower carbon footprint. This alignment with sustainability goals is increasingly important for corporate social responsibility initiatives and regulatory compliance in global markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific embodiments and examples provided within the patent documentation to clarify practical application details. Understanding these nuances helps stakeholders make informed decisions about adopting this methodology for their specific projects. The answers reflect the current state of the art as described in the intellectual property disclosure.
Q: What are the key advantages of this new synthesis process?
A: The process described in patent CN114269338A offers a more economical and simple method for preparing diazaspiro lactam compounds compared to conventional methods, utilizing efficient coupling agents to streamline the cyclization step.
Q: Which coupling agents are compatible with this reaction?
A: The methodology supports a wide range of art-recognized coupling agents including carbodiimides like DCC and DIC, as well as phosphonium and uronium salts such as HATU and PyBOP, providing flexibility for scale-up.
Q: Is this process suitable for large-scale pharmaceutical production?
A: Yes, the use of common organic solvents like THF and DCM, along with robust reagents, indicates strong potential for commercial scale-up and reliable supply chain integration for API intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diazaspiro Lactam Supplier
NINGBO INNO PHARMCHEM stands ready to leverage this advanced chemistry to support your development and commercialization goals with unmatched expertise. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining rigorous quality standards. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications for complex intermediates like diazaspiro lactams. We understand the critical nature of NMDA modulator projects and are committed to delivering materials that accelerate your timelines. Our team of experts is prepared to navigate the complexities of process optimization to ensure your supply chain remains robust and efficient.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific needs. Request a Customized Cost-Saving Analysis to understand the economic impact of switching to this more efficient manufacturing process. We are eager to provide specific COA data and route feasibility assessments that demonstrate our capability to be your trusted partner. Contact us today to initiate a conversation about securing a stable and cost-effective supply of these vital pharmaceutical intermediates for your pipeline.
