Advanced Metal-Free Synthesis of 1,3-Dihydroisobenzofuran Derivatives for Commercial Scale
Advanced Metal-Free Synthesis of 1,3-Dihydroisobenzofuran Derivatives for Commercial Scale
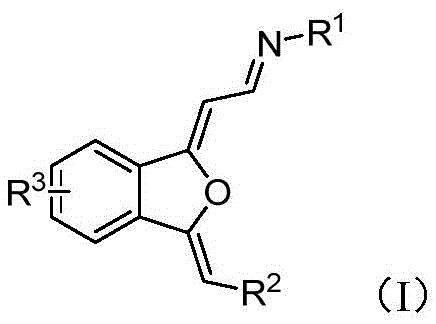
The landscape of organic synthesis for heterocyclic compounds is undergoing a significant transformation, driven by the urgent need for greener, more cost-effective, and scalable manufacturing processes. A pivotal development in this field is detailed in patent CN108947945B, which discloses a novel preparation method for 1,3-dihydroisobenzofuran derivatives. These compounds are not merely academic curiosities; they serve as critical structural units in a wide array of natural products, functional molecules, and increasingly, as key frameworks for molecular fluorescent probes used in detecting divalent copper and cyanide ions. The traditional reliance on multi-step syntheses involving precious metals has long been a bottleneck for industrial adoption. However, this new methodology leverages an ortho-alkynyl substituted amine ketene compound as a raw material, facilitating a direct, one-step catalytic cyclization. By shifting away from complex transition metal catalysis to a fluoride-mediated system, this technology offers a robust pathway for producing high-purity pharmaceutical intermediates and electronic chemical precursors with exceptional efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,3-dihydroisobenzofuran skeleton has been fraught with synthetic challenges that hinder large-scale commercialization. Prior art, including studies referenced in the background of the patent such as those by Shang et al. and Sekine et al., predominantly relies on the use of noble metal catalysts like gold or palladium salts. These conventional routes often necessitate rigorous exclusion of moisture and oxygen, requiring expensive inert gas setups that complicate reactor design and increase operational expenditures. Furthermore, the removal of trace heavy metal residues from the final active pharmaceutical ingredient (API) or intermediate is a stringent regulatory requirement, often demanding additional purification steps like scavenging or recrystallization, which inevitably erode overall yield. The substrate scope in these older methods is frequently limited, failing to accommodate diverse electronic environments without significant optimization, thereby restricting their utility in the rapid development of new drug candidates or functional materials.
The Novel Approach
In stark contrast, the innovative process described in CN108947945B introduces a paradigm shift by utilizing a simple, non-metallic catalytic system. The core of this advancement lies in the use of tetrabutylammonium fluoride (TBAF) to promote the cyclization of ortho-alkynyl substituted aminoenones. This approach operates under remarkably mild conditions, typically at temperatures around 35°C, and crucially, it is insensitive to air. This air tolerance is a game-changer for process chemistry, as it removes the absolute necessity for gloveboxes or Schlenk lines, drastically simplifying the engineering controls required for production. The reaction exhibits broad substrate universality, successfully tolerating various substituents including electron-withdrawing and electron-donating groups on the aryl rings, as well as silicon-based groups. This flexibility allows medicinal chemists and process engineers to access a diverse library of derivatives without redesigning the entire synthetic route for each new analog.
Mechanistic Insights into Fluoride-Catalyzed Cyclization
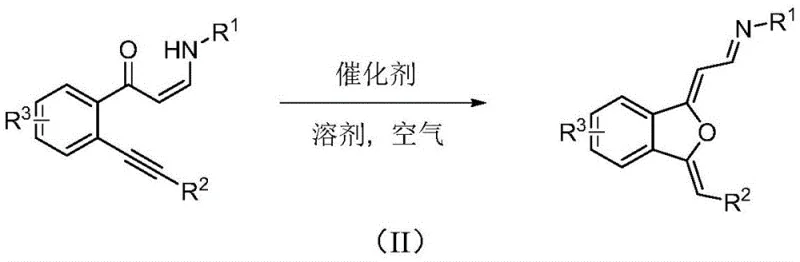
The mechanistic elegance of this transformation centers on the unique reactivity of the fluoride ion in organic media. Unlike traditional Lewis acid catalysis which activates electrophiles, the fluoride ion in this system likely acts as a base or a nucleophilic initiator to trigger the intramolecular attack of the oxygen or nitrogen functionality onto the activated alkyne moiety. The ortho-alkynyl substituted aminoenone substrate possesses a specific electronic arrangement where the alkyne is activated towards nucleophilic addition. Upon introduction of the TBAF catalyst in a solvent like Tetrahydrofuran (THF), the fluoride species facilitates the formation of a vinyl anion or a similar reactive intermediate, which subsequently undergoes cyclization to form the furan ring. This pathway avoids the high-energy barriers associated with oxidative addition and reductive elimination steps common in palladium cycles. Consequently, the energy input required is minimal, aligning perfectly with green chemistry principles by reducing the carbon footprint of the manufacturing process.
From an impurity control perspective, the mildness of the fluoride-catalyzed conditions is paramount. High-temperature reactions or those involving strong oxidants often lead to polymerization of the alkyne starting material or decomposition of the sensitive imine functionalities. By maintaining the reaction temperature at a moderate 35°C and avoiding harsh oxidants, the formation of side products is significantly suppressed. The patent data highlights isolated yields exceeding 95% for many substrates, indicating a clean reaction profile. For instance, the synthesis of specific derivatives demonstrates that even with steric hindrance or varying electronic properties, the catalyst maintains high turnover efficiency. This purity profile is critical for downstream applications, particularly in the synthesis of fluorescent probes where trace impurities can quench fluorescence or lead to false positives in sensing applications.
How to Synthesize 1,3-Dihydroisobenzofuran Derivatives Efficiently
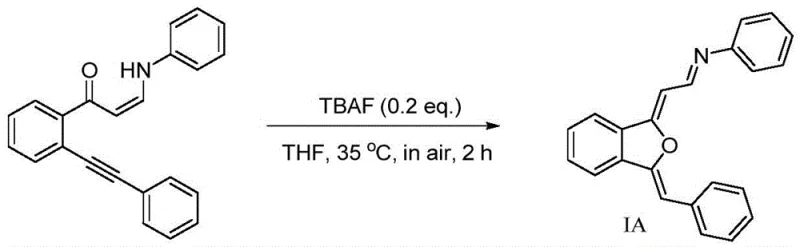
Implementing this synthesis in a laboratory or pilot plant setting is straightforward due to the robustness of the reaction conditions. The protocol eliminates the need for specialized air-free techniques, making it accessible to standard organic synthesis facilities. The process begins with the dissolution of the ortho-alkynyl substituted aminoenone precursor in a suitable organic solvent, with THF being the preferred medium due to its ability to solubilize both the organic substrate and the ionic catalyst. Following dissolution, the catalyst is introduced, and the mixture is heated gently. The simplicity of the work-up procedure further enhances its appeal; since no heavy metals are present, the product can often be isolated via simple filtration or extraction without the need for complex chromatographic purification or metal scavenging resins. For a detailed breakdown of the standardized operating procedures and safety parameters, please refer to the technical guide below.
- Dissolve the ortho-alkynyl substituted aminoenone raw material in an organic solvent such as Tetrahydrofuran (THF) or ethyl acetate within a reaction vessel open to air.
- Add the catalyst, preferably tetrabutylammonium fluoride (TBAF), maintaining a molar ratio between the substrate and catalyst of approximately 10: 2 to 5:1.
- Heat the reaction mixture to a mild temperature of 35°C and stir for 15 minutes to 3 hours, followed by standard separation and purification to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this metal-free methodology represents a substantial opportunity for cost optimization and risk mitigation. The elimination of noble metal catalysts directly impacts the bill of materials, removing one of the most volatile and expensive cost drivers in fine chemical manufacturing. Moreover, the supply chain for TBAF is stable and mature, unlike the geopolitical complexities often surrounding the sourcing of platinum group metals. The operational simplicity also translates to faster batch turnover times; without the need for extensive degassing or inert atmosphere maintenance, reactors can be charged and discharged more rapidly, increasing overall facility throughput. This efficiency gain is compounded by the high yields reported, which minimize raw material waste and maximize the output per batch, ensuring a more reliable supply of critical intermediates for downstream drug substance manufacturing.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the substitution of expensive noble metal catalysts with inexpensive fluoride salts. In traditional routes, the cost of the catalyst itself, combined with the downstream costs of metal removal and waste disposal, can be prohibitive. By utilizing TBAF, manufacturers can achieve drastic cost savings while simultaneously simplifying the purification train. The high atom economy and excellent yields mean that less raw material is required to produce the same amount of product, further driving down the cost per kilogram. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to lower utility costs over the lifecycle of the product.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the air-insensitive nature of this reaction. Conventional methods requiring strict inert gas protection are vulnerable to disruptions in nitrogen or argon supply and equipment failures in gas delivery systems. This new method can be run in standard vessels open to the atmosphere, reducing dependency on specialized infrastructure. Furthermore, the raw materials, ortho-alkynyl substituted aminoenones, are synthetically accessible from common building blocks, ensuring a steady flow of inputs. This reliability is crucial for maintaining continuous production schedules and meeting the tight delivery windows demanded by global pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden hazards, but the benign nature of this fluoride-catalyzed cyclization makes it highly amenable to scale-up. The absence of pyrophoric reagents or high-pressure hydrogenation steps reduces the safety risks associated with large-scale production. From an environmental standpoint, the process aligns with increasingly stringent regulations regarding heavy metal discharge. By generating waste streams free of toxic transition metals, the burden on wastewater treatment facilities is lightened, and the environmental compliance costs are minimized. This 'green' credential is increasingly becoming a prerequisite for suppliers aiming to partner with top-tier multinational corporations committed to sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these nuances is essential for R&D teams planning pilot runs and for procurement teams assessing vendor capabilities.
Q: Does this synthesis method require inert gas protection?
A: No, one of the distinct advantages of this patented process (CN108947945B) is its insensitivity to air. The reaction proceeds efficiently under ambient air conditions, eliminating the need for costly nitrogen or argon blanketing systems typically required for sensitive organometallic chemistry.
Q: What catalysts are used in this novel pathway?
A: The process utilizes non-metal catalysts, specifically tetrabutylammonium fluoride (TBAF) or a combination of tetrabutylammonium bromide and cesium fluoride. This avoids the use of expensive and toxic noble metal catalysts like palladium or gold found in conventional methods.
Q: What are the typical yields for this cyclization reaction?
A: The patent data indicates excellent yields, often exceeding 95% when TBAF is used as the catalyst. Specific examples in the documentation demonstrate isolated yields of up to 99% for various substituted derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Dihydroisobenzofuran Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN108947945B for the production of high-value heterocyclic intermediates. As a leading CDMO partner, we possess the technical expertise to translate this laboratory-scale innovation into robust, commercial-scale manufacturing processes. Our facilities are equipped to handle diverse synthetic pathways, and we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that consistency is key in the pharmaceutical supply chain; therefore, our operations are governed by stringent purity specifications and supported by rigorous QC labs that ensure every batch meets the highest standards of quality and safety required for API intermediates and functional materials.
We invite you to collaborate with us to leverage this advanced technology for your next project. Whether you require custom synthesis of specific 1,3-dihydroisobenzofuran derivatives or process optimization for existing routes, our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to contact our technical procurement team to request specific COA data for similar compounds and to discuss route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality supply chain for your critical chemical building blocks.
