Advanced Pd-Catalyzed C-H Activation for Scalable Polysubstituted Benzoic Acid Manufacturing
Advanced Pd-Catalyzed C-H Activation for Scalable Polysubstituted Benzoic Acid Manufacturing
The landscape of organic synthesis for complex aromatic scaffolds is undergoing a paradigm shift, driven by the urgent need for more sustainable and efficient manufacturing processes in the pharmaceutical and fine chemical sectors. Patent CN109867613B introduces a groundbreaking methodology for the synthesis of polysubstituted benzoic acids, a critical class of building blocks widely utilized in the construction of bioactive molecules and advanced material precursors. This technology leverages a sophisticated palladium-catalyzed C-H activation strategy that fundamentally alters the retrosynthetic logic traditionally applied to these structures. By transforming simple, commodity-grade benzoic acid derivatives and readily accessible aziridines into high-value polysubstituted products in a single operational step, this invention addresses long-standing bottlenecks related to step economy and atom efficiency. For R&D directors and process chemists, this represents a significant opportunity to streamline synthetic routes, while supply chain managers will recognize the inherent value in utilizing robust, air-stable reaction conditions that facilitate reliable commercial scale-up of complex pharmaceutical intermediates.
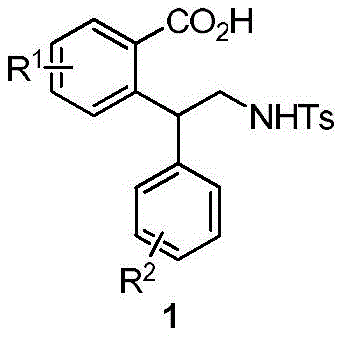
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted benzoic acid frameworks has relied heavily on classical electrophilic aromatic substitution reactions, such as the Friedel-Crafts alkylation, or transition-metal catalyzed cross-coupling reactions involving pre-functionalized aryl halides. These conventional pathways suffer from severe intrinsic limitations that hinder their efficiency in modern process chemistry. A primary drawback is the necessity for protecting group chemistry; the carboxylic acid moiety, being highly reactive and capable of coordinating with Lewis acids or metal catalysts, often requires tedious protection and deprotection sequences to prevent side reactions or catalyst poisoning. Furthermore, traditional methods frequently demand harsh reaction conditions, including strong acids, elevated temperatures, or cryogenic environments for organometallic reagents like Grignard or organolithium species. These aggressive conditions not only limit the tolerance of sensitive functional groups but also generate substantial quantities of hazardous waste, complicating downstream purification and increasing the environmental footprint of the manufacturing process.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in patent CN109867613B employs a direct C-H functionalization strategy that elegantly bypasses the need for pre-halogenation or carboxyl protection. By utilizing the carboxylic acid group itself as an intrinsic directing group, the process achieves high regioselectivity for ortho-substitution without additional synthetic manipulations. This innovative approach couples substituted benzoic acids with N-tosyl aziridines under mild thermal conditions, typically around 80°C, using a palladium catalyst system. The reaction proceeds with remarkable efficiency, delivering target products in yields ranging from 50% to 90% across a diverse array of substrates. This simplification of the synthetic sequence not only reduces the total number of unit operations but also significantly lowers the consumption of raw materials and solvents, thereby offering a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Pd-Catalyzed Ortho-C-H Activation
The core of this technological advancement lies in the intricate interplay between the palladium catalyst, the carboxyl directing group, and the specialized reaction medium. The mechanism initiates with the coordination of the palladium(II) species to the oxygen atoms of the carboxylic acid group on the benzoic acid substrate. This coordination event positions the metal center in close proximity to the ortho-C-H bond, facilitating a concerted metalation-deprotonation (CMD) process. This step is critical as it forms a stable five-membered palladacycle intermediate, which serves as the key organometallic species for the subsequent transformation. The presence of additives such as N-acetyl glycine plays a pivotal role in accelerating this C-H cleavage step, likely by acting as a bidentate ligand that stabilizes the active catalytic species and lowers the activation energy barrier for the metalation event.
Following the formation of the palladacycle, the reaction proceeds through the insertion of the aziridine substrate. The strained three-membered ring of the aziridine is highly susceptible to nucleophilic attack or oxidative addition, leading to ring opening and the formation of a new carbon-carbon bond at the ortho position of the benzene ring. The use of hexafluoroisopropanol (HFIP) as the solvent is particularly noteworthy; this polar protic solvent is known to stabilize cationic intermediates and enhance hydrogen bonding interactions, which can significantly improve the reaction rate and selectivity. The final step involves protonolysis or reductive elimination to release the polysubstituted benzoic acid product and regenerate the active palladium catalyst, completing the catalytic cycle. This mechanistic understanding underscores the robustness of the process, as it tolerates a wide range of electronic effects from substituents like halogens, nitro groups, and alkyl chains, ensuring consistent performance across different substrate classes.
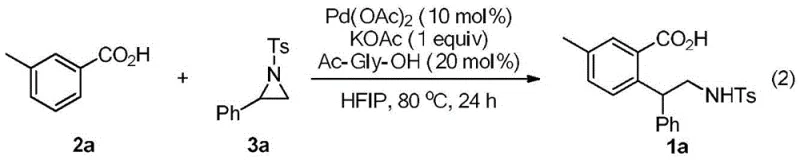
How to Synthesize Polysubstituted Benzoic Acid Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and purity. The protocol is designed to be user-friendly, operating under ambient air pressure rather than requiring stringent inert atmosphere techniques, which greatly simplifies the engineering controls needed for production. The standard procedure involves combining the benzoic acid derivative, the aziridine coupling partner, palladium acetate, potassium acetate, and N-acetyl glycine in hexafluoroisopropanol. The mixture is then heated to 80°C for a duration of approximately 24 hours. Following the reaction, standard workup procedures involving solvent removal and silica gel chromatography are sufficient to isolate the high-purity product. For detailed standardized operating procedures and safety guidelines, please refer to the technical documentation below.
- Charge a reaction vessel with substituted benzoic acid, palladium catalyst (e.g., Pd(OAc)2), aziridine derivative, base (e.g., KOAc), and amino acid additive (e.g., Ac-Gly-OH) in HFIP solvent.
- Heat the reaction mixture to 80°C under air atmosphere and stir for 24 hours to facilitate the C-H activation and ring-opening coupling.
- Upon completion, remove volatiles under reduced pressure and purify the crude residue via silica gel column chromatography to isolate the target polysubstituted benzoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers transformative benefits for procurement strategies and supply chain resilience. The primary advantage stems from the utilization of commodity chemicals as starting materials. Substituted benzoic acids and aziridines are widely available from global chemical suppliers at competitive price points, eliminating the dependency on exotic or custom-synthesized precursors that often plague complex API supply chains. This accessibility ensures a stable supply base and mitigates the risk of raw material shortages that can disrupt production schedules. Furthermore, the elimination of protection and deprotection steps translates directly into reduced consumption of auxiliary reagents and solvents, driving down the overall variable cost of goods sold (COGS) without compromising on quality.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by its exceptional step economy. By condensing what would traditionally be a multi-step sequence into a single catalytic transformation, manufacturers can realize substantial savings in labor, energy, and equipment usage. The avoidance of expensive protecting group reagents and the reduction in purification stages significantly lower the processing costs. Additionally, the high yields reported (50-90%) minimize material loss, ensuring that a greater proportion of the input mass is converted into valuable saleable product, thereby optimizing the overall material balance and enhancing profit margins.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply chain stability. Operating under air atmosphere at moderate temperatures (80°C) removes the need for specialized cryogenic cooling or high-pressure hydrogenation equipment, making the process adaptable to a wider range of manufacturing facilities. This flexibility allows for easier technology transfer between sites and reduces the lead time for establishing new production lines. The tolerance for diverse functional groups also means that supply chain disruptions for specific specialized substrates can be mitigated by switching to alternative analogues without redesigning the entire synthetic route.
- Scalability and Environmental Compliance: As regulatory pressures regarding waste disposal and environmental impact intensify, this green chemistry approach offers a distinct competitive advantage. The reduction in synthetic steps inherently reduces the E-factor (mass of waste per mass of product), aligning with sustainability goals. The use of recyclable solvents and the minimization of heavy metal waste through efficient catalysis simplify wastewater treatment protocols. This environmental compatibility facilitates smoother regulatory approvals and reduces the long-term liability associated with hazardous waste management, making it an ideal candidate for sustainable commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: What are the key advantages of this Pd-catalyzed method over traditional Friedel-Crafts alkylation?
A: Unlike traditional methods that require harsh conditions and pre-protection of the carboxyl group, this novel approach utilizes the carboxyl group directly as a directing group for ortho-C-H activation. This eliminates multiple synthetic steps, reduces waste generation, and allows for milder reaction conditions (80°C) with high functional group tolerance.
Q: Can this synthesis method be scaled for industrial production of API intermediates?
A: Yes, the process is highly amenable to scale-up. It utilizes commercially available and inexpensive starting materials such as substituted benzoic acids and aziridines. The reaction operates under air atmosphere without the need for inert gas protection, significantly simplifying the engineering requirements for large-scale reactors.
Q: What is the scope of substituents compatible with this synthetic route?
A: The method demonstrates broad substrate scope, tolerating a wide variety of electronic and steric environments. Substituents including methyl, methoxy, halogens (fluoro, chloro, bromo, iodo), nitro, cyano, and trifluoromethyl groups at ortho, meta, and para positions are well-tolerated, yielding products with 50-90% efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Benzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic methodologies in maintaining a competitive edge in the global pharmaceutical market. Our team of expert process chemists has thoroughly evaluated the technology described in patent CN109867613B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and risk-mitigated. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of polysubstituted benzoic acid derivatives we produce, meeting the exacting standards required by top-tier pharmaceutical companies.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains using this advanced technology. By leveraging our expertise in C-H activation chemistry, we can help you achieve significant operational efficiencies and cost savings. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term growth objectives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
