Revolutionizing Lamivudine Intermediate Synthesis via Continuous Flow Technology
Revolutionizing Lamivudine Intermediate Synthesis via Continuous Flow Technology
The pharmaceutical industry is constantly seeking more efficient, scalable, and cost-effective methods for producing critical antiviral intermediates. Patent CN107382725B introduces a groundbreaking continuous production method for L-menthyl glycolate (MGH), a pivotal precursor in the synthesis of Lamivudine, a potent nucleoside antiviral drug used globally for treating Hepatitis B Virus (HBV). This technology represents a significant leap forward from traditional batch processing, addressing long-standing challenges regarding yield, purity, and environmental impact. By transitioning to a steady-state continuous flow system, manufacturers can achieve superior control over reaction parameters, minimizing the formation of detrimental byproducts that have historically plagued the synthesis of this complex ester. For R&D directors and procurement specialists alike, understanding this shift is crucial for optimizing supply chains and ensuring the consistent availability of high-quality active pharmaceutical ingredients.
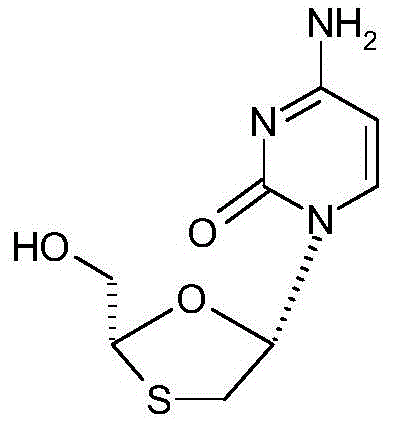
Lamivudine, chemically known as (2R-cis)-4-amino-1-(2-hydroxymethyl-1,3-oxathiolan-5-yl)-1H-pyrimidine-2-ketone, relies heavily on the quality of its starting materials. The structural integrity of the final drug depends on the stereochemical purity of the L-menthyl group introduced early in the synthesis. The patent highlights that MGH serves as the essential starting material for constructing the oxathiolane ring system found in Lamivudine. Consequently, any inefficiency in producing MGH directly cascades into increased costs and potential supply bottlenecks for the final API. The innovation detailed in this patent not only streamlines the production of MGH but also aligns with modern green chemistry principles by reducing wastewater treatment loads and energy consumption, making it an attractive proposition for environmentally conscious manufacturing facilities aiming to reduce their carbon footprint while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
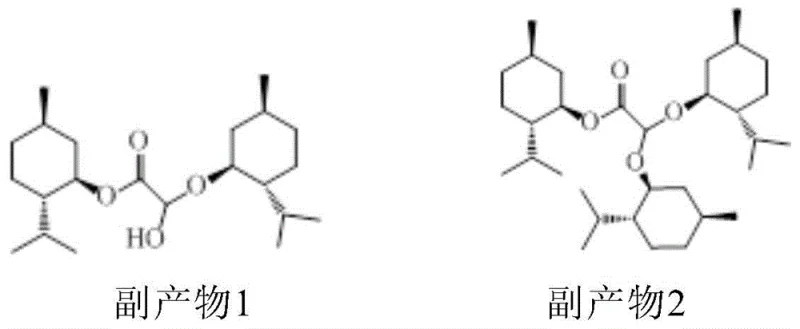
Historically, the industrial production of L-menthyl dihydroxyacetate has relied heavily on batch processing methods, which suffer from inherent inefficiencies and reproducibility issues. In the traditional esterification stage, achieving high conversion of glyoxylic acid often necessitates harsh conditions that promote the formation of significant amounts of byproducts. Specifically, the reaction tends to generate hemiacetal byproducts and acetal byproducts, with the latter potentially reaching concentrations as high as 26% in the reaction mixture. These impurities not only lower the overall yield of the desired MGH but also complicate downstream purification processes, requiring extensive washing and recrystallization steps that increase solvent usage and waste generation. Furthermore, the subsequent addition reaction with sodium bisulfite in batch reactors typically requires prolonged stirring times, often exceeding 24 hours, leading to excessive energy consumption and low equipment turnover rates. This sluggish kinetics creates a bottleneck in the production line, limiting the total output capacity and driving up the cost per kilogram of the intermediate.
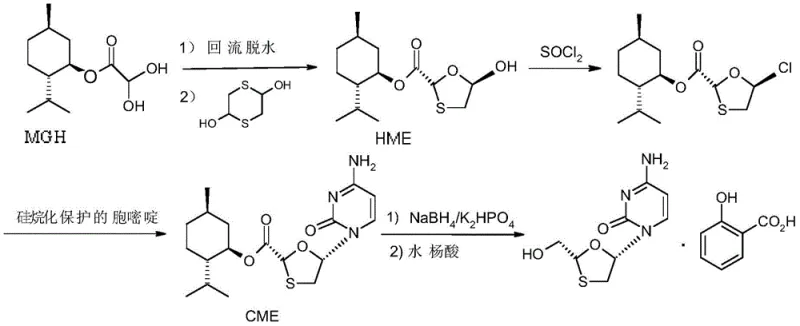
The accumulation of these inefficiencies makes batch processing economically unsustainable for large-scale operations aiming to meet the growing global demand for antiviral medications. The variability inherent in batch-to-batch processing also poses risks to quality control, as slight deviations in temperature or mixing efficiency can lead to fluctuating impurity profiles. Additionally, the handling of large volumes of reactive mixtures in single vessels increases safety risks associated with exothermic events. The formation of byproducts like those shown in the patent documentation underscores the lack of selectivity in conventional methods, where the equilibrium favors side reactions over the desired esterification. This necessitates a fundamental rethinking of the process architecture to move away from static batch vessels towards dynamic flow systems that can better manage reaction thermodynamics and kinetics.
The Novel Approach
The patented continuous production method offers a robust solution to these challenges by implementing a multi-stage flow reactor system that maintains precise control over reaction conditions. Instead of a single large vessel, the process utilizes a series of connected reactors for both the addition and reduction steps, allowing for a steady-state operation where reactants are continuously fed and products are continuously removed. This approach drastically reduces the residence time required for the bisulfite addition step, eliminating the need for the 24-hour stirring periods characteristic of batch methods. By controlling the flow rates and temperature gradients across the reactor series, the system effectively suppresses the formation of hemiacetal and acetal byproducts, thereby enhancing the selectivity for the target MGH compound. The result is a process that not only achieves higher yields but also produces a cleaner crude product, simplifying the subsequent purification workload and reducing the overall environmental burden of the manufacturing process.
Furthermore, the continuous process enables better management of the exothermic nature of the reactions involved. In a flow system, the surface-area-to-volume ratio is significantly higher than in a batch tank, facilitating more efficient heat dissipation and preventing hot spots that could degrade the product or trigger runaway reactions. The patent describes a specific configuration where the esterification is performed in a single kettle with continuous overflow, while the addition and reduction steps utilize three-kettle series systems. This modular design allows for independent optimization of each stage; for instance, the pH can be precisely adjusted between stages using automated dosing of regulators like sodium carbonate. Such granular control ensures that the reaction environment remains optimal throughout the entire synthesis, maximizing raw material utilization and minimizing the generation of waste streams that require costly treatment. This level of process intensification is key to achieving the economic and operational advantages required for modern pharmaceutical manufacturing.
Mechanistic Insights into Continuous Flow Esterification and Reduction
The core of this innovative process lies in the intricate interplay between esterification kinetics and the subsequent nucleophilic addition and reduction steps. In the initial stage, L-menthol reacts with glyoxylic acid in an inert solvent such as cyclohexane, catalyzed by sulfuric acid. The continuous removal of water via azeotropic distillation in the flow setup drives the equilibrium towards the ester product, preventing the reverse hydrolysis reaction. Crucially, the continuous flow prevents the local accumulation of glyoxylic acid, which is a primary driver for the formation of the troublesome acetal byproducts seen in batch processes. By maintaining a low concentration of free aldehyde through immediate consumption or controlled feeding, the system favors the formation of the mono-ester MGH over the di-ester or acetal derivatives. This kinetic control is further enhanced by the precise temperature regulation, typically maintained between 70°C and 100°C, which provides sufficient activation energy for esterification without promoting thermal degradation or side reactions.

Following esterification, the process moves to the formation of the alpha-hydroxysulfonic acid sodium salt via reaction with sodium bisulfite. In this step, the continuous flow ensures rapid mixing of the organic ester phase with the aqueous bisulfite phase, creating a large interfacial area for the reaction to occur. The patent emphasizes the importance of pH control in this stage, maintaining the system between 4.0 and 6.0 to optimize the nucleophilicity of the bisulfite ion while preventing the decomposition of the sensitive intermediate. The subsequent reduction with formaldehyde is equally critical, where the pH is adjusted to a slightly neutral range of 6.0 to 8.0 to facilitate the hydride transfer or equivalent reduction mechanism that converts the sulfonate intermediate into the final hydroxymethyl group. The use of a three-kettle series for this reduction allows for a staged addition of formaldehyde and base, ensuring complete conversion while minimizing the risk of over-reaction or polymerization of the formaldehyde. This mechanistic precision is what allows the process to achieve GC purities exceeding 99%, a benchmark that is difficult to reach with conventional batch techniques.
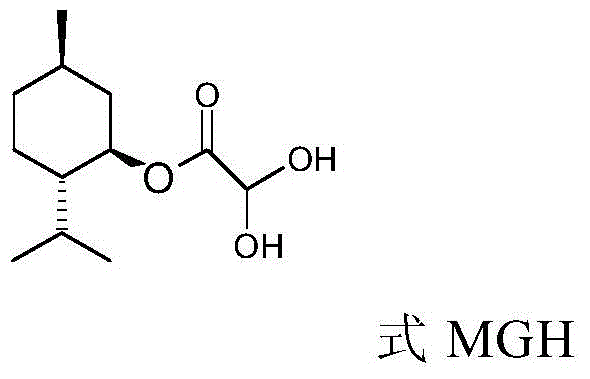
How to Synthesize L-Menthyl Glycolate Efficiently
The synthesis of L-menthyl glycolate via this continuous method involves a carefully orchestrated sequence of unit operations designed to maximize throughput and purity. The process begins with the preparation of a menthol solution in cyclohexane, which is then fed into the esterification reactor alongside a mixture of glyoxylic acid and sulfuric acid. The reaction mixture is heated to reflux, and the water generated is continuously separated, driving the reaction to completion. The resulting esterification liquid is then washed and separated before entering the addition reactor series, where it meets the sodium bisulfite solution under controlled pH conditions. Finally, the addition product undergoes reduction with formaldehyde in a third reactor series, followed by crystallization and drying to yield the final white solid product. For a detailed breakdown of the specific flow rates, temperatures, and residence times required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Perform continuous esterification of menthol and glyoxylic acid in an inert solvent like cyclohexane using sulfuric acid catalysis at controlled reflux temperatures.
- Mix the esterification solution with sodium bisulfite in a series of reactors to form the alpha-hydroxysulfonic acid sodium salt intermediate under strict pH control.
- React the addition solution with formaldehyde in a multi-stage reduction process, followed by crystallization and purification to isolate the final MGH product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this continuous flow technology translates into tangible strategic advantages that go beyond simple yield improvements. The shift from batch to continuous processing fundamentally alters the cost structure of MGH manufacturing by significantly enhancing equipment utilization rates. In a batch plant, reactors sit idle during charging, heating, cooling, and discharging phases; in contrast, a continuous plant operates at steady state, meaning the same footprint of equipment can produce a vastly larger volume of product over time. This intensification reduces the capital expenditure required for new capacity expansions and lowers the fixed cost allocation per kilogram of product. Moreover, the reduction in reaction time from days to hours means that working capital is tied up for shorter periods, improving cash flow dynamics for the manufacturer and, by extension, offering more competitive pricing structures to downstream buyers.
- Cost Reduction in Manufacturing: The elimination of long stirring times and the reduction in byproduct formation lead to substantial savings in energy and raw material costs. By avoiding the formation of up to 26% acetal byproducts, the process ensures that nearly every mole of expensive chiral menthol is converted into valuable product rather than waste. Additionally, the simplified purification train reduces the consumption of solvents and the associated costs of solvent recovery and disposal. The use of common, inexpensive catalysts like sulfuric acid instead of precious metals further keeps the variable costs low, making the process economically resilient against fluctuations in raw material prices.
- Enhanced Supply Chain Reliability: Continuous manufacturing inherently provides a more consistent and predictable output profile compared to batch processing. The steady-state nature of the reaction minimizes batch-to-batch variability, ensuring that every shipment of MGH meets the same stringent quality specifications. This reliability is critical for pharmaceutical supply chains, where deviations in intermediate quality can halt API production lines. The ability to run the plant for extended periods without shutdowns for cleaning and turnaround means that suppliers can maintain higher inventory levels and respond more quickly to sudden spikes in demand for Lamivudine, thereby reducing the risk of stockouts for global health programs.
- Scalability and Environmental Compliance: The modular nature of the flow reactor system allows for straightforward scale-up by simply increasing the run time or numbering up the reactor units, avoiding the engineering challenges associated with scaling large batch tanks. From an environmental perspective, the process generates significantly less wastewater due to higher conversion rates and reduced washing requirements. The lower energy intensity of the process also contributes to a smaller carbon footprint, helping pharmaceutical companies meet their sustainability goals. The reduced volume of hazardous waste simplifies compliance with increasingly strict environmental regulations, mitigating the risk of regulatory fines or production stoppages.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the continuous production of L-menthyl glycolate. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing supply chains or for R&D groups looking to optimize their own synthesis routes. The clarity provided here aims to bridge the gap between academic patent claims and practical industrial application.
Q: How does the continuous process improve yield compared to batch methods?
A: The continuous process effectively controls side reactions such as hemiacetal and acetal formation by maintaining steady-state reaction conditions, resulting in significantly higher selectivity and raw material utilization compared to traditional batch stirring which often yields up to 26% acetal byproducts.
Q: What solvents are compatible with this synthesis route?
A: The patent specifies the use of inert solvents such as cyclohexane, hexane, or toluene, with cyclohexane being particularly preferred for the recrystallization and extraction steps due to its efficiency in phase separation and product recovery.
Q: Is this method suitable for large-scale industrial manufacturing?
A: Yes, the method utilizes a multi-kettle series continuous process which greatly improves equipment unit volume productivity, reduces the number of required reactors, and allows for automatic production, making it highly suitable for industrial mass production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Menthyl Glycolate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against viral diseases. Our team of expert process chemists has thoroughly analyzed the continuous flow technology described in Patent CN107382725B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering L-menthyl glycolate that meets the most stringent purity specifications, utilizing our rigorous QC labs to ensure every batch is free from the hemiacetal and acetal impurities that compromise downstream synthesis. Our facility is equipped with state-of-the-art continuous flow reactors that allow us to harness the full efficiency of this patented method, guaranteeing a stable supply of this vital building block for the pharmaceutical industry.
We invite procurement directors and supply chain managers to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized continuous process, we can offer significant value propositions that traditional batch manufacturers cannot match. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. Let us partner with you to secure a resilient, cost-effective, and high-quality supply of L-menthyl glycolate for your Lamivudine production needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
