Advanced Copper-Catalyzed Synthesis of 3-Amino-2-indenecarbonitrile for Commercial Scale-up
Advanced Copper-Catalyzed Synthesis of 3-Amino-2-indenecarbonitrile for Commercial Scale-up
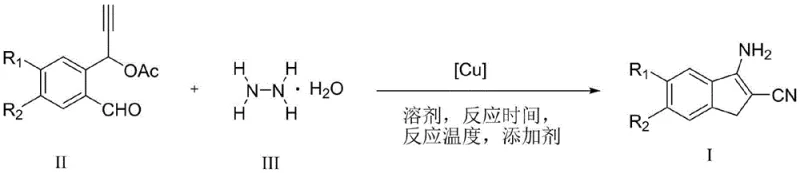
The pharmaceutical and material science industries are constantly seeking efficient, scalable routes to complex heterocyclic scaffolds, and the recent disclosure in patent CN114436890B offers a transformative solution for the production of 3-amino-2-indenecarbonitrile derivatives. This groundbreaking technology leverages a novel copper-catalyzed system to facilitate the synthesis of these valuable intermediates through an innovative N,N-bond cleavage mechanism involving hydrazine hydrate and propargyl compounds. Unlike conventional pathways that often rely on scarce and expensive noble metals, this method utilizes abundant copper salts to drive the cyclization with exceptional efficiency, achieving yields as high as 90% under remarkably mild conditions. For R&D directors and procurement strategists, this represents a significant opportunity to optimize the manufacturing of key pharmaceutical intermediates and functional materials, ensuring both economic viability and supply chain resilience in an increasingly competitive global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indene core, particularly the 3-amino-2-indenecarbonitrile motif, has been a formidable challenge in organic synthesis, often necessitating the use of sophisticated and costly catalytic systems. Traditional literature reports, such as those describing the Thorpe-Ziegler reaction, typically depend on noble metal catalysts like Iridium to effect the necessary nitrile catalytic cross-coupling reactions. These legacy processes are fraught with limitations, including the prohibitive cost of the catalysts themselves, which can severely impact the overall cost of goods sold (COGS) for large-scale production. Furthermore, these older methods frequently require harsh reaction conditions, such as elevated temperatures or stringent anhydrous environments, which complicate process safety and increase energy consumption. The reliance on precious metals also introduces significant supply chain vulnerabilities, as the availability of metals like Iridium is geographically concentrated and subject to volatile market fluctuations, posing a risk to long-term production continuity for critical drug substances.
The Novel Approach
In stark contrast to these legacy challenges, the methodology outlined in patent CN114436890B introduces a paradigm shift by employing a readily available copper catalyst system to drive the transformation. This innovative route utilizes the cleavage of the N,N-bond in hydrazine hydrate to construct the target indene framework, a mechanistic pathway that is both atom-economical and operationally simple. The reaction proceeds efficiently at mild temperatures, typically around 40°C, in common organic solvents like dichloromethane, eliminating the need for energy-intensive heating or specialized high-pressure equipment. By replacing expensive noble metals with inexpensive copper salts, this approach drastically reduces the raw material burden while simultaneously simplifying the downstream purification process, as copper residues are generally easier to manage and remove than heavy noble metal contaminants. This robustness allows for the convenient synthesis of various functionalized derivatives, making it an ideal candidate for the rapid generation of diverse compound libraries in drug discovery programs.
Mechanistic Insights into Copper-Catalyzed N,N-Bond Cleavage Cyclization
The core of this technological advancement lies in the unique ability of the copper catalyst to activate the propargyl substrate and facilitate the subsequent nucleophilic attack by hydrazine hydrate. The mechanism likely involves the coordination of the copper center to the alkyne moiety of the propargyl compound, increasing its electrophilicity and rendering it susceptible to attack by the nitrogen nucleophile. Following the initial addition, the copper species promotes the critical N,N-bond cleavage of the hydrazine fragment, a step that is energetically demanding in the absence of a suitable catalyst. This cleavage event triggers a cascade of intramolecular cyclization steps, ultimately forging the five-membered ring of the indene system and establishing the crucial amino and cyano functionalities in a single operational sequence. The elegance of this mechanism is further highlighted by its tolerance to a wide array of substituents on the aromatic ring, including alkyl, alkoxy, halogen, and nitro groups, which suggests that the catalytic cycle is robust against steric and electronic variations in the substrate structure.
From an impurity control perspective, this copper-catalyzed pathway offers distinct advantages over radical-based or high-temperature thermal cyclizations. The mild reaction conditions minimize the formation of thermal degradation byproducts and polymerization side reactions that often plague indene syntheses. Moreover, the use of hydrazine hydrate as a nitrogen source is highly selective under these catalytic conditions, reducing the likelihood of over-alkylation or the formation of complex hydrazine-derived oligomers. The specific choice of additives, such as anhydrous sodium sulfate or molecular sieves, plays a pivotal role in sequestering water generated during the reaction or stabilizing the active copper species, thereby maintaining high catalytic turnover numbers. This level of control over the reaction environment ensures a clean crude profile, which translates directly to higher isolated yields and reduced solvent consumption during the final purification stages, a critical factor for green chemistry compliance in modern manufacturing.
How to Synthesize 3-Amino-2-indenecarbonitrile Efficiently
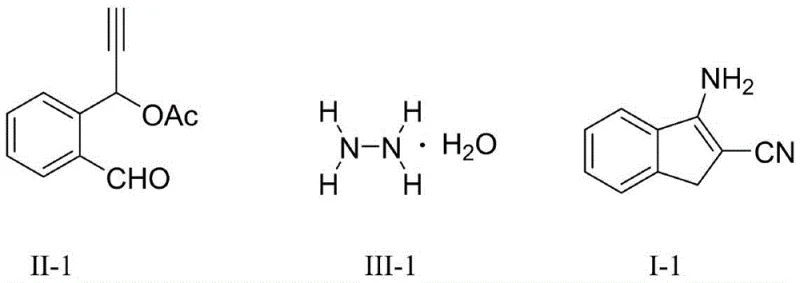
Implementing this synthesis in a laboratory or pilot plant setting is straightforward and aligns well with standard organic processing capabilities. The procedure begins with the preparation of a catalyst mixture, where a copper salt such as copper(II) acetate monohydrate is combined with a drying agent like anhydrous sodium sulfate in a suitable reaction vessel. The substrates, specifically the functionalized propargyl compound and hydrazine hydrate, are dissolved in a solvent like dichloromethane and introduced to the catalyst bed under stirring. The reaction is allowed to proceed at a moderate temperature, typically between 25°C and 60°C, for a duration of 1 to 24 hours depending on the specific substrate reactivity. Upon completion, the workup involves simple concentration under reduced pressure followed by standard silica gel chromatography, a process that is familiar to most synthetic chemistry teams and requires no specialized equipment beyond standard glassware and rotary evaporators.
- Prepare the reaction mixture by adding a copper catalyst (e.g., Cu(OAc)2·H2O) and an additive (e.g., anhydrous sodium sulfate) into a reaction container.
- Dissolve the propargyl compound and hydrazine hydrate in a reaction medium such as dichloromethane, then add this solution to the stirred catalyst mixture.
- Stir the reaction at 25-60°C for 1-24 hours, then concentrate under reduced pressure and purify via silica gel column chromatography to obtain the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this copper-catalyzed methodology presents a compelling value proposition centered on cost stability and operational flexibility. The transition from noble metal catalysts to base metal copper represents a fundamental shift in the cost structure of the synthesis, removing the exposure to the volatile pricing of rare earth and platinum group metals. This substitution not only lowers the direct material costs but also mitigates the regulatory and environmental burdens associated with the disposal and recycling of heavy metal waste streams. Furthermore, the mild reaction conditions imply lower energy requirements for heating and cooling, contributing to a reduced carbon footprint and lower utility costs per kilogram of product produced. These factors combine to create a more resilient supply chain that is less susceptible to external shocks in the commodities market.
- Cost Reduction in Manufacturing: The elimination of expensive Iridium catalysts in favor of ubiquitous copper salts results in a drastic reduction in catalyst expenditure, which is a major component of the variable cost in fine chemical synthesis. Additionally, the high yields reported (up to 90%) mean that less starting material is wasted, improving the overall mass balance and reducing the cost per unit of the active pharmaceutical ingredient (API) intermediate. The simplicity of the workup procedure further reduces labor and solvent costs, as extensive extraction or complex crystallization steps are often unnecessary when the crude purity is high.
- Enhanced Supply Chain Reliability: Sourcing copper salts and hydrazine hydrate is significantly more reliable than procuring specialized noble metal complexes, as these are commodity chemicals produced at massive scales globally. This abundance ensures that production schedules are not disrupted by catalyst shortages, a common issue with bespoke ligand-metal systems. The broad substrate scope also means that a single set of reaction conditions can be applied to a wide variety of analogues, allowing for flexible manufacturing campaigns where different intermediates can be produced on the same multipurpose equipment without extensive requalification.
- Scalability and Environmental Compliance: The reaction operates at near-ambient pressure and moderate temperatures, making it inherently safer and easier to scale from gram to ton quantities without the need for specialized high-pressure reactors. The use of common solvents like dichloromethane or ethanol facilitates solvent recovery and recycling, aligning with strict environmental regulations regarding volatile organic compound (VOC) emissions. Moreover, the avoidance of toxic heavy metals simplifies the wastewater treatment process, reducing the cost and complexity of environmental compliance and waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the experimental data and scope defined in the patent documentation. These insights are intended to clarify the operational parameters and potential applications for stakeholders evaluating this route for their own manufacturing pipelines. Understanding these nuances is essential for making informed decisions about process adoption and technology transfer.
Q: What are the optimal reaction conditions for this copper-catalyzed synthesis?
A: According to patent CN114436890B, the optimal conditions involve using Cu(OAc)2·H2O as the catalyst in dichloromethane at 40°C for 4 hours, yielding up to 90%.
Q: Can this method tolerate various functional groups on the substrate?
A: Yes, the method demonstrates wide substrate applicability, tolerating substituents such as alkyl, cycloalkyl, phenyl, benzyl, and heterocyclic groups on the aromatic ring.
Q: How does this method compare to traditional noble metal catalysis?
A: Unlike traditional methods requiring expensive Iridium catalysts for Thorpe-Ziegler reactions, this novel approach utilizes inexpensive copper salts, significantly reducing raw material costs while maintaining high yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Amino-2-indenecarbonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of robust and scalable synthetic routes like the one described in CN114436890B for the development of next-generation therapeutics and advanced materials. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-amino-2-indenecarbonitrile delivered meets the highest standards of quality required by global regulatory bodies. We are committed to leveraging our technical expertise to optimize this copper-catalyzed process further, ensuring maximum yield and minimal environmental impact for our partners.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be integrated into your supply chain to drive down costs and accelerate your time to market. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring that you have the reliable support necessary to bring your most challenging chemical projects to fruition.
