Scalable Synthesis of 1-(2-Halobiphenyl-4-yl)-Cyclopropanecarboxylic Acid Derivatives for Neurodegenerative Therapies
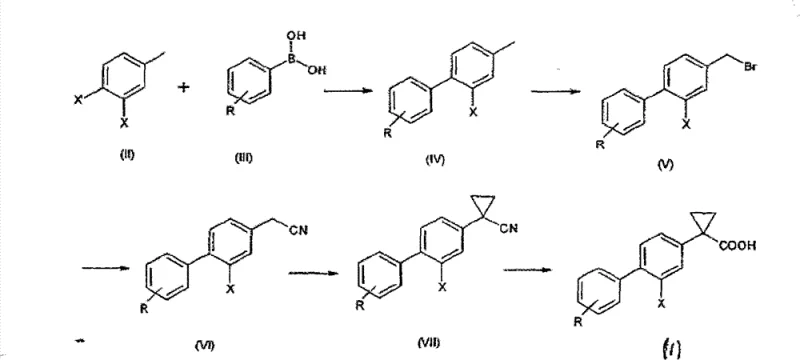
The pharmaceutical industry's relentless pursuit of effective treatments for neurodegenerative disorders, particularly Alzheimer's disease, has placed a premium on the efficient synthesis of gamma-secretase modulators. Patent CN102046576B discloses a groundbreaking methodology for preparing derivatives of 1-(2-halobiphenyl-4-yl)-cyclopropanecarboxylic acid, represented by general formula (I), which serve as critical intermediates in this therapeutic class. Unlike prior art methods that suffered from low yields and environmental hazards, this invention introduces a robust, scalable pathway that prioritizes safety and operational efficiency. The core innovation lies in restructuring the synthetic sequence to perform the biaryl bond formation as the inaugural step, thereby establishing the molecular scaffold early in the process. This strategic shift not only mitigates the formation of hazardous by-products but also facilitates superior purification protocols that are essential for regulatory compliance in active pharmaceutical ingredient (API) manufacturing.
![Chemical structure of specific compound 1-(3',4'-dichloro-2-fluoro[1,1'-biphenyl]-4-yl)-cyclopropanecarboxylic acid](/insights/img/halobiphenyl-cyclopropane-carboxylic-acid-pharma-supplier-20260315090302-06.webp)
For procurement specialists and supply chain directors, the implications of this technological advancement are profound, offering a reliable pharmaceutical intermediates supplier pathway that drastically reduces lead time for high-purity intermediates. By eliminating the reliance on ozone-depleting solvents and cumbersome purification techniques, the process aligns with modern green chemistry principles while simultaneously driving down the cost of goods sold (COGS). The ability to produce these complex scaffolds with an overall yield exceeding 30% represents a significant leap forward from the historical benchmarks of 12-14%, ensuring a more stable and cost-effective supply chain for downstream drug development programs targeting amyloid-beta modulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the state-of-the-art synthesis, as detailed in WO 2004/074232, relied on a linear sequence that initiated with the functionalization of a simple toluene derivative. This conventional approach necessitated a radical bromination step using carbon tetrachloride (CCl4), a solvent now recognized for its severe toxicity, ozone-depleting potential, and status as a greenhouse gas. Furthermore, this early-stage bromination often resulted in the formation of significant quantities of dihalogenated by-products, which detrimentally impacted the overall yield and complicated downstream purification. The subsequent Suzuki coupling, performed late in the sequence, frequently delivered poor yields, and the final isolation of the target molecule required silica gel chromatography. From an industrial perspective, silica gel chromatography is notoriously difficult to scale, requiring vast volumes of solvent and generating substantial solid waste, rendering the process economically unviable for commercial-scale production.
The Novel Approach
The methodology disclosed in CN102046576B fundamentally re-engineers this workflow by executing the Suzuki-Miyaura cross-coupling reaction as the very first step. This strategic inversion allows for the construction of the biphenyl core using commercially available and cost-effective starting materials, such as 4-bromo-3-fluoro-toluene and substituted phenylboronic acids. By establishing the carbon-carbon bond early, the process avoids the harsh conditions associated with early-stage radical bromination on a simple toluene ring. Subsequent steps, including the radical bromination of the methyl group, are performed on the more robust biphenyl scaffold using safer reagents like N-bromosuccinimide (NBS) in acetonitrile. Crucially, the final product can be purified to high chemical purity, typically greater than 95%, through standard crystallization techniques using solvent mixtures like n-heptane and isopropanol, completely obviating the need for chromatographic purification.
Mechanistic Insights into Suzuki-Miyaura Coupling and Radical Functionalization
The cornerstone of this improved synthesis is the initial palladium-catalyzed cross-coupling reaction, which joins the halogenated toluene derivative with a phenylboronic acid species. The patent highlights the preference for heterogeneous catalysts, specifically palladium on carbon (Pd/C), over homogeneous alternatives like tetrakis(triphenylphosphine)palladium. This choice is mechanistically significant as heterogeneous catalysts offer easier separation via filtration, reducing the risk of residual heavy metal contamination in the final API—a critical quality attribute for pharmaceutical regulators. The reaction proceeds efficiently in alcoholic solvents such as ethanol, often in the presence of inorganic bases like sodium carbonate, facilitating the transmetallation step essential for the catalytic cycle. This step typically achieves yields higher than 80%, providing a robust foundation for the subsequent transformations.
Following the biaryl formation, the methyl group on the toluene moiety undergoes selective radical bromination. This transformation is meticulously controlled to minimize over-bromination, utilizing a slight molar excess of NBS (approximately 1.05 equivalents) in the presence of a radical initiator like benzoyl peroxide. The use of acetonitrile as the solvent medium enhances the selectivity for the monobrominated species, yielding the benzyl bromide intermediate in excess of 90%. This intermediate is then converted to a nitrile via nucleophilic substitution with sodium cyanide, followed by a phase-transfer catalyzed cyclopropanation using 1,2-dibromoethane. The final hydrolysis of the nitrile to the carboxylic acid is conducted under basic conditions, completing the synthesis of the target gamma-secretase modulator scaffold with high fidelity and minimal impurity generation.
How to Synthesize 1-(2-Halobiphenyl-4-yl)-Cyclopropanecarboxylic Acid Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and purity at every stage. The process begins with the coupling of the aryl halide and boronic acid, followed by sequential functional group manipulations that build the cyclopropane ring. Each step has been optimized to utilize common industrial reagents and solvents, ensuring that the technology is readily transferable to manufacturing facilities. The detailed standardized synthesis steps, including specific stoichiometry, temperature profiles, and workup procedures, are outlined below to guide process chemists in replicating this high-efficiency route.
- Perform a Suzuki-Miyaura coupling between a dihalo-toluene derivative and a phenylboronic acid using a Pd/C catalyst to form the biphenyl core.
- Conduct radical bromination of the methyl group using N-bromosuccinimide (NBS) and benzoyl peroxide in acetonitrile to generate the benzyl bromide intermediate.
- Convert the benzyl bromide to a nitrile derivative using sodium cyanide, followed by cyclopropanation with 1,2-dibromoethane under phase transfer conditions.
- Hydrolyze the resulting cyclopropanenitrile using a strong base like KOH in methanol/water to yield the final carboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For organizations managing the procurement of complex pharmaceutical intermediates, the adoption of this novel synthesis route offers substantial strategic benefits. The elimination of toxic and regulated solvents like carbon tetrachloride simplifies environmental compliance and reduces the overhead costs associated with hazardous waste disposal. Furthermore, the shift from chromatographic purification to crystallization represents a major operational improvement, as crystallization is inherently more scalable, consumes less solvent, and generates less waste than column chromatography. These factors collectively contribute to a more resilient supply chain capable of meeting the rigorous demands of clinical and commercial drug production.
- Cost Reduction in Manufacturing: The replacement of expensive homogeneous palladium catalysts with heterogeneous Pd/C significantly lowers raw material costs while simplifying catalyst recovery. Additionally, the avoidance of silica gel chromatography removes a major cost driver associated with solvent consumption and stationary phase disposal. The improved overall yield, which more than doubles compared to prior art methods, directly translates to a lower cost per kilogram of the final intermediate, enhancing the economic viability of the entire drug development program.
- Enhanced Supply Chain Reliability: The starting materials for this process, including various substituted phenylboronic acids and halogenated toluenes, are widely available from global chemical suppliers, reducing the risk of raw material shortages. The robustness of the synthetic steps, characterized by high yields and tolerance to minor variations in conditions, ensures consistent production output. This reliability is crucial for maintaining continuous supply lines for long-term clinical trials and eventual commercial launch, minimizing the risk of production delays.
- Scalability and Environmental Compliance: Designed with industrial application in mind, the process utilizes unit operations that are standard in fine chemical manufacturing, such as reflux, filtration, and crystallization. The exclusion of ozone-depleting substances and the reduction of solvent intensity align with increasingly stringent global environmental regulations. This forward-looking approach ensures that the manufacturing process remains compliant with future regulatory frameworks, safeguarding the long-term sustainability of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 1-(2-halobiphenyl-4-yl)-cyclopropanecarboxylic acid derivatives. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, offering clarity on the process capabilities and advantages.
Q: What are the primary advantages of this new synthesis route over previous methods?
A: The new process described in CN102046576B reverses the synthetic order by performing the Suzuki coupling first, which avoids the use of toxic carbon tetrachloride (CCl4) in early stages and eliminates the need for difficult-to-scale silica gel chromatography, resulting in significantly higher overall yields exceeding 30%.
Q: How does the process ensure high purity without chromatography?
A: High chemical purity greater than 95% is achieved through optimized crystallization steps, specifically using solvent systems like n-heptane and isopropanol, which allows for the removal of impurities without the solvent intensity and operational complexity of column chromatography.
Q: Is the catalyst system suitable for large-scale manufacturing?
A: Yes, the process utilizes palladium on carbon (Pd/C), a heterogeneous catalyst that is less expensive and easier to handle and recover than homogeneous palladium complexes like Pd(PPh3)4, making it highly suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(2-Halobiphenyl-4-yl)-Cyclopropanecarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation neurodegenerative therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch supports your regulatory filings and clinical objectives with unwavering consistency.
We invite you to collaborate with us to optimize your supply chain for these vital compounds. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can accelerate your project timelines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
