Scalable Green Synthesis of N-methyl-2-cyano-3-arylpyrrole Intermediates via Molecular Iodine Catalysis
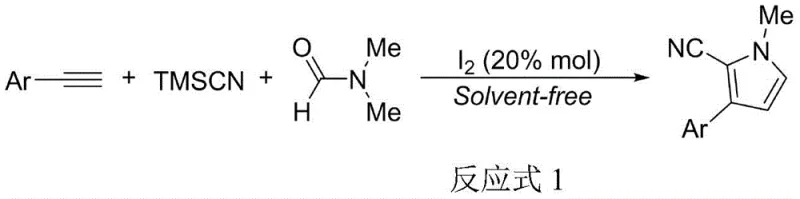
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways for synthesizing complex heterocyclic scaffolds, particularly pyrrole derivatives which serve as critical pharmacophores in anticancer and antibacterial agents. Patent CN110746336B, published in May 2021, introduces a groundbreaking green preparation method for N-methyl-2-cyano-3-arylpyrrole compounds that fundamentally shifts the paradigm from traditional transition-metal catalysis to a benign molecular iodine system. This innovation addresses long-standing challenges in atom economy and waste generation by utilizing a solvent-free, three-component tandem reaction involving terminal aromatic alkynes, trimethylnitrile silane (TMSCN), and N,N-dimethylformamide (DMF). For R&D directors and process chemists, this patent represents a significant leap forward, offering a route that achieves high yields under mild conditions while drastically reducing the Process Mass Intensity (PMI) compared to prior art methods that relied on toxic oxidants and expensive copper catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of polysubstituted pyrroles often relied on harsh conditions that were ill-suited for large-scale industrial application. As illustrated in the background art of the patent, previous methodologies, such as those reported by Professor Wang Shaohua, utilized copper triflate as a catalyst alongside dichloro dicyanoquinone (DDQ) as a stoichiometric oxidant. These conventional routes suffered from severe drawbacks, including the requirement for inert argon atmospheres, prolonged reaction times of up to 24 hours, and the generation of virulent hydrocyanic acid byproducts. Furthermore, the atom efficiency was notoriously low due to the need for excessive equivalents of reagents—often 10 equivalents of TMSCN and 15 equivalents of DMF—leading to massive waste streams. The purification process was equally burdensome, typically necessitating complex chromatographic separation to remove metal residues and side products, which significantly inflated production costs and extended lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel approach disclosed in CN110746336B utilizes a catalytic amount of molecular iodine to drive a highly efficient three-component addition reaction under solvent-free conditions. This method operates at equimolar ratios of the starting materials, representing a dramatic improvement in atom utilization and cost reduction in pharmaceutical intermediate manufacturing. The reaction proceeds smoothly at 80°C, completing in merely 2 hours, which is a twelve-fold reduction in time compared to traditional copper-catalyzed methods. By eliminating the need for transition metals and external oxidants, this process not only simplifies the downstream processing but also enhances the environmental profile of the synthesis. The resulting N-methyl-2-cyano-3-arylpyrrole compounds are obtained with excellent selectivity and can be isolated through simple extraction, bypassing the need for energy-intensive chromatography.
Mechanistic Insights into Iodine-Catalyzed [3+2] Cycloaddition
The mechanistic pathway of this transformation is both elegant and distinct from previous copper-mediated oxidations. Under the action of the molecular iodine catalyst, the trimethylnitrile silane reacts directly with N,N-dimethylformamide to generate a key zwitterionic intermediate, designated as IM1 in the patent literature. This intermediate then undergoes an intermolecular [3+2] cycloaddition with the terminal aromatic alkyne to construct the pyrrole ring system. This mechanism avoids the formation of toxic hydrocyanic acid intermediates seen in older pathways and ensures that the cyano group is incorporated efficiently into the final structure. The use of iodine as a Lewis acid catalyst facilitates the activation of the nitrile species without introducing heavy metal contaminants, which is a critical consideration for regulatory compliance in API synthesis. The robustness of this mechanism allows for a wide range of functional group tolerance, ensuring that sensitive moieties on the aryl ring remain intact during the cyclization process.
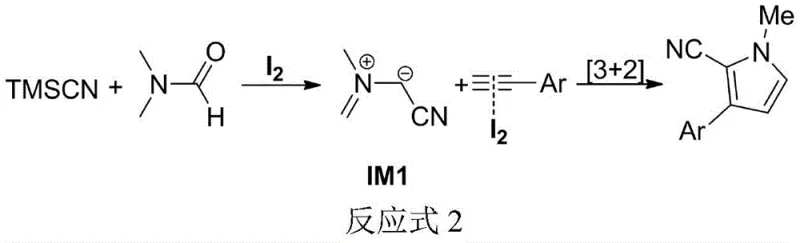
From an impurity control perspective, this mechanism offers substantial advantages for quality assurance teams. Because the reaction does not employ transition metals like copper, the risk of heavy metal leaching into the final product is effectively eliminated, reducing the burden on analytical QC labs to detect trace metals below ppm levels. Additionally, the high regioselectivity of the [3+2] cycloaddition minimizes the formation of structural isomers, leading to a cleaner crude reaction profile. The patent data indicates that even with diverse substrates ranging from electron-rich methoxy-substituted alkynes to electron-deficient nitro-derivatives, the reaction maintains high fidelity. This predictability in impurity profiles simplifies the validation process for commercial scale-up of complex pharmaceutical intermediates, ensuring consistent batch-to-batch quality essential for GMP manufacturing environments.
How to Synthesize N-methyl-2-cyano-3-arylpyrrole Efficiently
The operational simplicity of this synthesis makes it highly attractive for process development teams looking to transfer technology from the lab to the pilot plant. The procedure involves charging a reaction vessel with equimolar amounts of the terminal aryne, TMSCN, and DMF, followed by the addition of 20 mol% molecular iodine. The mixture is then heated to 80°C under solvent-free conditions, where it reacts vigorously to form the target pyrrole. Detailed standardized synthetic steps, including specific workup procedures involving saturated sodium sulfite washing and ethyl acetate extraction, are outlined in the experimental section of the patent to ensure reproducibility.
- Charge a reaction vessel with equimolar amounts of terminal aromatic alkyne, trimethylnitrile silane (TMSCN), and N,N-dimethylformamide (DMF).
- Add molecular iodine catalyst (20 mol%) to the mixture and stir at 80°C under solvent-free conditions for approximately 2 hours.
- Upon completion, wash the reaction system with saturated sodium sulfite solution and extract the aqueous phase with ethyl acetate to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-catalyzed methodology translates into tangible operational efficiencies and risk mitigation. The shift from a solvent-based system to a solvent-free protocol significantly reduces the volume of raw materials required, thereby lowering logistics costs associated with solvent transport and storage. Moreover, the elimination of chromatographic purification steps drastically shortens the production cycle time, allowing for faster turnover of inventory and improved responsiveness to market demand. The use of molecular iodine, a commodity chemical, instead of specialized copper catalysts or toxic oxidants like DDQ, ensures a more stable and cost-effective supply chain, reducing dependency on scarce or regulated reagents. This stability is crucial for maintaining continuous production schedules and avoiding delays caused by raw material shortages.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic reduction in Process Mass Intensity (PMI), which the patent reports decreased by over 85 times compared to prior art. By operating with equimolar reagents and eliminating the need for expensive oxidants and transition metal catalysts, the direct material costs are significantly lowered. Furthermore, the removal of chromatography columns and the associated silica gel or resin consumables results in substantial savings in waste disposal and operational overhead. The high atom efficiency means that a greater proportion of the input mass ends up in the final product, maximizing the return on investment for every kilogram of raw material purchased.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more reliable supply chain by minimizing the risk of batch failures. The tolerance of the method to air and moisture, as evidenced by the experimental data showing no difference between nitrogen and air atmospheres, simplifies the engineering controls required for the reactor setup. This flexibility allows manufacturers to utilize standard equipment without the need for specialized inert gas systems, reducing capital expenditure. Additionally, the broad substrate scope ensures that supply chains for various substituted pyrrole derivatives can be consolidated under a single, versatile manufacturing platform, enhancing overall agility.
- Scalability and Environmental Compliance: Scaling this process to commercial volumes is facilitated by the absence of hazardous solvents and the exothermic nature of the reaction which is easily managed at 80°C. The environmental footprint is further minimized by the generation of benign byproducts that can be treated with standard aqueous workups, aligning with increasingly stringent global environmental regulations. The high EcoScale score reported in the patent underscores the sustainability of this approach, making it an ideal candidate for companies aiming to improve their green chemistry metrics. This alignment with environmental standards not only reduces regulatory risk but also enhances the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this green synthesis technology. These answers are derived directly from the experimental data and claims within patent CN110746336B, providing a reliable foundation for process evaluation. Understanding these specifics is vital for technical teams assessing the feasibility of integrating this route into existing production lines.
Q: What is the optimal catalyst loading for this iodine-catalyzed reaction?
A: According to patent CN110746336B, the optimal loading of molecular iodine is 20 mol% relative to the terminal alkyne. Loadings below 15% result in reduced yields, while increasing beyond 22% offers no significant improvement in conversion.
Q: Does this method require chromatographic purification?
A: No, one of the primary advantages of this green preparation method is that it eliminates the need for column chromatography. The target N-methyl-2-cyano-3-arylpyrrole compounds can be obtained in high purity simply through washing with saturated sodium sulfite and extraction with ethyl acetate.
Q: What is the substrate scope for the terminal aromatic alkyne?
A: The method demonstrates broad substrate tolerance, successfully reacting with phenylacetylenes bearing electron-donating groups (methyl, methoxy), electron-withdrawing groups (fluoro, bromo, iodo, nitro, trifluoromethyl, ester), as well as heterocyclic alkynes like pyridine and thiophene derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-methyl-2-cyano-3-arylpyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iodine-catalyzed synthesis described in CN110746336B for the production of high-value pyrrole intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry are fully realized in a GMP-compliant setting. Our rigorous QC labs and stringent purity specifications guarantee that every batch of N-methyl-2-cyano-3-arylpyrrole meets the exacting standards required for downstream API synthesis, providing our partners with peace of mind regarding quality and consistency.
We invite pharmaceutical and agrochemical companies to leverage our expertise to optimize their supply chains for these critical heterocyclic building blocks. By collaborating with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and innovation in your drug development programs.
