Advanced Synthesis of N-Nitro-N-Allyl Pyridine-2-Amine for High-Performance Energetic Materials
The landscape of energetic material synthesis is undergoing a significant transformation, driven by the urgent need for safer, more selective, and environmentally benign manufacturing processes. Patent CN110540522B introduces a groundbreaking synthetic methodology for producing N-nitro-N-allyl pyridine-2-amine compounds, a class of structures pivotal for developing next-generation high-energy density materials. Unlike conventional approaches that rely on hazardous mixed-acid systems, this innovation employs a mild oxidative nitration strategy using nitrite salts coupled with modern oxidants. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a paradigm shift towards sustainable chemistry. The process operates under remarkably gentle conditions, typically between 0-50°C, ensuring that sensitive functional groups remain untouched while achieving high conversion rates. This technical advancement not only mitigates the safety risks associated with handling explosive nitramine precursors but also streamlines the purification workflow, offering substantial operational advantages for industrial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitramine compounds has been dominated by the use of aggressive nitrating agents such as mixed nitric and sulfuric acids, nitric acid-acetic anhydride complexes, or fuming sulfuric acid mixtures. These traditional methods suffer from severe drawbacks that hinder their applicability in modern fine chemical manufacturing. Firstly, the harsh acidic environment often leads to poor chemoselectivity, resulting in unwanted electrophilic substitution on aromatic rings or the degradation of sensitive alkene moieties like the allyl group. Secondly, the exothermic nature of mixed-acid nitration poses significant safety hazards, requiring specialized corrosion-resistant reactors and rigorous temperature control systems to prevent runaway reactions. Furthermore, the generation of large volumes of acidic waste creates a heavy environmental burden, necessitating costly neutralization and disposal procedures. For a procurement manager, these factors translate into higher capital expenditure for equipment, increased operational costs for waste management, and potential supply chain disruptions due to stringent environmental regulations.
The Novel Approach
The methodology disclosed in CN110540522B elegantly circumvents these challenges by replacing strong mineral acids with a combination of a nitro source (such as sodium nitrite or silver nitrite) and a mild oxidant (like N-chlorosuccinimide or potassium persulfate). This system generates the active nitrating species in situ under neutral or weakly acidic conditions, preserving the integrity of the substrate. As illustrated in the general reaction scheme below, the transformation proceeds with exceptional specificity, targeting the amine nitrogen exclusively while leaving the carbon-carbon double bond of the allyl group completely intact.
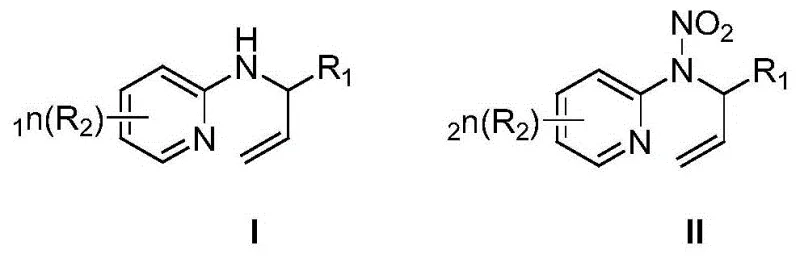
This chemoselectivity is a critical advantage, as it eliminates the need for protecting group strategies that add steps and cost to the synthesis. The reaction typically completes within 2 to 6 hours at temperatures ranging from 0°C to 50°C, drastically reducing energy consumption compared to refluxing acid conditions. Moreover, the workup procedure is simplified to a standard silica gel column chromatography, avoiding the complex extraction and neutralization steps required for acid-based methods. This streamlined approach facilitates cost reduction in energetic material manufacturing by minimizing raw material usage, lowering energy inputs, and reducing the time required for batch turnover.
Mechanistic Insights into Oxidative Nitrosation
The core of this innovation lies in the mechanistic pathway of oxidative nitrosation. In the presence of an oxidant such as N-chlorosuccinimide (NCS), the nitrite anion is activated to form a reactive nitro-species, likely a nitro radical or an electrophilic nitronium equivalent, which attacks the lone pair of the secondary amine nitrogen. This mechanism differs fundamentally from the electrophilic aromatic substitution seen in mixed-acid nitration. Because the reaction center is the nucleophilic nitrogen rather than the electron-rich aromatic ring or alkene, the process exhibits remarkable functional group tolerance. The patent data demonstrates that various substituents on the phenyl ring, including electron-withdrawing groups like cyano and nitro, as well as electron-donating groups like methoxy and methyl, are well-tolerated. This broad substrate scope is essential for creating a diverse library of derivatives for structure-activity relationship studies.
Furthermore, the preparation of the starting material, N-(1-phenylallyl)pyridin-2-amine, is itself a robust two-step sequence involving condensation followed by Grignard addition, as depicted in the synthesis route below. This ensures that high-purity precursors are readily available, which is a prerequisite for achieving the high yields (up to 88%) reported in the nitration step.
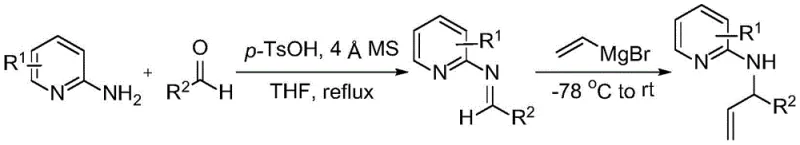
Impurity control is another critical aspect addressed by this mechanism. Traditional nitration often produces poly-nitrated byproducts or ring-nitrated impurities that are difficult to separate due to similar polarity. In contrast, the mild oxidative conditions minimize side reactions, resulting in a cleaner crude product profile. The absence of strong acids also prevents the hydrolysis of the allyl group or the pyridine ring, which can occur under harsh acidic conditions. This purity profile is vital for downstream applications, particularly in the synthesis of energetic materials where trace impurities can affect stability and performance. The ability to subsequently reduce the nitramine to a hydrazine derivative, as shown in the following transformation, further underscores the versatility of this scaffold in medicinal and materials chemistry.
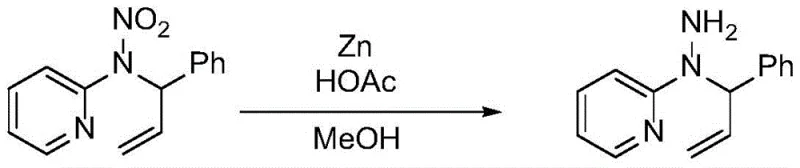
How to Synthesize N-Nitro-N-Allyl Pyridine-2-Amine Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction monitoring. The patent specifies a molar ratio of substrate to nitro source to oxidant of approximately 1:2:2 for optimal results, although ratios ranging from 1:1:1 to 1:3:3 are feasible. Acetonitrile is identified as the preferred solvent due to its ability to dissolve both organic substrates and inorganic salts effectively. The detailed standardized synthesis steps, including precise quenching and purification protocols, are outlined in the guide below to ensure reproducibility and safety.
- Prepare the N-(1-phenylallyl)pyridin-2-amine substrate via condensation and Grignard addition.
- Mix the amine substrate with a nitro source (e.g., sodium nitrite) and an oxidant (e.g., N-chlorosuccinimide) in acetonitrile.
- Stir the reaction mixture at 0-50°C for 2-6 hours, then purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this synthetic route offers compelling economic and logistical benefits beyond mere technical superiority. The shift from hazardous mixed acids to solid or soluble oxidants and nitrites simplifies raw material sourcing and storage. Solid reagents like N-chlorosuccinimide and sodium nitrite are stable, easy to handle, and do not require the specialized tank farms needed for bulk sulfuric or nitric acid. This flexibility enhances supply chain reliability by reducing dependency on bulk acid suppliers who may be subject to seasonal demand fluctuations or regulatory shipping restrictions. Additionally, the mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors without the need for exotic alloys resistant to hot concentrated acids, thereby lowering capital investment barriers for contract manufacturing organizations.
- Cost Reduction in Manufacturing: The elimination of corrosive mixed acids removes the significant costs associated with acid-resistant equipment maintenance and replacement. Furthermore, the simplified workup procedure, which avoids extensive aqueous washing and neutralization, reduces water consumption and wastewater treatment costs. The high selectivity of the reaction minimizes the formation of hard-to-remove impurities, leading to higher overall yields and reduced loss of valuable starting materials. By avoiding the need for cryogenic conditions often required for controlling exothermic acid nitrations, the process also achieves substantial energy savings, contributing to a lower carbon footprint and reduced utility expenses.
- Enhanced Supply Chain Reliability: The use of commodity chemicals like sodium nitrite and common organic oxidants ensures a stable and diversified supply base. Unlike specialized nitrating agents that may have limited suppliers, the reagents used in this protocol are widely available globally. The robustness of the reaction across a wide temperature range (0-50°C) provides operational flexibility, allowing manufacturers to adapt to varying ambient conditions without compromising product quality. This resilience is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage is straightforward due to the absence of violent exotherms and gas evolution typically seen in acid nitrations. The reaction can be safely performed in larger batches with standard cooling systems. From an environmental perspective, the process generates significantly less hazardous waste. The absence of spent sulfuric acid sludge simplifies waste disposal and aligns with increasingly strict global environmental regulations. This compliance advantage reduces the risk of regulatory fines and shutdowns, ensuring long-term operational continuity for commercial scale-up of complex heterocyclic intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitration technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on safety, efficiency, and applicability for potential partners.
Q: Why is this nitration method safer than traditional mixed acid nitration?
A: Traditional mixed acid nitration involves strong corrosives and high exothermic risks. This patent utilizes mild oxidants like N-chlorosuccinimide with nitrites at near-room temperature, significantly reducing thermal hazards and equipment corrosion.
Q: Does this method preserve the allyl double bond during nitration?
A: Yes, the method exhibits excellent chemoselectivity. Unlike electrophilic nitration which often attacks electron-rich alkenes, this oxidative protocol specifically targets the amine nitrogen, leaving the sensitive allyl group intact for further functionalization.
Q: What represents the primary cost driver in scaling this process?
A: The primary cost drivers are the stoichiometric oxidant and nitrite salt. However, the elimination of expensive acid waste treatment and the high yield (up to 88%) offset reagent costs, making it economically viable for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Nitro-N-Allyl Pyridine-2-Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced synthetic methodologies like the one described in CN110540522B for the development of high-performance materials. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of nitramine derivatives or large-scale supply of key intermediates, our infrastructure is designed to meet the demanding standards of the global pharmaceutical and agrochemical industries.
We invite you to collaborate with us to leverage this innovative technology for your specific applications. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you optimize your supply chain and accelerate your product development timeline.
