Scalable Asymmetric Synthesis of (-)-Kainic Acid via Rhodium-Catalyzed Addition for Pharmaceutical Applications
Introduction to Advanced Asymmetric Synthesis of (-)-Kainic Acid
The preparation of optically active amino acids remains a cornerstone challenge in modern pharmaceutical chemistry, particularly for complex marine natural products like (-)-Kainic Acid. Patent CN103373948A introduces a groundbreaking methodology that addresses the longstanding limitations of traditional extraction and synthesis. This technology leverages a highly efficient six-step synthetic pathway, achieving a remarkable total yield of 40% while maintaining exceptional stereochemical integrity. The core innovation lies in the utilization of rhodium-catalyzed enantioselective 1,4-addition reactions combined with substrate-induced asymmetric protonation. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this patent represents a paradigm shift from resource-intensive isolation to precise, scalable chemical manufacturing. The process not only ensures high enantiomeric excess but also simplifies the operational complexity typically associated with poly-substituted proline derivatives.
Historically, (-)-Kainic Acid has been valued as a potent agonist for L-glutamate receptors, serving as a critical tool in neuroscience research for studying epilepsy, Alzheimer's disease, and Huntington's disease. However, reliance on marine extraction has resulted in volatile pricing and inconsistent supply chains. The synthetic approach detailed in this patent mitigates these risks by establishing a robust, reproducible chemical route. By starting from simple, commercially available substrates and employing sophisticated chiral catalysis, the method delivers optical purity that rivals natural sources. This transition from biological sourcing to chemical synthesis is essential for securing the commercial scale-up of complex pharmaceutical intermediates required by the global neuropharmacology market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for obtaining (-)-Kainic Acid have predominantly relied on extraction from marine plants such as Digenea simplex. This biological sourcing is inherently flawed due to seasonal variability, geographical constraints, and the labor-intensive nature of isolation processes. Furthermore, previous chemical synthesis attempts reported in literature often suffered from excessive step counts, frequently exceeding ten steps, which drastically reduced overall throughput and increased waste generation. Low diastereoselectivity in earlier routes necessitated cumbersome purification protocols, often involving repeated recrystallizations or preparative HPLC, which are economically unviable for large-scale production. These inefficiencies created a bottleneck for researchers requiring high-purity materials for receptor binding studies and drug discovery programs.
The Novel Approach
The novel approach presented in patent CN103373948A circumvents these historical bottlenecks through a concise six-step sequence that prioritizes atom economy and stereocontrol. The strategy initiates with a rhodium-catalyzed conjugate addition, establishing the first chiral center with high fidelity, followed by a substrate-controlled protonation to set the second center. This dual-asymmetric induction strategy eliminates the need for resolution steps that typically halve yields in racemic syntheses. The route is designed for operational simplicity, utilizing standard organic solvents and reagents that are readily accessible in industrial settings. 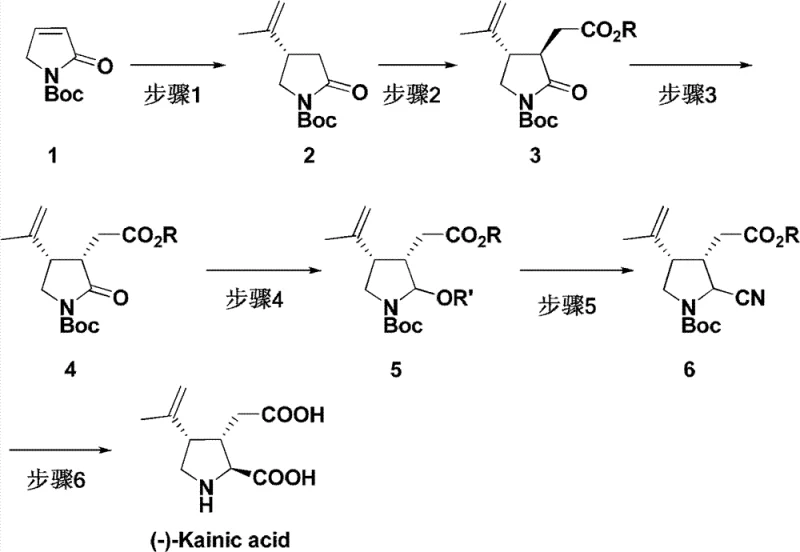 This streamlined pathway significantly reduces the cost reduction in pharmaceutical intermediate manufacturing by minimizing unit operations and maximizing material throughput at each stage. The ability to generate the target molecule with three adjacent chiral centers in such a short sequence demonstrates a superior level of synthetic design compared to legacy methods.
This streamlined pathway significantly reduces the cost reduction in pharmaceutical intermediate manufacturing by minimizing unit operations and maximizing material throughput at each stage. The ability to generate the target molecule with three adjacent chiral centers in such a short sequence demonstrates a superior level of synthetic design compared to legacy methods.
Mechanistic Insights into Rh-Catalyzed Asymmetric 1,4-Addition
The heart of this synthetic innovation is the rhodium-catalyzed asymmetric 1,4-addition of alkenyl boron reagents to unsaturated five-membered lactams. This transformation is mediated by chiral rhodium complexes, specifically dimeric structures such as [Rh(OH)L]2 or [Rh(Cl)L]2, where L represents a chiral diene ligand. The choice of ligand is critical, as it dictates the facial selectivity of the olefin insertion into the rhodium-alkenyl bond. The reaction proceeds under mild conditions, typically between 25°C and 60°C, using organic bases like triethylamine or DIPEA to facilitate transmetallation. The use of potassium alkenyltrifluoroborates as nucleophiles offers enhanced stability and reactivity compared to traditional boronic acids, ensuring consistent performance across different batches. 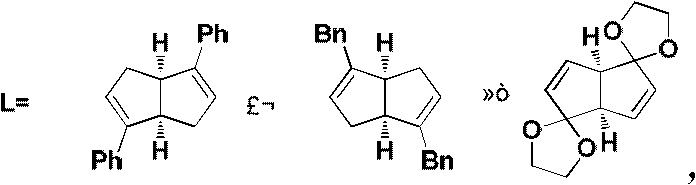 This catalytic cycle is highly efficient, requiring only 3-10 mol% of the rhodium catalyst to drive the reaction to completion within 24 to 72 hours.
This catalytic cycle is highly efficient, requiring only 3-10 mol% of the rhodium catalyst to drive the reaction to completion within 24 to 72 hours.
Following the initial addition, the synthesis employs a substrate-induced asymmetric protonation to install the second stereocenter. This step involves the generation of an enolate intermediate using strong bases such as LiHMDS or NaHMDS at cryogenic temperatures (-78°C to 0°C). The subsequent quenching with a chiral proton source, preferably (-)-camphorsulfonic acid, ensures that the protonation occurs from the less hindered face of the enolate, guided by the existing chirality of the molecule. 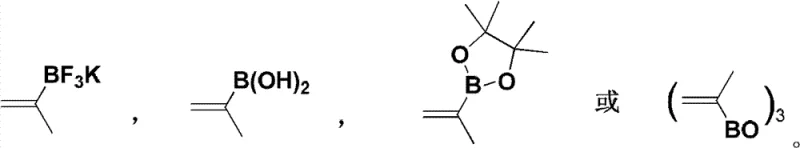 The precision of this protonation step is vital for controlling the relative stereochemistry between the C3 and C4 positions of the pyrrolidine ring. By combining metal-catalyzed C-C bond formation with organocatalytic-like proton transfer, the process achieves a level of impurity control that is difficult to replicate with non-catalytic methods. This mechanistic elegance translates directly into higher purity profiles, reducing the burden on downstream purification teams.
The precision of this protonation step is vital for controlling the relative stereochemistry between the C3 and C4 positions of the pyrrolidine ring. By combining metal-catalyzed C-C bond formation with organocatalytic-like proton transfer, the process achieves a level of impurity control that is difficult to replicate with non-catalytic methods. This mechanistic elegance translates directly into higher purity profiles, reducing the burden on downstream purification teams.
How to Synthesize (-)-Kainic Acid Efficiently
Executing this synthesis requires careful attention to reaction conditions, particularly temperature control during the enolization and protonation steps. The process begins with the preparation of the chiral rhodium catalyst in situ or using pre-formed complexes, followed by the addition of the unsaturated lactam substrate and the alkenyl boron reagent in a biphasic or homogeneous solvent system. Subsequent steps involve sequential functional group manipulations including alkylation, reduction, and cyanation, all of which are optimized for high conversion rates. The final deprotection and hydrolysis steps utilize aqueous conditions to reveal the free amino acid functionality. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized protocol below which outlines the critical process parameters for each transformation.
- Perform Rh-catalyzed asymmetric 1,4-addition of alkenyl boron reagents to unsaturated lactam to form the first chiral center.
- Execute alpha-alkylation with alkyl alpha-haloacetate followed by kinetic protonation to introduce the second chiral center.
- Complete the synthesis through carbonyl reduction, acetal formation, cyanation, and final hydrolysis to yield optically pure (-)-Kainic Acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this synthetic route offers profound strategic benefits beyond mere technical feasibility. The primary advantage lies in the decoupling of supply from biological sources, which are subject to environmental fluctuations and regulatory scrutiny regarding marine harvesting. By adopting a fully synthetic pathway, organizations can secure a stable, year-round supply of (-)-Kainic Acid that is immune to seasonal disruptions. Furthermore, the use of commodity chemicals and standard catalytic systems means that raw material costs are predictable and manageable, avoiding the price volatility associated with rare natural extracts. This stability is crucial for long-term project planning and budget forecasting in drug development pipelines.
- Cost Reduction in Manufacturing: The streamlined six-step sequence significantly lowers the cost of goods sold (COGS) by reducing the number of isolation and purification events. Traditional routes often require extensive chromatographic separations after every step, which consumes vast amounts of silica gel and solvents. In contrast, this patent describes efficient workups involving extractions and crystallizations that are more amenable to bulk processing. The elimination of transition metal removal steps, often required in palladium or nickel catalysis, further simplifies the downstream processing, as the rhodium catalyst loading is low and the metal can be effectively scavenged or left within acceptable limits for intermediate use. These cumulative efficiencies result in substantial cost savings without compromising quality.
- Enhanced Supply Chain Reliability: The reliance on commercially available reagents such as potassium isopropenyltrifluoroborate and standard protecting group reagents (Boc anhydride derivatives) ensures that the supply chain is robust and resilient. There is no dependency on custom-synthesized starting materials that might have long lead times. The synthetic intermediates generated in steps 2 through 5 are stable and can be stockpiled if necessary, providing a buffer against demand spikes. This flexibility allows suppliers to respond rapidly to urgent requests from research institutions, thereby reducing lead time for high-purity pharmaceutical intermediates and strengthening customer relationships through consistent delivery performance.
- Scalability and Environmental Compliance: From an environmental perspective, the process is designed with green chemistry principles in mind. The high atom economy of the addition reactions minimizes waste generation, and the use of recyclable solvents like toluene and ethyl acetate aligns with modern sustainability goals. The final purification via ion exchange resin is a scalable technique widely used in the amino acid industry, avoiding the need for preparative HPLC which is difficult to scale. This makes the process ideal for commercial scale-up of complex pharmaceutical intermediates from pilot plant to multi-ton production. The reduced solvent intensity and waste profile also lower the environmental compliance burden, facilitating easier permitting and operation in regulated manufacturing zones.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. They are derived from the specific advantages and operational details outlined in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these nuances is key to successfully integrating this methodology into existing production workflows.
Q: What are the primary advantages of this synthetic route over natural extraction?
A: Unlike natural extraction which is costly and supply-limited, this synthetic method offers a consistent, scalable supply chain with high optical purity (up to 99% ee) and a streamlined 6-step process.
Q: How is stereocontrol achieved in the key steps?
A: Stereocontrol is primarily achieved through a chiral rhodium catalyst in the initial 1,4-addition step and subsequently reinforced by substrate-induced asymmetric protonation using chiral acids like camphorsulfonic acid.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes commercially available reagents and standard organic transformations such as column chromatography and ion exchange resin purification, making it highly amenable to scale-up from kilogram to multi-ton levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (-)-Kainic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in advancing neurological research and drug discovery. Our team of expert chemists has thoroughly analyzed the synthetic route described in patent CN103373948A and is fully prepared to execute this chemistry at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee enantiomeric excess levels that match or exceed the patent examples. We are committed to being a partner that delivers both technical excellence and supply security.
We invite you to engage with our technical sales team to discuss how this advanced synthesis can optimize your supply chain. Whether you require custom kilogram quantities for preclinical studies or metric ton volumes for commercial API manufacturing, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you secure a reliable source of (-)-Kainic Acid that drives your research forward without supply chain interruptions.
