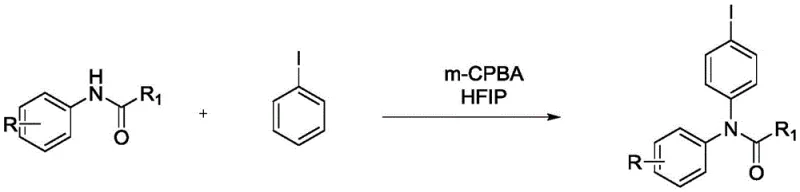
Scalable Metal-Free Synthesis of N-(4-Iodophenyl) Amides for Pharma Intermediates
The pharmaceutical and agrochemical industries constantly seek robust, scalable pathways to access functionalized aryl amines, particularly those bearing halogen handles for subsequent cross-coupling reactions. Patent CN111646917A introduces a significant advancement in this domain by detailing a preparation method for iodobenzene para-aminated compounds, specifically N-(4-iodophenyl) amides. This technology leverages a metal-free oxidative system promoted by meta-chloroperoxybenzoic acid (m-CPBA) in hexafluoroisopropanol (HFIP). Unlike traditional methods that rely on expensive and waste-generating hypervalent iodine reagents, this approach utilizes simple iodobenzene as the substrate, activated in situ by the peracid oxidant. For R&D directors and process chemists, this represents a pivotal shift towards more atom-economical and environmentally benign synthesis of key intermediates used in drug discovery and development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct C-H amination of aromatic rings to install nitrogen functionality at specific positions has been a formidable challenge, often requiring harsh conditions or precious metal catalysts. Traditional routes to N-aryl amides frequently employ stoichiometric amounts of hypervalent iodine(III) reagents, such as diacetoxyiodobenzene (PhI(OAc)2), to activate the aromatic ring. While effective, these reagents are costly to manufacture and generate stoichiometric amounts of iodobenzene waste upon reduction, leading to poor atom economy and increased disposal costs. Furthermore, many metal-catalyzed alternatives require rigorous exclusion of air and moisture, along with complex ligand systems that can be difficult to remove from the final API intermediate, posing significant risks for supply chain continuity and regulatory compliance regarding heavy metal residues.
The Novel Approach
The methodology described in the patent data offers a transformative solution by replacing stoichiometric hypervalent iodine sources with a catalytic-like activation of simple iodobenzene using m-CPBA. In this novel system, the peracid serves as the terminal oxidant to generate the active iodine species in situ, which then facilitates the para-selective amination by the acetanilide nucleophile. This shift drastically reduces the raw material costs associated with specialized iodine reagents and simplifies the reaction profile. By operating in HFIP, a solvent known for stabilizing cationic intermediates and enhancing electrophilicity, the reaction achieves high yields under mild thermal conditions (40-80°C). This approach not only improves the environmental footprint by minimizing heavy metal usage but also streamlines the workup procedure, making it highly attractive for cost-sensitive commercial manufacturing.
Mechanistic Insights into m-CPBA Promoted Oxidative Amination
The core of this chemical transformation lies in the unique interaction between the peracid oxidant and the iodobenzene substrate within the HFIP solvent matrix. Mechanistically, m-CPBA is believed to oxidize the iodobenzene to a reactive hypervalent iodine(III) species, potentially an iodosylbenzene or related cationic intermediate, which is stabilized by the strong hydrogen-bond donating capability of HFIP. This activated species becomes highly electrophilic at the para-position relative to the iodine atom, inviting nucleophilic attack by the nitrogen lone pair of the substituted acetanilide. The use of HFIP is critical, as it lowers the energy barrier for this oxidative addition step and suppresses side reactions, ensuring that the reaction proceeds with high regioselectivity. This precise control over the reaction pathway allows for the consistent formation of the desired N-(4-iodophenyl) bond without significant ortho-substitution or over-oxidation byproducts.
From an impurity control perspective, the metal-free nature of this catalytic cycle is a substantial advantage for pharmaceutical applications. The absence of transition metals like palladium or copper eliminates the risk of metal leaching into the final product, a common bottleneck in API synthesis that requires expensive scavenging steps. The primary byproducts of this reaction are meta-chlorobenzoic acid (from the reduced m-CPBA) and water, both of which are easily removed during the aqueous workup and extraction phases described in the patent examples. This clean reaction profile ensures that the crude product obtained after solvent removal is of high purity, reducing the burden on downstream purification processes like column chromatography or recrystallization, thereby enhancing the overall throughput and yield of the manufacturing process.
How to Synthesize N-(4-Iodophenyl) Amides Efficiently
The synthesis protocol outlined in the patent provides a straightforward, operationally simple procedure suitable for both laboratory scale-up and industrial adaptation. The process begins by charging a reactor with the specific substituted acetanilide, iodobenzene, m-CPBA, and hexafluoroisopropanol in defined molar ratios. The mixture is then subjected to controlled heating under reflux conditions, allowing the oxidative coupling to proceed to completion over a period of 4 to 10 hours. Following the reaction, a standard aqueous workup involving extraction with organic solvents such as ethyl acetate effectively isolates the crude material. For detailed operational parameters, safety considerations, and specific stoichiometric optimizations tailored to your specific substrate, please refer to the standardized synthesis guide below.
- Charge reactor with substituted acetanilide, iodobenzene, m-CPBA, and hexafluoroisopropanol (HFIP).
- Heat the mixture to 40-80°C under reflux with magnetic stirring for 4-10 hours.
- Quench with water, extract with organic solvent, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this m-CPBA promoted synthesis route offers tangible benefits in terms of cost stability and supplier reliability. By eliminating the dependency on scarce and volatile transition metal catalysts, manufacturers can insulate their production costs from the fluctuations of the precious metals market. The use of commodity chemicals like iodobenzene and m-CPBA ensures a robust supply chain with multiple global sourcing options, reducing the risk of single-source bottlenecks. Furthermore, the mild reaction conditions translate to lower energy consumption and reduced wear on reactor equipment, contributing to significant long-term operational expenditure savings. The simplified purification process also means faster batch turnover times, enabling suppliers to respond more agilely to fluctuating market demands for these critical intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive hypervalent iodine reagents with inexpensive iodobenzene and m-CPBA fundamentally alters the cost structure of the synthesis. Since the oxidant and substrate are commodity chemicals available in bulk quantities, the raw material costs are drastically lower compared to traditional methods. Additionally, the removal of transition metal catalysts eliminates the need for costly metal scavenger resins and the associated validation testing for residual metals, further driving down the total cost of goods sold. This economic efficiency allows for more competitive pricing strategies when supplying high-purity pharmaceutical intermediates to downstream clients.
- Enhanced Supply Chain Reliability: Sourcing complex ligands or specialized metal catalysts often involves long lead times and geopolitical risks. In contrast, the reagents required for this process—iodobenzene, m-CPBA, and HFIP—are widely produced by major chemical manufacturers globally. This abundance ensures a continuous and reliable supply of raw materials, minimizing the risk of production stoppages due to material shortages. The robustness of the chemistry also means that the process is less sensitive to minor variations in reagent quality, providing a buffer against supply chain disruptions and ensuring consistent delivery schedules for critical project milestones.
- Scalability and Environmental Compliance: The metal-free nature of this reaction aligns perfectly with increasingly stringent environmental regulations regarding heavy metal discharge and waste management. Scaling this process from kilograms to metric tons does not require complex engineering controls for metal containment or specialized waste treatment facilities. The byproducts are organic acids that can be managed through standard waste streams, simplifying the environmental permitting process for new production lines. This ease of scale-up ensures that the technology can seamlessly transition from pilot plant studies to full commercial production without significant re-engineering, securing the long-term viability of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative amination technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, aiming to clarify the feasibility and advantages of adopting this route for your specific application needs. Understanding these nuances is essential for making informed decisions about process integration and vendor selection.
Q: What are the advantages of using m-CPBA over hypervalent iodine reagents?
A: Using m-CPBA with simple iodobenzene avoids the high cost and waste generation associated with stoichiometric hypervalent iodine reagents like PhI(OAc)2, significantly improving atom economy.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the reaction operates at mild temperatures (40-80°C) without transition metals, simplifying downstream purification and making it highly scalable for industrial production.
Q: What is the regioselectivity of this amination reaction?
A: The method exhibits high para-selectivity, specifically functionalizing the para-position of the iodobenzene ring to form N-(4-iodophenyl) derivatives exclusively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(4-Iodophenyl) Amides Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-(4-iodophenyl) amides meets the exacting standards required for pharmaceutical synthesis. Our commitment to quality and consistency makes us a trusted partner for companies seeking to secure their supply of complex fine chemical intermediates.
We invite you to leverage our technical expertise to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your project timelines and quality objectives. Let us collaborate to bring your next generation of therapeutics to market faster and more efficiently.
