Scalable Manufacturing of Alpha Alpha Terpyridine for Advanced Coordination Chemistry
Scalable Manufacturing of Alpha Alpha Terpyridine for Advanced Coordination Chemistry
The landscape of fine chemical manufacturing is constantly evolving to meet the rigorous demands of modern coordination chemistry and pharmaceutical development. A significant breakthrough in this domain is documented in patent CN113354573A, which details a robust method for the large-scale production of alpha, alpha-terpyridine (CAS 1148-79-4). This heterocyclic compound serves as a pivotal tridentate ligand, renowned for its ability to form stable complexes with various transition metals, making it indispensable in fields ranging from antitumor drug design to the engineering of advanced optical materials and photosensitizers for solar energy conversion. The structural integrity and electronic properties of alpha, alpha-terpyridine allow it to act as both an electron donor and acceptor, facilitating its widespread application in supramolecular chemistry and biological probing systems where high purity is non-negotiable.
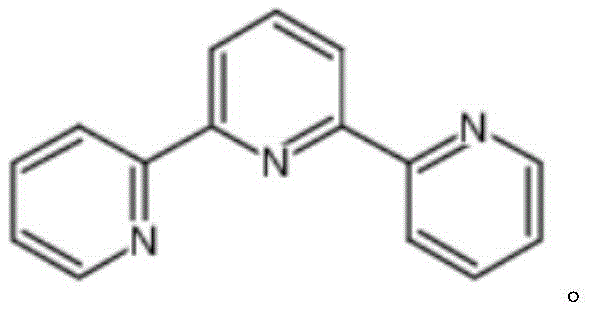
Historically, the synthesis of terpyridine derivatives has been plagued by significant technical bottlenecks that hindered their widespread commercial adoption. Conventional methodologies predominantly relied on cross-coupling reactions that necessitated the use of highly toxic tin reagents and expensive palladium catalysts. These traditional approaches not only incurred substantial raw material costs but also introduced severe downstream processing challenges, particularly regarding the removal of trace metal residues which are unacceptable in pharmaceutical and electronic grade applications. Furthermore, earlier attempts at base-mediated synthesis, such as those disclosed in CN109096339A, suffered from critically low yields of approximately 15% and required labor-intensive flash column chromatography for purification, rendering them economically unviable for kilogram-to-ton scale manufacturing operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on transition metal catalysis in legacy synthetic routes presents a multifaceted problem for supply chain managers and quality assurance teams alike. The use of palladium and tin introduces a high risk of heavy metal contamination, requiring sophisticated and costly purification steps such as scavenging resins or repeated recrystallizations to meet stringent regulatory limits. Additionally, the environmental footprint of these processes is considerable due to the toxicity of the reagents and the generation of hazardous waste streams. From an economic perspective, the volatility of precious metal prices directly impacts the cost of goods sold, creating financial instability for long-term procurement contracts. The low efficiency of previous non-metallic routes, characterized by poor conversion rates and the necessity for chromatographic separation, further exacerbates these issues by limiting throughput and increasing solvent consumption.
The Novel Approach
In stark contrast, the methodology outlined in patent CN113354573A offers a transformative solution by employing a metal-free organocatalytic strategy combined with efficient base-mediated cyclization. This innovative route utilizes readily available guanidine derivatives, such as guanylurea sulfate, to catalyze the formation of a key enamine intermediate, thereby bypassing the need for toxic tin reagents entirely. The subsequent cyclization step leverages potassium tert-butoxide in anhydrous tetrahydrofuran, followed by a streamlined workup involving ammonium acetate and acetic acid. This approach not only simplifies the operational workflow but also dramatically enhances the overall process mass intensity, allowing for the direct isolation of high-purity product through simple recrystallization rather than complex chromatography.
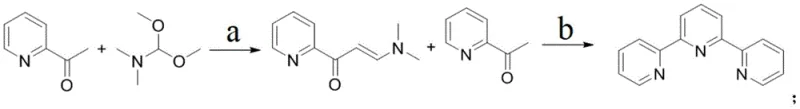
Mechanistic Insights into Guanidine-Catalyzed Enamine Formation and Cyclization
The core of this technological advancement lies in the precise mechanistic control exerted during the initial enamine formation step. The utilization of guanidine-based catalysts facilitates the condensation of 2-acetylpyridine with N,N-dimethylformamide dimethyl acetal under mild reflux conditions. These organic bases act as efficient proton shuttles, activating the carbonyl group of the acetylpyridine while stabilizing the transition state, which leads to the selective formation of 1-(3-pyridyl)-3-(dimethylamino)-2-propen-1-one. The choice of solvent plays a critical role here; the patent highlights that while tetrahydrofuran is effective, switching to chloroform or toluene can significantly boost the molar yield of this intermediate from roughly 42% to nearly 90%, demonstrating the tunability of the reaction parameters for optimal performance.
Following the isolation of the enamine intermediate, the second stage involves a sophisticated cascade of aldol-like condensation and cyclization events. The addition of potassium tert-butoxide generates a reactive enolate species from a second equivalent of 2-acetylpyridine, which then attacks the electrophilic carbon of the enamine intermediate. The subsequent introduction of ammonium acetate and acetic acid creates a buffered acidic environment that promotes the dehydration and aromatization necessary to close the central pyridine ring. This careful modulation of pH and reaction conditions ensures that side reactions are minimized, resulting in a crude product profile that is clean enough to yield 99.4% pure alpha, alpha-terpyridine after a single recrystallization from n-heptane, effectively eliminating the need for silica gel chromatography.
How to Synthesize Alpha Alpha Terpyridine Efficiently
Implementing this synthesis requires strict adherence to the optimized conditions regarding stoichiometry and solvent ratios to maximize the reported 68.4% total yield. The process begins with the preparation of the enamine precursor using a guanidine catalyst in a refluxing organic solvent, followed by isolation via extraction and recrystallization. The second phase involves the careful addition of strong base and the intermediate to anhydrous THF, followed by the acidic cyclization step. For detailed operational parameters, safety guidelines, and specific temperature profiles required to replicate this high-efficiency pathway in a GMP environment, please refer to the standardized technical protocol provided below.
- React 2-acetylpyridine with N,N-dimethylformamide dimethyl acetal using a guanidine catalyst in refluxing solvent to form the enamine intermediate.
- Treat the intermediate with potassium tert-butoxide and additional 2-acetylpyridine in anhydrous THF to initiate condensation.
- Complete the cyclization by adding ammonium acetate and acetic acid under reflux, followed by recrystallization from n-heptane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from metal-catalyzed coupling to this organocatalytic route represents a significant opportunity for cost optimization and risk mitigation. By eliminating the dependency on palladium and tin, manufacturers can decouple their production costs from the volatile precious metals market, leading to more stable pricing structures for long-term contracts. The removal of heavy metal catalysts also drastically reduces the complexity of the purification train, shortening the batch cycle time and reducing the consumption of specialized scavenging materials. This streamlining of the downstream process translates directly into enhanced throughput capabilities, allowing suppliers to respond more agilely to fluctuating market demands without compromising on the stringent purity specifications required by the pharmaceutical and electronic sectors.
- Cost Reduction in Manufacturing: The patent explicitly discloses that this method can reduce production costs by at least 40% compared to prior art, primarily driven by the substitution of expensive noble metal catalysts with inexpensive organocatalysts and the elimination of chromatographic purification. The high molar yield of the intermediate step, which can reach up to 89.2% when using chloroform as a solvent, further contributes to raw material efficiency, ensuring that less starting material is wasted to side products. Additionally, the ability to use common industrial solvents like toluene and THF, which are easily recovered and recycled, minimizes waste disposal costs and aligns with green chemistry principles.
- Enhanced Supply Chain Reliability: Sourcing high-purity ligands often faces bottlenecks due to the limited number of vendors capable of removing trace metal impurities to ppb levels. This synthetic route inherently avoids the introduction of such impurities, thereby reducing the risk of batch rejection and supply disruption. The robustness of the reaction conditions, which operate at ambient pressure and moderate temperatures, allows for production in standard glass-lined or stainless steel reactors, broadening the pool of qualified contract manufacturing organizations (CMOs) capable of executing the synthesis. This diversification of manufacturing capacity ensures a more resilient supply chain for critical intermediates used in high-value applications.
- Scalability and Environmental Compliance: The transition from laboratory scale to industrial production is facilitated by the simplicity of the workup procedures, which rely on standard liquid-liquid extraction and crystallization rather than specialized equipment. The patent examples demonstrate successful execution on a multi-kilogram scale, proving the feasibility of scaling from 100 kgs to 100 MT annual production volumes. Furthermore, the avoidance of toxic tin reagents simplifies environmental compliance and wastewater treatment, reducing the regulatory burden on manufacturing sites and supporting corporate sustainability goals regarding hazardous waste reduction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of alpha, alpha-terpyridine synthesized via this novel pathway. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a transparent view of the technology's capabilities. Understanding these nuances is essential for R&D teams evaluating this intermediate for new drug candidates or material science applications.
Q: How does this method improve upon traditional palladium-catalyzed coupling?
A: Traditional methods rely on toxic tin reagents and expensive palladium catalysts which leave difficult-to-remove metal residues. This novel route utilizes organocatalysts and base-mediated cyclization, eliminating heavy metal contamination risks and simplifying purification.
Q: What purity levels can be achieved with this synthetic route?
A: The patent data demonstrates that through optimized recrystallization protocols, specifically using n-heptane, the final alpha,alpha-terpyridine product can achieve HPLC purity levels exceeding 99.4% without the need for complex column chromatography.
Q: Is this process suitable for industrial scale-up?
A: Yes, the methodology is designed for large-scale production, utilizing common solvents like THF and toluene and avoiding hazardous reagents. The total yield reaches over 68%, significantly higher than previous base-mediated attempts which yielded only 15%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha Alpha Terpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance ligands like alpha, alpha-terpyridine play in the development of next-generation therapeutics and functional materials. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, consistently delivering material that meets the exacting standards of the global pharmaceutical and electronic industries.
We invite you to leverage our technical expertise to optimize your supply chain and accelerate your time to market. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our implementation of this patented technology can drive value and reliability for your organization.
