Advanced Catalyst-Free Synthesis of Quaternary Carbon Center Containing Symmetric Gem-Difluoroolefin Derivatives
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex molecular architectures, particularly those featuring all-carbon quaternary centers which are prevalent in bioactive molecules. Patent CN111205202A introduces a groundbreaking approach to synthesizing symmetric gem-difluoroolefin derivatives containing these challenging quaternary carbon centers. This technology addresses a significant gap in organic synthesis, as prior art has rarely reported efficient methods for creating symmetric gem-difluoroolefins with all-carbon quaternary substitution. The innovation lies in its ability to utilize readily available starting materials to generate high-value building blocks that serve as effective isosteres for carbonyl groups, thereby enhancing the metabolic stability and biological activity of potential drug candidates. By leveraging a catalyst-free, base-mediated pathway, this process offers a streamlined route that bypasses the complexities often associated with transition metal catalysis, presenting a compelling opportunity for reliable pharmaceutical intermediate supplier partnerships focused on next-generation medicinal chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of all-carbon quaternary centers has been a formidable challenge in organic synthesis, often requiring harsh reaction conditions, stoichiometric amounts of expensive reagents, or sophisticated transition metal catalysts that necessitate rigorous removal steps to meet regulatory standards for residual metals in APIs. Conventional routes to gem-difluoroolefins frequently suffer from limited substrate scope, poor functional group tolerance, and the generation of significant hazardous waste, which complicates the environmental compliance profile of the manufacturing process. Furthermore, many existing methods struggle to achieve symmetry in the final product without multiple protection and deprotection steps, leading to lower overall yields and increased production costs. The reliance on precious metal catalysts not only inflates the raw material expenditure but also introduces supply chain vulnerabilities related to the availability and price volatility of these metals, making the economic viability of large-scale production uncertain for many process development teams.
The Novel Approach
In stark contrast to these traditional limitations, the method disclosed in the patent utilizes a direct coupling strategy between active methylene compounds and α-trifluoromethyl styrene derivatives under mild basic conditions. This novel approach eliminates the need for any catalyst or ligand, relying instead on simple bases such as cesium carbonate or alkali metal tert-butoxides to drive the reaction forward efficiently. The process operates at moderate temperatures ranging from 60°C to 100°C, ensuring energy efficiency and operational safety while maintaining high conversion rates. As illustrated in the reaction scheme below, this methodology allows for the direct formation of the symmetric gem-difluoroolefin scaffold with a quaternary center in a single pot, drastically simplifying the synthetic workflow.

The versatility of this reaction is further evidenced by its compatibility with a broad array of substituents on both the methylene and styrene components, enabling the rapid generation of diverse chemical libraries for drug discovery programs without the need for extensive process optimization for each new analog.
Mechanistic Insights into Base-Mediated Difluoroolefination
The core of this synthetic breakthrough involves the nucleophilic activation of the active methylene compound by the base to form a carbanion species, which subsequently attacks the electron-deficient double bond of the α-trifluoromethyl styrene. This addition-elimination sequence is facilitated by the strong electron-withdrawing nature of the trifluoromethyl group, which activates the olefin towards nucleophilic attack while simultaneously stabilizing the intermediate anionic species. The subsequent elimination of a fluoride ion restores the double bond character, resulting in the formation of the gem-difluoroolefin moiety. This mechanism is particularly elegant because it avoids the formation of unstable intermediates that often plague other fluorination strategies, ensuring a clean reaction profile with minimal byproduct formation. The absence of transition metals means there are no competing oxidative addition or reductive elimination pathways that could lead to complex mixtures of regioisomers or stereoisomers, thereby simplifying the downstream purification process significantly.
From an impurity control perspective, the mild reaction conditions play a crucial role in maintaining the integrity of sensitive functional groups present on the aromatic rings of the substrates. The use of polar aprotic solvents like dimethyl sulfoxide (DMSO) or N,N-dimethylformamide (DMF) ensures excellent solubility of the ionic intermediates, promoting homogeneous reaction kinetics that prevent localized hot spots which could trigger decomposition. The general structure of the resulting products, as shown in the formula below, highlights the symmetrical nature of the molecule where the quaternary carbon serves as a central hub connecting two identical gem-difluoroolefin arms, a structural feature that is difficult to achieve with high fidelity using asymmetric synthesis methods.
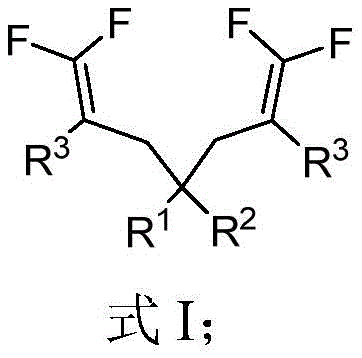
This structural symmetry not only simplifies the spectroscopic characterization of the final product but also suggests potential advantages in crystal packing and solid-state properties, which are critical factors for the formulation of pharmaceutical ingredients. The robustness of this mechanism across various electronic environments on the aromatic rings confirms its utility as a general platform technology for constructing complex fluorinated scaffolds.
How to Synthesize Symmetric Gem-Difluoroolefin Derivatives Efficiently
To implement this synthesis effectively, one must carefully control the stoichiometry of the base relative to the α-trifluoromethyl styrene, with a molar ratio typically ranging from 3:1 to 3:4 to ensure complete deprotonation and driving force for the elimination step. The reaction is conducted under an inert atmosphere, such as nitrogen or argon, to prevent moisture interference which could deactivate the base or hydrolyze sensitive intermediates. Following the reaction period of 3 to 24 hours, the workup procedure involves a straightforward aqueous quench followed by extraction with common organic solvents like ethyl acetate, allowing for the easy separation of the organic product from inorganic salts. The detailed standardized synthesis steps see the guide below for precise operational parameters.
- Dissolve active methylene compound, alpha-trifluoromethyl styrene compound, and a base such as cesium carbonate or lithium tert-butoxide in an organic solvent like DMF or DMSO under a protective nitrogen atmosphere.
- Stir the reaction mixture at a temperature between 60°C and 100°C for a duration of 3 to 24 hours to ensure complete conversion to the target derivative.
- Quench the reaction with water, extract the product using ethyl acetate or dichloromethane, remove the solvent via rotary evaporation, and purify the crude product using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalyst-free synthesis route presents a transformative opportunity to optimize the cost structure and reliability of their raw material supply chains. By removing the dependency on scarce and expensive transition metal catalysts, the direct material costs associated with the production of these high-value intermediates are substantially reduced, freeing up capital for other critical R&D investments. Furthermore, the use of commodity chemicals such as benzyl cyanides and substituted styrenes as starting materials ensures a stable and diversified supply base, mitigating the risks associated with single-source suppliers of specialized reagents. The simplicity of the reaction setup, which does not require specialized high-pressure equipment or cryogenic cooling, allows for manufacturing in standard glass-lined or stainless steel reactors, facilitating easier technology transfer between different production sites globally.
- Cost Reduction in Manufacturing: The elimination of costly ligands and noble metal catalysts removes a significant portion of the variable cost per kilogram, while the high atom economy of the reaction minimizes waste disposal fees. Additionally, the mild reaction temperatures reduce energy consumption compared to processes requiring extreme heating or cooling, contributing to a lower overall carbon footprint and operational expenditure. The simplified purification process, often achievable through standard column chromatography or crystallization, reduces the consumption of silica gel and solvents, further driving down the cost of goods sold without compromising on the quality of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Since the key starting materials are bulk chemicals produced by numerous global manufacturers, the risk of supply disruption due to geopolitical issues or factory outages is significantly minimized. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in raw material quality, allowing for greater flexibility in vendor selection and qualification. This resilience ensures consistent delivery schedules for downstream customers, enabling them to maintain lean inventory levels and respond more agilely to market demands for new drug formulations containing these fluorinated motifs.
- Scalability and Environmental Compliance: The absence of heavy metals simplifies the regulatory approval process for new drug applications, as there is no need for extensive validation of metal clearance steps, accelerating the time to market for new therapies. The process generates primarily inorganic salt waste and spent solvent, which are easier to treat and recycle compared to the complex hazardous waste streams generated by transition metal-catalyzed reactions. This aligns with modern green chemistry principles and corporate sustainability goals, making the manufacturing process more attractive to environmentally conscious stakeholders and regulatory bodies worldwide.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this method for the production of complex fluorinated intermediates.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented method described in CN111205202A operates without the need for any transition metal catalysts or ligands, utilizing simple inorganic or organic bases instead, which significantly reduces raw material costs and eliminates heavy metal contamination risks.
Q: What is the substrate scope for the active methylene component?
A: The method demonstrates excellent functional group tolerance, accommodating a wide range of active methylene compounds including various substituted benzyl cyanides, phenylacetates, and heterocyclic derivatives, allowing for diverse structural modifications of the final pharmaceutical intermediate.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the reaction conditions are mild (60-100°C) and the operation is simple and safe, involving standard extraction and chromatography techniques, making it highly adaptable for commercial scale-up from kilogram to ton-level production without complex engineering requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Symmetric Gem-Difluoroolefin Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality, structurally complex intermediates to accelerate your drug discovery pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify the identity and quality of every batch. Our expertise in fluorine chemistry allows us to navigate the unique challenges of handling fluorinated reagents safely and effectively, guaranteeing a supply of high-purity pharmaceutical intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to contact our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this catalyst-free method for your specific target molecules. We encourage you to reach out for specific COA data and route feasibility assessments to validate the performance of these symmetric gem-difluoroolefin derivatives in your own biological assays, ensuring a successful partnership that drives value for your organization.
