Scalable Production of (R)-1-Boc-3-aminopiperidine: A Breakthrough in Diabetes Drug Intermediates
The pharmaceutical landscape for type 2 diabetes treatment continues to evolve, driven by the demand for safer and more efficient synthesis routes for key active pharmaceutical ingredients (APIs). Patent CN115557882A introduces a robust preparation method for (R)-1-Boc-3-aminopiperidine, a critical chiral intermediate used in the manufacture of DPP-IV inhibitors such as alogliptin benzoate and trexagliptin. This technology addresses long-standing challenges in the industry by replacing hazardous reagents and complex high-pressure operations with a streamlined, chiral resolution-based approach. For global procurement teams and R&D directors, this patent represents a significant shift towards greener chemistry that does not compromise on yield or purity. By leveraging R-mandelic acid for stereochemical control and utilizing a classic Hofmann degradation for the final functional group transformation, the process offers a compelling alternative to legacy synthetic pathways that have historically plagued supply chains with safety concerns and inconsistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of piperidine-based amine intermediates has been fraught with significant technical and safety hurdles that impede efficient commercial scale-up. Early literature, such as the work by Brian R. de Costa, relied heavily on the use of azide compounds, which are notoriously unstable and pose severe explosion risks, particularly when handled in large quantities required for industrial production. Furthermore, other established routes, like those reported in Heterocycles, necessitated high-pressure hydrogenation steps to reduce pyridine derivatives. These high-pressure conditions not only require expensive, specialized reactor equipment but often result in incomplete hydrogenation and the formation of difficult-to-remove by-products. The cumulative effect of these limitations is a manufacturing process that is capital intensive, operationally hazardous, and prone to batch-to-batch variability, ultimately driving up the cost of goods and extending lead times for downstream API manufacturers.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a温和 (mild) and highly controllable four-step sequence starting from readily available ethyl 3-piperidinecarboxylate. Instead of dangerous azides or high-pressure reduction, the process employs a classical chiral resolution strategy using R-mandelic acid to establish the necessary stereocenter with exceptional precision. This is followed by a standard Boc protection, an aminolysis to form an amide, and finally a Hofmann degradation to generate the primary amine. This strategic pivot away from extreme reaction conditions allows the synthesis to be conducted in standard glass-lined or stainless steel reactors without the need for specialized high-pressure infrastructure. The result is a process that is inherently safer, easier to purify, and significantly more robust, providing a reliable foundation for the consistent supply of high-purity pharmaceutical intermediates.
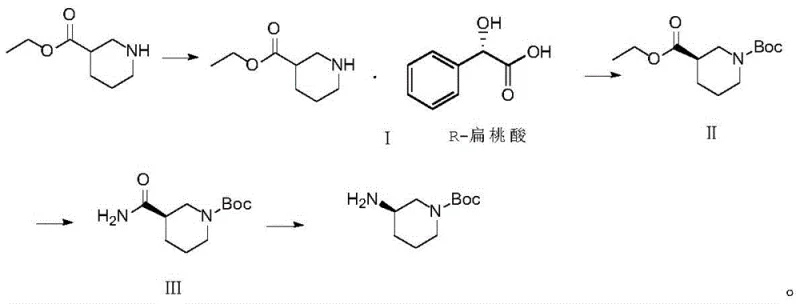
Mechanistic Insights into Chiral Resolution and Hofmann Degradation
The cornerstone of this synthesis lies in the initial chiral resolution step, which effectively separates the racemic ethyl 3-piperidinecarboxylate into its desired enantiomer. By reacting the racemic ester with R-mandelic acid in ethyl acetate at controlled temperatures between 40°C and 50°C, a diastereomeric salt is formed. The differential solubility of these salts allows for the selective crystallization of the target (R)-enantiomer salt. Subsequent recrystallization from ethanol further enriches the optical purity, consistently achieving an enantiomeric excess (ee) value greater than 99%. This high level of stereochemical control at the very beginning of the synthesis is crucial, as it prevents the propagation of chiral impurities through subsequent steps, thereby simplifying the purification burden for the final API and ensuring compliance with stringent regulatory standards for chiral drugs.
The final transformation involves the Hofmann degradation, a powerful tool for converting primary amides into primary amines with one fewer carbon atom. In this specific application, the intermediate (R)-1-Boc-3-piperidinecarboxamide is treated with sodium hypochlorite in a strongly alkaline environment provided by sodium hydroxide. The mechanism proceeds through the formation of an N-chloroamide, which rearranges to an isocyanate intermediate before hydrolyzing to the final amine. The patent specifies precise temperature controls, maintaining the reaction between 0°C and 5°C during the addition of the oxidant to prevent side reactions, followed by warming to 40-45°C to drive the rearrangement to completion. This careful thermal management ensures high conversion rates while minimizing the formation of urea by-products or over-oxidation, resulting in a clean reaction profile that facilitates straightforward isolation of the final product.
How to Synthesize (R)-1-Boc-3-aminopiperidine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing reproducibility and safety at every stage. The process begins with the dissolution of the starting material in ethyl acetate, followed by the slow addition of the resolving agent to ensure uniform crystal growth. Subsequent steps involve standard organic workups, including aqueous washes and drying over anhydrous sodium sulfate, which are familiar to any process chemist. The detailed experimental conditions, such as the specific molar ratios of reagents and the precise temperature ramps for crystallization, are designed to maximize yield and purity without requiring exotic catalysts or conditions. For a comprehensive understanding of the operational parameters, the detailed standardized synthesis steps are provided in the guide below.
- Perform chiral resolution of ethyl 3-piperidinecarboxylate using R-mandelic acid in ethyl acetate to obtain the chiral salt with >99% ee.
- Conduct condensation reaction with Boc anhydride in dichloromethane using triethylamine as a base to protect the nitrogen atom.
- Execute aminolysis with ammonia water in THF to convert the ester group into a primary amide intermediate.
- Complete the synthesis via Hofmann degradation using sodium hypochlorite and sodium hydroxide to yield the final amine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial benefits that directly address the pain points of procurement managers and supply chain directors. By eliminating the need for hazardous azide reagents and high-pressure hydrogenation equipment, the process significantly reduces the capital expenditure required for facility setup and the operational costs associated with safety compliance and waste disposal. The reliance on commodity chemicals like R-mandelic acid, Boc anhydride, and ammonia water ensures a stable and diverse supply base, mitigating the risk of raw material shortages that can disrupt production schedules. Furthermore, the mild reaction conditions and high yields contribute to a more predictable manufacturing timeline, allowing for better inventory planning and faster response to market demands for diabetes medications.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the avoidance of high-pressure processing equipment lead to a drastic simplification of the production infrastructure. Without the need for specialized hydrogenation reactors or extensive safety measures for explosive azides, the overhead costs per kilogram of product are significantly lowered. Additionally, the high yields reported in the patent, particularly the quantitative conversion in the aminolysis step, mean that less raw material is wasted, further driving down the variable costs associated with large-scale manufacturing.
- Enhanced Supply Chain Reliability: The use of widely available starting materials and reagents ensures that the supply chain is resilient against geopolitical or logistical disruptions. Unlike processes that rely on bespoke or scarce catalysts, this method utilizes chemicals that are produced in massive volumes globally, guaranteeing consistent availability. The robustness of the process also means that production can be easily transferred between different manufacturing sites without significant re-validation, providing flexibility in sourcing and reducing the risk of single-point failures in the supply network.
- Scalability and Environmental Compliance: The process is inherently green, generating fewer hazardous by-products and avoiding the use of toxic heavy metals. This aligns perfectly with modern environmental regulations and corporate sustainability goals, reducing the burden of wastewater treatment and hazardous waste disposal. The simplicity of the workup procedures, involving standard extractions and crystallizations, makes the process highly scalable from kilogram to multi-ton quantities without the engineering complexities often associated with scaling up high-pressure or cryogenic reactions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity on safety, purity, and scalability for potential partners and stakeholders evaluating this technology for their supply chains.
Q: Why is this new synthesis method safer than previous azide-based routes?
A: Previous methods utilized hazardous azide compounds which pose significant explosion risks during industrial handling. This patented process eliminates azides entirely, relying instead on stable chiral resolution and standard organic transformations, drastically improving operational safety.
Q: How does the chiral resolution step ensure high optical purity?
A: The process utilizes R-mandelic acid as a resolving agent in ethyl acetate. Through controlled crystallization and recrystallization cycles, the method consistently achieves an enantiomeric excess (ee) value greater than 99%, ensuring the strict stereochemical requirements for downstream API synthesis are met.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the method operates under mild reaction conditions without the need for high-pressure hydrogenation equipment. The use of common solvents and reagents, combined with high yields and simplified post-treatment procedures, makes it highly adaptable for ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-1-Boc-3-aminopiperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and production of life-saving medications. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis or large-scale supply of this key diabetes drug intermediate, our infrastructure is designed to support your most demanding projects with reliability and precision.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this safer, more efficient method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific volume requirements, ensuring that your project moves forward with the highest level of technical and commercial confidence.
