Advanced Manufacturing of VEGFR2 Inhibitor SU1498: A Scalable and Green Synthetic Route
Advanced Manufacturing of VEGFR2 Inhibitor SU1498: A Scalable and Green Synthetic Route
The pharmaceutical industry's relentless pursuit of potent anti-angiogenic agents has placed Vascular Endothelial Growth Factor Receptor 2 (VEGFR2) inhibitors at the forefront of oncology research. Among these, SU1498 (2-cyano-3-(4-hydroxy-3,5-diisopropylphenyl)-N-(3-phenylpropyl)acrylamide) stands out as a critical selective inhibitor with significant therapeutic potential. However, the commercial viability of such complex molecules often hinges on the efficiency and safety of their synthesis. The recent disclosure in patent CN110862335B introduces a transformative preparation method that addresses longstanding bottlenecks in SU1498 manufacturing. By replacing hazardous reagents with greener alternatives and optimizing reaction conditions, this technology offers a pathway to high-purity intermediates and final active pharmaceutical ingredients (APIs). For global procurement teams and R&D directors, understanding this shift is vital for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality at scale.
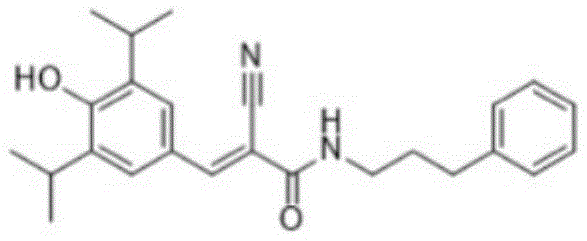
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of SU1498 has been plagued by significant operational and environmental challenges, primarily stemming from the reliance on outdated formylation techniques. As documented in prior art such as US5792771A, the conventional route necessitates the use of urotropine (hexamethylenetetramine) in trifluoroacetic acid to introduce the crucial aldehyde functionality onto the phenolic ring. This approach is inherently flawed for modern industrial applications due to the highly toxic and corrosive nature of the reagents involved. The use of urotropine generates substantial amounts of difficult-to-treat waste streams, posing severe environmental compliance risks and escalating disposal costs. Furthermore, the reaction conditions are often harsh, leading to side reactions that complicate downstream purification. The resulting low yields and impure intermediates create a ripple effect, diminishing the overall efficiency of the synthesis and making the final cost of goods sold (COGS) prohibitively high for large-scale commercialization.
The Novel Approach
In stark contrast, the methodology outlined in CN110862335B represents a paradigm shift towards sustainable and efficient chemical manufacturing. This novel approach abandons the problematic urotropine route in favor of a classic yet highly optimized Vilsmeier-Haack formylation using phosphorus oxychloride (POCl3) and N,N-dimethylformamide (DMF). This substitution not only utilizes readily available and cost-effective raw materials but also significantly streamlines the post-reaction workup. The process is further distinguished by its innovative second step, which employs task-specific ionic liquids as both solvent and catalyst for the Knoevenagel condensation. This eliminates the need for large volumes of volatile organic compounds (VOCs), thereby reducing the facility's environmental footprint. The culmination of these improvements is a robust three-step sequence that delivers the target molecule with superior purity and yield, establishing a new benchmark for cost reduction in pharmaceutical intermediates manufacturing.
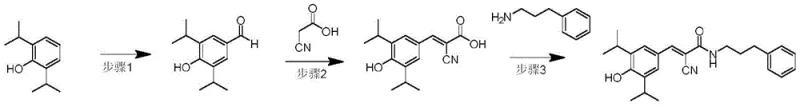
Mechanistic Insights into Ionic Liquid Mediated Condensation and Amidation
The core innovation of this synthetic strategy lies in the meticulous optimization of each transformation, particularly the condensation step. The initial Vilsmeier-Haack reaction proceeds through the formation of an iminium salt intermediate from DMF and POCl3, which acts as a potent electrophile attacking the electron-rich aromatic ring of 2,6-diisopropylphenol. The steric hindrance provided by the two isopropyl groups directs the formylation exclusively to the para-position, ensuring high regioselectivity. Following hydrolysis, the resulting 4-hydroxy-3,5-diisopropylbenzaldehyde is obtained in high yield (approximately 90%), providing a clean substrate for subsequent steps. This precision in the early stages is critical for minimizing impurity carryover, a key concern for R&D directors focused on impurity profiles.
The subsequent Knoevenagel condensation between the aldehyde and 2-cyanoacetic acid is where the process truly differentiates itself through green chemistry principles. By utilizing ionic liquids such as tris(2-hydroxyethyl)ammonium acetate, the reaction benefits from a unique microenvironment that stabilizes the transition state and facilitates proton transfer without the need for additional toxic bases or solvents. The ionic liquid acts as a dual-function medium, dissolving the reactants effectively while catalyzing the dehydration step required to form the alpha,beta-unsaturated nitrile. This results in the formation of (E)-2-cyano-3-(4-hydroxy-3,5-diisopropylphenyl)acrylic acid with excellent stereocontrol and purity. Finally, the amidation step employs standard peptide coupling reagents like EDCI and HOBt, ensuring mild conditions that preserve the sensitive cyano and phenolic functionalities while efficiently linking the 3-phenylpropylamine side chain.
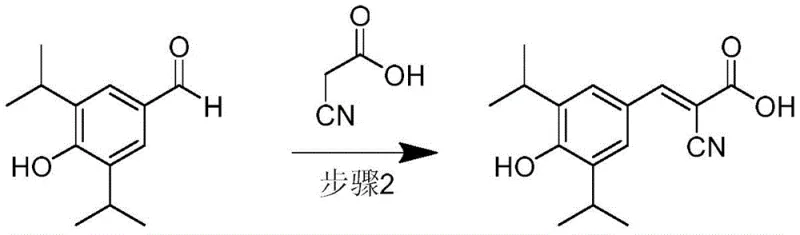
How to Synthesize SU1498 Efficiently
Implementing this advanced synthetic route requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process is designed to be modular, allowing for distinct quality control checkpoints after each of the three major transformations. Operators must carefully control the temperature during the exothermic Vilsmeier reagent formation and maintain precise stoichiometric ratios during the ionic liquid condensation to maximize conversion. The following guide outlines the critical operational phases derived from the patent examples, serving as a foundational reference for process engineers aiming to replicate this high-efficiency workflow in a GMP-compliant setting.
- Perform Vilsmeier-Haack formylation on 2,6-diisopropylphenol using POCl3 and DMF in 1,2-dichloroethane at 50-100°C to yield 4-hydroxy-3,5-diisopropylbenzaldehyde.
- Conduct Knoevenagel condensation between the aldehyde intermediate and 2-cyanoacetic acid using tris(2-hydroxyethyl)ammonium acetate ionic liquid at 60-100°C.
- Execute amide coupling between the resulting acrylic acid derivative and 3-phenylpropylamine using EDCI and HOBt in DMF at room temperature to finalize SU1498.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere technical superiority. The elimination of hazardous reagents like urotropine directly translates to a simplified regulatory compliance landscape, reducing the administrative burden and liability associated with handling toxic substances. Moreover, the use of inexpensive, commodity-grade starting materials such as 2,6-diisopropylphenol and 3-phenylpropylamine ensures a stable supply chain that is less susceptible to market volatility compared to specialized, high-cost reagents. The simplified post-processing, particularly the ease of product isolation via filtration in the ionic liquid step, drastically reduces solvent consumption and energy usage during concentration and drying phases.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of expensive and difficult-to-dispose-of reagents. By substituting the urotropine/TFA system with POCl3/DMF and ionic liquids, the facility avoids the high costs associated with neutralizing corrosive acidic waste and treating amine-containing effluents. Additionally, the high yields reported in each step (ranging from 80% to 90%) minimize raw material waste, ensuring that a greater proportion of input mass is converted into saleable product. This efficiency gain allows for a more competitive pricing structure without compromising on the stringent quality standards required for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals mitigates the risk of supply disruptions that often plague specialty reagent markets. Since the raw materials are common industrial chemicals, sourcing can be diversified across multiple vendors, preventing single-source bottlenecks. Furthermore, the robustness of the reaction conditions—operating at moderate temperatures and atmospheric pressure—means the process is less prone to batch failures due to minor equipment fluctuations. This reliability ensures consistent delivery schedules, a critical factor for partners managing just-in-time inventory for clinical or commercial production runs.
- Scalability and Environmental Compliance: From a scalability perspective, the absence of cryogenic conditions or high-pressure reactors makes this route ideal for rapid scale-up from kilogram to multi-ton production. The green chemistry attributes, specifically the use of recyclable ionic liquids and reduced VOC emissions, align perfectly with increasingly stringent global environmental regulations. This forward-looking approach future-proofs the manufacturing asset against tightening emission caps and waste disposal restrictions, ensuring long-term operational continuity and enhancing the corporate sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this SU1498 synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this method. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this intermediate into their broader drug development pipelines.
Q: Why is the new synthetic route for SU1498 preferred over the traditional Urotropine method?
A: The traditional method utilizes toxic and corrosive urotropine in trifluoroacetic acid, leading to difficult post-processing, environmental pollution, and lower yields. The novel route replaces this with a standard Vilsmeier-Haack reaction using POCl3/DMF, which is safer, easier to handle, and provides higher purity intermediates suitable for large-scale production.
Q: What is the specific advantage of using ionic liquids in Step 2 of the synthesis?
A: Using ionic liquids like tris(2-hydroxyethyl)ammonium acetate serves as both a solvent and a catalyst for the Knoevenagel condensation. This eliminates the need for volatile organic solvents, simplifies the work-up process through simple acidification and filtration, and aligns with green chemistry principles by reducing hazardous waste generation.
Q: What purity levels can be achieved with this optimized manufacturing process?
A: The patented process demonstrates exceptional control over impurities, achieving final product purity levels of approximately 99% after standard column chromatography purification. The intermediate steps also show high purity (95-97%), ensuring a robust quality profile for the final VEGFR2 inhibitor.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable SU1498 Supplier
At NINGBO INNO PHARMCHEM, we recognize that the complexity of synthesizing VEGFR2 inhibitors like SU1498 demands a partner with deep technical expertise and unwavering commitment to quality. Our team has extensively analyzed the pathways described in CN110862335B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that achieving the reported 99% purity requires not just the right recipe, but rigorous process control and state-of-the-art analytical capabilities. Our facilities are equipped with rigorous QC labs and advanced purification technologies to ensure that every batch meets the stringent purity specifications demanded by top-tier pharmaceutical clients.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your next project. Whether you require custom synthesis for preclinical studies or bulk supply for commercial formulation, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized process can enhance your supply chain efficiency and reduce your overall time to market.
