Scalable Gold-Catalyzed Halogenation for High-Purity Pharmaceutical Intermediates
Scalable Gold-Catalyzed Halogenation for High-Purity Pharmaceutical Intermediates
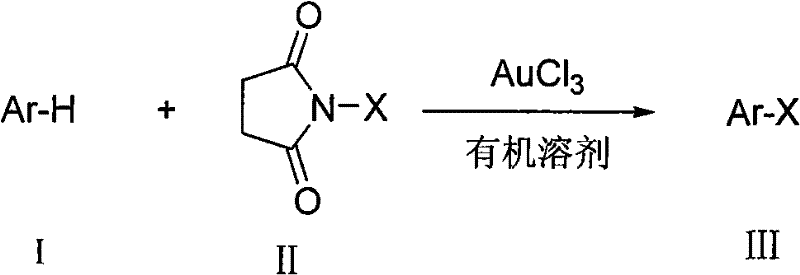
The synthesis of aromatic halogenated compounds represents a cornerstone operation in the manufacturing of advanced pharmaceutical intermediates and fine chemicals. Patent CN101993318A introduces a transformative methodology that leverages gold trichloride (AuCl3) as a highly efficient catalyst to facilitate the direct halogenation of aromatic hydrocarbons. Unlike traditional methods that often rely on harsh Lewis acids or hazardous elemental halogens, this innovation utilizes N-halosuccinimides as mild halogen sources under remarkably benign conditions. The technology enables the production of chlorinated, brominated, and iodinated aromatics with exceptional regioselectivity and yield. For R&D directors and process chemists, this patent offers a robust pathway to complex molecular building blocks essential for cross-coupling reactions such as Suzuki-Miyaura or Buchwald-Hartwig aminations. The ability to conduct these reactions in air without stringent moisture exclusion marks a significant departure from conventional protocols, promising enhanced operational simplicity and safety profiles for industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of aromatic halides has been fraught with significant operational challenges and safety hazards that complicate large-scale manufacturing. Traditional electrophilic aromatic substitution often necessitates the use of stoichiometric or even excess amounts of strong Lewis acids like aluminum chloride or iron(III) chloride, which generate substantial quantities of acidic waste streams requiring costly neutralization and disposal. Furthermore, many classical protocols employ elemental chlorine or liquid bromine as halogenating agents, substances that are highly corrosive, toxic, and difficult to handle safely in a production environment. These reactions frequently demand strictly anhydrous and oxygen-free conditions, requiring specialized equipment such as gloveboxes or extensive nitrogen purging systems, which increases capital expenditure and operational complexity. Additionally, the harsh reaction conditions associated with these legacy methods often lead to poor functional group tolerance, resulting in side reactions, over-halogenation, or decomposition of sensitive moieties within the substrate molecule.
The Novel Approach
The methodology disclosed in CN101993318A fundamentally redefines the halogenation landscape by introducing a catalytic system based on gold trichloride that operates under mild, aerobic conditions. By utilizing N-halosuccinimides (NCS, NBS, NIS) as the halogen source, the process eliminates the need for handling dangerous elemental halogens, thereby enhancing workplace safety and reducing regulatory burdens associated with hazardous material storage. The reaction proceeds efficiently in common organic solvents such as 1,2-dichloroethane or dichloromethane at moderate temperatures ranging from 0°C to 80°C. Crucially, the system demonstrates remarkable tolerance to atmospheric moisture and oxygen, allowing reactions to be performed in standard glassware without the need for inert gas protection. This simplification of reaction conditions not only lowers the barrier to entry for laboratory synthesis but also translates directly into reduced engineering controls and lower operating costs when scaling up to commercial production volumes.
Mechanistic Insights into AuCl3-Catalyzed Electrophilic Aromatic Substitution
The core of this technological advancement lies in the unique ability of the gold(III) center to activate the N-halo bond of the succinimide reagent, generating a highly reactive electrophilic halogen species in situ. Mechanistically, the gold trichloride catalyst coordinates with the nitrogen or carbonyl oxygen of the N-halosuccinimide, polarizing the N-X bond and facilitating the transfer of the halogen cation equivalent to the electron-rich aromatic ring. This activation mode allows for the generation of the active halogenating agent at much lower concentrations than would be required in uncatalyzed thermal processes, thereby minimizing non-selective radical pathways that often lead to poly-halogenated byproducts. The electrophilic attack follows standard aromatic substitution rules, where the incoming halogen is directed to positions ortho or para to electron-donating groups, ensuring high regioselectivity that is critical for the synthesis of specific pharmaceutical intermediates. The catalytic cycle is sustained by the regeneration of the active gold species, allowing for turnover numbers that far exceed those of stoichiometric Lewis acid promoters.
From an impurity control perspective, the mild nature of this catalytic system offers distinct advantages in maintaining product purity profiles required for GMP manufacturing. Because the reaction avoids the extreme acidity and oxidative stress associated with traditional methods, there is a significant reduction in the formation of tar-like polymeric byproducts and structural isomers that are difficult to separate. The low catalyst loading, typically between 0.01% and 1%, ensures that residual heavy metal content in the final product is minimized, simplifying downstream purification steps such as crystallization or chromatography. Furthermore, the compatibility with diverse functional groups—including esters, ethers, amides, and existing halogens—means that complex substrates can be halogenated late-stage in a synthetic sequence without requiring extensive protecting group strategies. This chemoselectivity is paramount for R&D teams aiming to streamline synthetic routes and improve overall process mass intensity.
How to Synthesize Aromatic Halogenated Compounds Efficiently
The practical implementation of this gold-catalyzed halogenation protocol is designed for ease of execution while maintaining high standards of reproducibility and yield. The general procedure involves dissolving the aromatic substrate and the appropriate N-halosuccinimide reagent in a selected organic solvent, followed by the addition of the gold trichloride catalyst solution. The mixture is then heated to the specified temperature, typically between room temperature and 80°C, and monitored via GC-MS or TLC until the starting material is fully consumed. Following reaction completion, the workup is straightforward, involving simple concentration of the solvent and purification via column chromatography or recrystallization to afford the target aryl halide in high purity. Detailed standardized synthesis steps for specific substrates are provided in the guide below to ensure consistent results across different batches.
- Combine aromatic hydrocarbon substrate and N-halosuccinimide in an organic solvent such as 1,2-dichloroethane.
- Add a catalytic amount of gold trichloride (0.01-1 mol%) to the reaction mixture under ambient air conditions.
- Heat the mixture between 0°C and 80°C until completion, then purify via concentration and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this gold-catalyzed halogenation technology presents a compelling value proposition centered on cost efficiency and operational resilience. The shift from stoichiometric Lewis acids to a catalytic gold system dramatically reduces the consumption of raw materials, particularly the expensive and hazardous reagents traditionally used in halogenation. By eliminating the need for specialized inert atmosphere equipment and rigorous drying protocols, facilities can utilize existing infrastructure more effectively, reducing both capital investment and maintenance overheads. The simplified workflow also translates to shorter batch cycles and higher throughput, enabling manufacturers to respond more agilely to market demands for key pharmaceutical intermediates. Furthermore, the use of solid N-halosuccinimides instead of liquid halogens simplifies logistics and storage, mitigating risks associated with the transport of corrosive chemicals.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic reduction in catalyst loading and the elimination of expensive waste treatment protocols. Since the gold catalyst is used in trace amounts (as low as 0.01 mol%), the cost contribution of the precious metal to the final product is negligible, especially when considering the high turnover efficiency. Additionally, the avoidance of stoichiometric Lewis acids removes the burden of neutralizing large volumes of acidic waste, leading to substantial savings in environmental compliance and disposal fees. The mild reaction conditions also imply lower energy consumption for heating and cooling compared to processes requiring cryogenic temperatures or prolonged reflux, further optimizing the utility costs associated with production.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly bolstered by the use of commercially available and stable reagents such as N-bromosuccinimide and gold trichloride, which are readily sourced from multiple global suppliers. The robustness of the reaction against atmospheric moisture and oxygen means that production schedules are less vulnerable to disruptions caused by equipment failure in inert gas systems or variations in solvent quality. This reliability ensures consistent delivery timelines for critical intermediates, reducing the risk of stockouts that could halt downstream API synthesis. Moreover, the broad substrate scope allows for the flexible production of various halogenated building blocks using a unified platform technology, simplifying inventory management and reducing the need for specialized reactor setups for different products.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the inherent safety and simplicity of the reaction conditions. The absence of hazardous elemental halogens and the operation under ambient pressure make the process inherently safer for large-scale reactors, reducing the need for exotic metallurgy or complex safety interlocks. From an environmental standpoint, the atom economy is improved by the use of N-halosuccinimides, which produce succinimide as a benign byproduct that can potentially be recycled or easily disposed of. The reduction in heavy metal waste and acidic effluents aligns with increasingly stringent global environmental regulations, positioning manufacturers who adopt this technology as leaders in sustainable chemical production and green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this halogenation technology. These answers are derived directly from the experimental data and claims presented in the patent literature to provide accurate guidance for potential adopters. Understanding these nuances is essential for evaluating the feasibility of integrating this method into existing manufacturing workflows.
Q: Does this gold-catalyzed halogenation require inert atmosphere conditions?
A: No, a key advantage of the method described in CN101993318A is that it proceeds smoothly in air without the need for strict anhydrous or oxygen-free conditions, significantly simplifying operational requirements.
Q: What is the typical catalyst loading for this transformation?
A: The process utilizes extremely low catalyst loadings, typically ranging from 0.01% to 1% molar ratio relative to the aromatic substrate, which drastically reduces heavy metal contamination risks and costs.
Q: Is this method compatible with sensitive functional groups?
A: Yes, the reaction exhibits excellent functional group tolerance, successfully accommodating substrates with alkyl, alkoxy, ester, amide, and existing halogen substituents without degradation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Halogenated Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality aromatic halides play in the development of next-generation therapeutics and advanced materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical techniques to verify identity and assay. By leveraging advanced catalytic technologies like the gold-mediated halogenation described in CN101993318A, we can offer cost-effective solutions without compromising on the quality required for GMP environments.
We invite you to collaborate with us to optimize your supply chain for aromatic intermediates. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how our process improvements can impact your bottom line. Please contact our technical procurement team today to request specific COA data for our catalog items or to discuss route feasibility assessments for your custom synthesis projects. Let us be your partner in driving innovation and efficiency in your chemical supply chain.
