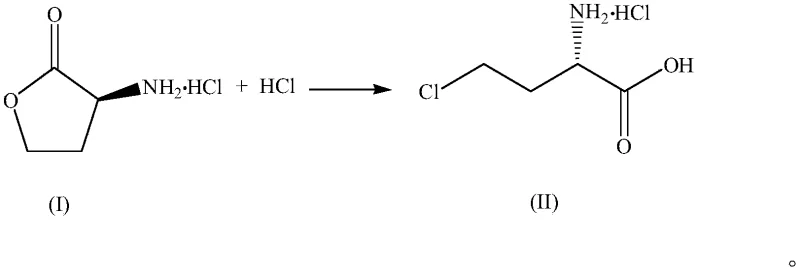
Scalable Synthesis of (S)-4-Chloro-2-aminobutyric Acid Derivatives via Efficient HCl Chlorination
The global demand for high-efficiency, low-toxicity herbicides continues to drive innovation in the synthesis of key chiral intermediates, particularly for the production of L-glufosinate-ammonium. As detailed in the recent patent publication CN116171270A, a significant technological breakthrough has been achieved in the preparation of (S)-4-chloro-2-aminobutyric acid hydrochloride and its corresponding esters. This patent introduces a novel ring-opening chlorination strategy that fundamentally shifts the paradigm from traditional thionyl chloride-based methods to a more sustainable and cost-effective hydrogen chloride (HCl) mediated process. For R&D directors and procurement specialists in the agrochemical and pharmaceutical sectors, this development represents a critical opportunity to optimize supply chains for high-purity API intermediates. The methodology not only addresses the environmental concerns associated with sulfur-containing waste but also enhances the overall economic viability of producing these essential building blocks for asymmetric synthesis.
Furthermore, the versatility of this synthetic route allows for the seamless transition from the chloro-acid intermediate to various ester derivatives, which are pivotal in the downstream synthesis of biologically active compounds. By leveraging concentrated hydrochloric acid as both the solvent and the chlorinating agent, the process minimizes the number of unit operations required, thereby reducing the potential for yield loss during isolation steps. This technical advancement aligns perfectly with the industry's push towards greener chemistry principles without compromising on the stringent purity specifications required for commercial-scale manufacturing of complex chiral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (S)-4-chloro-2-aminobutyrate has relied heavily on the use of thionyl chloride (SOCl2) as the primary chlorinating agent, as disclosed in prior art such as US20060135602A1. While effective in converting the hydroxyl group to a chloride, this conventional approach suffers from severe drawbacks that impact both operational safety and cost efficiency. The stoichiometric requirement for thionyl chloride is often excessive, leading to inflated raw material costs and the generation of substantial quantities of corrosive waste acid and sulfur dioxide gas. For a reliable agrochemical intermediate supplier, managing the disposal of these hazardous byproducts adds a significant layer of complexity and expense to the manufacturing process. Additionally, the harsh reaction conditions associated with thionyl chloride can promote side reactions, resulting in a complex impurity profile that necessitates rigorous and costly purification steps to meet pharmaceutical grade standards.
The Novel Approach
In stark contrast, the method described in CN116171270A utilizes hydrogen chloride (HCl), typically in the form of concentrated hydrochloric acid (30-38% w/w), to effect the ring-opening chlorination of L-homoserine lactone hydrochloride. This innovative approach drastically reduces the relative usage of chlorinating agents and eliminates the introduction of sulfur contaminants into the reaction matrix. The process is remarkably flexible, capable of being conducted under atmospheric pressure or elevated pressure (0.1-1.0 MPa), offering manufacturers the ability to tune reaction kinetics based on their specific equipment capabilities. Moreover, the HCl used in the reaction can be potentially recovered and recycled, creating a closed-loop system that significantly mitigates environmental impact. This shift not only simplifies the waste treatment protocol but also enhances the overall atom economy of the synthesis, making it a superior choice for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into HCl-Mediated Ring-Opening Chlorination
The core of this technological advancement lies in the acid-catalyzed ring-opening of the lactone moiety followed by nucleophilic substitution. The reaction initiates with the protonation of the lactone oxygen by the strong acidic medium, rendering the carbonyl carbon highly susceptible to nucleophilic attack. In the presence of high concentrations of chloride ions provided by the hydrochloric acid, the ring opens to form the linear chloro-acid structure. The stereochemistry at the alpha-carbon is preserved throughout this transformation, ensuring the optical purity of the final product, which is critical for the biological activity of downstream herbicides. The patent data indicates that maintaining a molar ratio of HCl to substrate between 3:1 and 4:1 is optimal for driving the equilibrium towards the desired product while minimizing the co-precipitation of unreacted starting material during the crystallization phase.
Temperature control plays a pivotal role in the mechanistic pathway and the subsequent isolation of the product. The reaction is preferably conducted at temperatures ranging from 80°C to 130°C, with 100°C identified as the optimal setpoint for balancing reaction rate and product stability. Following the reaction, the crystallization behavior is heavily influenced by the cooling profile. The patent highlights that allowing the reaction mixture to cool naturally within the oil bath environment, rather than rapid external cooling, facilitates the formation of high-purity crystals. This slow cooling rate prevents the occlusion of impurities and unreacted lactone within the crystal lattice, a phenomenon often observed when the mixture is quenched rapidly to room temperature or below. This nuanced understanding of crystallization thermodynamics is essential for achieving the high relative content (>90%) reported in the experimental examples.
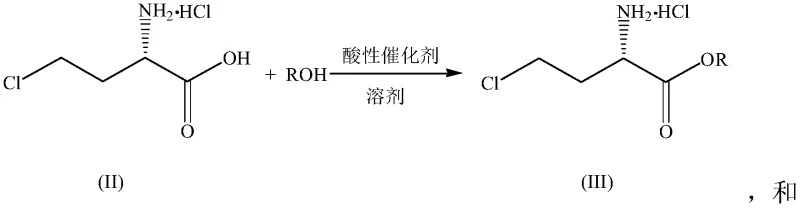
Following the chlorination, the resulting (S)-4-chloro-2-aminobutyric acid hydrochloride can be directly subjected to esterification. This step involves reacting the acid with an alcohol (ROH), preferably ethanol, in the presence of an acidic catalyst such as concentrated sulfuric acid. The mechanism follows a standard Fischer esterification pathway, where the acid catalyst protonates the carboxylic acid, facilitating nucleophilic attack by the alcohol. The use of sulfuric acid in catalytic amounts (0.05-0.4 eq) ensures high conversion rates without introducing excessive acid waste. The subsequent neutralization step, typically using ammonia water, liberates the free amine ester, which can then be extracted into organic solvents like dichloromethane or methyl tert-butyl ether. This modular approach allows for the efficient production of various ester derivatives tailored to specific synthetic needs.
How to Synthesize (S)-4-Chloro-2-aminobutyric Acid Hydrochloride Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production. It begins with the precise weighing of L-homoserine lactone hydrochloride and its suspension in concentrated hydrochloric acid. The addition of a catalytic amount of sulfuric acid can further accelerate the reaction, although it is not strictly mandatory. The mixture is then heated to reflux or maintained at 100°C for approximately 16 hours to ensure complete conversion. Upon completion, the critical step of crystallization is managed by ceasing heat input and allowing the system to equilibrate slowly, maximizing the recovery of pure product. For detailed operational parameters, safety guidelines, and specific workup procedures, please refer to the standardized synthesis guide below.
- Perform ring-opening chlorination of L-homoserine lactone hydrochloride using concentrated hydrochloric acid at elevated temperatures (80-130°C) to generate the chloro-acid intermediate.
- Conduct acid-catalyzed esterification of the chloro-acid intermediate with ethanol to form the corresponding ethyl ester hydrochloride salt.
- Neutralize the ester hydrochloride salt with a base such as ammonia water and extract with organic solvents to isolate the final free base ester product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this HCl-based chlorination technology offers substantial benefits that extend beyond mere technical feasibility. For procurement managers tasked with optimizing the cost structure of agrochemical intermediate supply chains, the elimination of thionyl chloride represents a direct reduction in raw material expenditure. Thionyl chloride is not only more expensive per mole compared to hydrochloric acid but also incurs higher logistics and storage costs due to its hazardous classification. By switching to a commodity chemical like concentrated HCl, manufacturers can achieve significant cost savings while simultaneously simplifying their inventory management protocols. Furthermore, the ability to recycle unreacted starting materials, as demonstrated in the patent's cyclic experiments, ensures that the effective yield of the process approaches theoretical maximums, thereby reducing the cost per kilogram of the final active ingredient.
- Cost Reduction in Manufacturing: The replacement of expensive thionyl chloride with readily available hydrochloric acid drastically lowers the direct material costs associated with the chlorination step. Additionally, the reduction in hazardous waste generation translates to lower disposal fees and reduced burden on effluent treatment plants, contributing to a leaner overall manufacturing budget. The process also minimizes the need for complex purification steps to remove sulfur-containing impurities, further streamlining production expenses.
- Enhanced Supply Chain Reliability: Hydrochloric acid is a globally available commodity chemical with a stable supply chain, unlike specialized reagents that may be subject to market volatility or regulatory restrictions. This availability ensures consistent production schedules and reduces the risk of supply disruptions. The robustness of the reaction conditions, which tolerate a range of pressures and temperatures, also means that the process can be easily transferred between different manufacturing sites without requiring specialized high-pressure equipment, enhancing supply flexibility.
- Scalability and Environmental Compliance: The process is inherently scalable, having been validated from gram to multi-kilogram scales in the patent examples. The environmental profile is significantly improved due to the absence of sulfur dioxide emissions and the potential for acid recycling. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, making the supply of these intermediates more resilient against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on process optimization and quality control measures. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this method into existing production lines.
Q: What are the advantages of using HCl over thionyl chloride for this synthesis?
A: Using hydrogen chloride (HCl) significantly reduces raw material costs compared to thionyl chloride and eliminates the generation of large amounts of waste acid and sulfur-containing byproducts, leading to a cleaner impurity profile and easier downstream processing.
Q: How does the crystallization method affect the purity of Compound II?
A: Natural cooling of the reaction mixture within the oil bath environment after heating cessation yields crystals with higher relative content and purity compared to rapid cooling or external room temperature cooling, as it prevents the co-precipitation of unreacted starting materials.
Q: Can the unreacted starting material be recycled in this process?
A: Yes, the process allows for the recovery of unreacted L-homoserine lactone hydrochloride from the filtrate, which can be recycled into subsequent batches, enabling a total cumulative yield of over 90% for the chlorination step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-4-Chloro-2-aminobutyric Acid Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity chiral intermediates in the development of next-generation agrochemicals and pharmaceuticals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify the identity and optical purity of every batch. Our infrastructure is designed to support the complex chemistry required for amino acid derivatives, guaranteeing a consistent and reliable supply for your R&D and commercial needs.
We invite you to collaborate with us to leverage this advanced HCl-mediated chlorination technology for your specific applications. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this process can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to drive efficiency and innovation in your supply chain.
