Advanced Ultrasound-Assisted Synthesis of 2-Arylformyl Benzoxazole Pharmaceutical Intermediates
Advanced Ultrasound-Assisted Synthesis of 2-Arylformyl Benzoxazole Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign methodologies for constructing complex heterocyclic scaffolds. A significant breakthrough in this domain is documented in Chinese Patent CN112961116B, which details a novel synthetic route for 2-arylformyl benzoxazole compounds. These structures serve as critical pharmacophores in the development of anticancer, antitumor, and antibacterial agents, making their efficient production a priority for reliable pharmaceutical intermediate suppliers. The patented technology leverages a synergistic combination of ultrasonic irradiation, acidic deep eutectic solvents (DES), and cuprous salt catalysis to drive an intramolecular cyclization-oxidation tandem reaction. This approach represents a paradigm shift from traditional fossil-fuel-based solvent systems to sustainable biomass-derived media, offering a compelling solution for cost reduction in API manufacturing while maintaining rigorous purity standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-arylformyl benzoxazole derivatives has relied heavily on the use of toxic, volatile organic solvents such as dichloromethane or dimethylformamide, which pose significant environmental and safety hazards during large-scale production. Conventional protocols often necessitate the use of stoichiometric amounts of harsh oxidants and strong acid or base additives to drive the cyclization process, leading to complex workup procedures and substantial waste generation. Furthermore, the recovery and recycling of these fossil-based solvents are energetically intensive and economically inefficient, often resulting in high operational expenditures that erode profit margins for chemical manufacturers. The reliance on such non-green chemistries also creates supply chain vulnerabilities, as regulatory pressures increasingly restrict the use of hazardous reagents, forcing procurement managers to seek alternative, compliant sourcing strategies for high-purity OLED material and pharmaceutical precursors.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN112961116B introduces a streamlined, one-pot protocol that utilizes an acidic choline chloride/oxalic acid deep eutectic solvent as both the reaction medium and a co-catalyst. This biomass-derived solvent system is not only biodegradable and non-toxic but also facilitates the activation of the N-(2-hydroxyphenyl)-2-arylacetamide substrate under mild thermal conditions. By integrating ultrasonic energy into the process, the method overcomes the mass transfer limitations typically associated with viscous ionic liquids, ensuring rapid and homogeneous mixing without the need for aggressive mechanical stirring. The result is a highly selective transformation that proceeds efficiently under an ambient air atmosphere, eliminating the need for expensive pure oxygen sources or dangerous peroxide oxidants, thereby significantly simplifying the commercial scale-up of complex polymer additives and drug intermediates.

Mechanistic Insights into CuI-Catalyzed Cyclization-Oxidation
The mechanistic pathway of this transformation involves a sophisticated interplay between the copper catalyst, the acidic solvent environment, and ultrasonic energy to achieve high-yield cyclization. Initially, the acidic deep eutectic solvent activates the amide carbonyl of the starting material through hydrogen bonding interactions, promoting an intramolecular nucleophilic attack by the ortho-hydroxyl group to form a cyclic hemiaminal intermediate. This intermediate subsequently undergoes dehydration and aromatization to generate a stable 2-arylmethylbenzoxazole species, which serves as the precursor to the final ketone product. The presence of cuprous iodide is critical at this stage, as it mediates the oxidation step through a redox cycle where Cu(I) is oxidized by molecular oxygen from the air to form a reactive Cu(II)-peroxo species. This active oxidant then selectively converts the benzylic methylene group into the corresponding carbonyl functionality, regenerating the Cu(I) catalyst to close the loop.
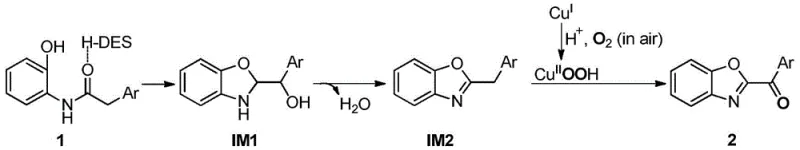
Understanding the impurity control mechanisms inherent in this process is vital for R&D directors focused on purity profiles and downstream processing. The mild reaction temperature of 60°C prevents thermal degradation of the sensitive benzoxazole ring, which is a common issue in high-temperature thermal cyclizations. Moreover, the specific acidity of the choline chloride/oxalic acid system suppresses side reactions such as over-oxidation or polymerization of the aryl rings, ensuring a clean crude product profile. The ultrasonic cavitation effect further contributes to impurity minimization by preventing the aggregation of copper species, which can otherwise lead to heterogeneous hotspots and uneven reaction rates. This level of control allows for the direct isolation of high-purity intermediates with minimal chromatographic purification, aligning perfectly with the stringent quality requirements for reducing lead time for high-purity pharmaceutical intermediates in a GMP environment.
How to Synthesize 2-Arylformyl Benzoxazole Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to precise parameters regarding catalyst loading, solvent composition, and ultrasonic settings to maximize efficiency. The standard protocol involves dissolving the N-(2-hydroxyphenyl)-2-arylacetamide substrate in the pre-formed acidic deep eutectic solvent, followed by the addition of 25 mol% cuprous iodide. The reaction vessel is then subjected to ultrasonic irradiation at a power of 50W and a frequency of 68KHz while maintaining a constant temperature of 60°C under an open air atmosphere. Detailed standardized synthesis steps for this procedure are provided in the guide below.
- Prepare the reaction mixture by combining N-(2-hydroxyphenyl)-2-arylacetamide substrate with 25 mol% cuprous iodide catalyst in an acidic choline chloride/oxalic acid deep eutectic solvent.
- Subject the mixture to ultrasonic irradiation at 50W power and 68KHz frequency while maintaining the temperature at 60°C under an air atmosphere for approximately 30 minutes.
- Upon completion, extract the product using ethyl acetate, allowing the deep eutectic solvent and catalyst raffinate to be recycled for subsequent batches.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ultrasound-assisted DES methodology offers transformative economic and logistical benefits that extend far beyond simple yield improvements. By replacing volatile organic solvents with recyclable deep eutectic solvents, manufacturers can drastically reduce the costs associated with solvent purchase, storage, and hazardous waste disposal, leading to substantial cost savings in the overall production budget. The ability to recycle the catalytic system for multiple batches without significant performance degradation means that the effective cost of the copper catalyst per kilogram of product is minimized, enhancing the overall cost-effectiveness of the supply chain. Furthermore, the use of ambient air as the terminal oxidant eliminates the safety risks and infrastructure costs linked to handling high-pressure oxygen or unstable peroxide reagents, thereby improving facility safety profiles and insurance premiums.
- Cost Reduction in Manufacturing: The elimination of expensive fossil solvents and stoichiometric oxidants directly lowers the bill of materials, while the recyclability of the reaction medium reduces waste treatment fees significantly. This process optimization allows for a leaner manufacturing model where resource utilization is maximized, and the dependency on fluctuating petrochemical markets is reduced, ensuring more stable pricing for long-term contracts.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including choline chloride, oxalic acid, and cuprous iodide, are commodity chemicals with robust global supply chains, minimizing the risk of production stoppages due to raw material shortages. The simplicity of the workup procedure, which involves basic extraction and solvent removal, shortens the production cycle time, enabling faster turnaround for urgent orders and improving the responsiveness of the supply chain to market demands.
- Scalability and Environmental Compliance: The green nature of this process aligns with increasingly strict environmental regulations, future-proofing the production facility against potential bans on hazardous solvents. The scalability is enhanced by the efficient heat and mass transfer provided by ultrasound, which mitigates the engineering challenges typically faced when scaling up viscous solvent systems, ensuring consistent product quality from gram-scale R&D to multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route, derived directly from the experimental data and technical specifications outlined in the patent documentation. These insights are intended to assist technical teams in evaluating the feasibility of adopting this methodology for their specific intermediate manufacturing needs. Comprehensive answers regarding catalyst recovery, substrate scope, and reaction kinetics are detailed in the section below.
Q: Can the deep eutectic solvent system be recycled?
A: Yes, the acidic deep eutectic solvent and cuprous salt catalytic system can be recycled and reused for more than 5 cycles without significant loss in catalytic activity or product yield.
Q: What is the optimal catalyst loading for this transformation?
A: Experimental data indicates that 25 mol% of cuprous iodide (CuI) provides the optimal balance between reaction efficiency and cost, yielding over 80% conversion under standard conditions.
Q: Is this method compatible with various functional groups?
A: The method exhibits excellent functional group tolerance, successfully accommodating substrates with electron-donating groups like methoxy and methyl, as well as electron-withdrawing halogens.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Arylformyl Benzoxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative, green chemistry solutions to meet the evolving demands of the global pharmaceutical market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality intermediates regardless of volume. Our state-of-the-art facilities are equipped with advanced ultrasonic reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-arylformyl benzoxazole delivered meets the highest industry standards for potency and impurity profiles.
We invite potential partners to engage with our technical procurement team to discuss how this patented technology can be integrated into your supply chain to drive efficiency and sustainability. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your project volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in the synthesis of complex organic intermediates.
