Revolutionizing Quinolone Manufacturing: Safe One-Pot Synthesis via Solid Carbonyl Sources
The pharmaceutical and fine chemical industries are constantly seeking safer, more efficient pathways to synthesize complex heterocyclic scaffolds, particularly quinolones, which serve as vital intermediates for a wide array of bioactive molecules. A groundbreaking approach detailed in Chinese patent CN110204489B introduces a novel "one-pot" methodology that fundamentally shifts the paradigm of carbonylation chemistry. Instead of relying on hazardous high-pressure carbon monoxide gas, this invention utilizes chromium hexacarbonyl as a solid, manageable carbonyl source activated by a unique amine system. This technical breakthrough not only mitigates significant safety risks associated with traditional carbonylation but also streamlines the operational workflow, making it an attractive candidate for large-scale commercial production. By operating under mild conditions in an air atmosphere, this process addresses the critical pain points of equipment complexity and toxicity control that have long plagued the synthesis of nitrogen-containing heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolone derivatives via carbonylation has been dominated by methods requiring gaseous carbon monoxide as the C1 building block. Pioneering work by the Torii group in 1991 and subsequent improvements by Genelot established the feasibility of reacting 2-iodoanilines with terminal alkynes under CO pressure. However, these conventional techniques suffer from severe inherent drawbacks that limit their industrial applicability. The primary concern is the extreme toxicity and flammability of carbon monoxide gas, which necessitates the use of specialized high-pressure autoclaves and rigorous safety protocols to prevent leaks and exposure. Furthermore, controlling the stoichiometry of a gaseous reagent in a liquid-phase reaction is notoriously difficult, often leading to inconsistent reaction kinetics and variable yields. The requirement for inert atmospheres, typically nitrogen or argon, adds another layer of operational cost and complexity, making these processes less desirable for cost-sensitive manufacturing environments where safety and simplicity are paramount.
The Novel Approach
In stark contrast to the hazardous traditional methods, the technology disclosed in patent CN110204489B offers a remarkably safe and efficient alternative by employing chromium hexacarbonyl as a solid carbonyl donor. This solid source is stable at room temperature and easy to handle, effectively eliminating the need for high-pressure gas cylinders and complex delivery systems. The innovation lies in the activation mechanism: under an air atmosphere, a specific combination of triethylamine and piperazine activates the chromium hexacarbonyl to release carbon monoxide in situ at a controlled rate. This slow release perfectly matches the kinetics of the palladium-catalyzed cross-coupling reaction, ensuring that the CO is available exactly when needed for the cyclization step.  This "one-pot" strategy not only simplifies the reactor setup to standard glassware capable of handling ambient pressure but also drastically reduces the environmental footprint by avoiding the venting of excess toxic gas, representing a significant leap forward in green chemistry principles for heterocycle synthesis.
This "one-pot" strategy not only simplifies the reactor setup to standard glassware capable of handling ambient pressure but also drastically reduces the environmental footprint by avoiding the venting of excess toxic gas, representing a significant leap forward in green chemistry principles for heterocycle synthesis.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
The core of this synthetic strategy relies on a sophisticated palladium-catalyzed cascade involving Sonogashira-type coupling followed by carbonylation and intramolecular cyclization. The catalytic cycle begins with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the 2-iodoaniline substrate. Simultaneously, the terminal alkyne undergoes transmetallation, facilitated by the base system. The critical divergence from standard Sonogashira coupling occurs here, where the in situ generated carbon monoxide inserts into the palladium-carbon bond to form an acyl-palladium intermediate. This acyl species then undergoes nucleophilic attack by the adjacent amino group on the aniline ring, closing the quinolone skeleton and regenerating the active palladium catalyst. The elegance of this mechanism is heavily dependent on the precise timing of CO availability; too rapid a release could lead to polymerization or side reactions, while too slow a release would stall the catalytic cycle. The patent data indicates that the specific interplay between the palladium catalyst, the Xantphos ligand, and the amine activators creates a harmonious kinetic profile that drives the reaction to completion with high selectivity.
Furthermore, the substrate scope of this reaction is exceptionally broad, demonstrating robust tolerance to various electronic and steric environments. As illustrated in the structural variations allowed for the starting materials, the 2-iodoaniline component can bear diverse substituents such as halogens (chloro, fluoro) or alkyl groups at multiple positions without compromising the reaction efficiency. 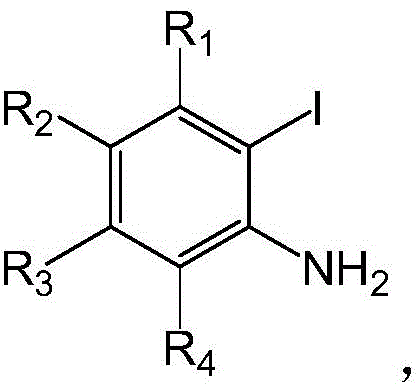 Similarly, the terminal alkyne partner accepts a wide range of aryl and heteroaryl groups, including electron-rich methoxy-substituted phenyl rings and electron-deficient nitrile-containing aromatics, as well as heterocycles like thiophene. This versatility is crucial for medicinal chemists who need to rapidly generate libraries of quinolone analogs for structure-activity relationship (SAR) studies. The ability to maintain high yields across such a diverse set of substrates suggests that the catalytic system is highly resilient to impurities and functional group interference, a key indicator of a robust manufacturing process capable of handling real-world feedstock variability.
Similarly, the terminal alkyne partner accepts a wide range of aryl and heteroaryl groups, including electron-rich methoxy-substituted phenyl rings and electron-deficient nitrile-containing aromatics, as well as heterocycles like thiophene. This versatility is crucial for medicinal chemists who need to rapidly generate libraries of quinolone analogs for structure-activity relationship (SAR) studies. The ability to maintain high yields across such a diverse set of substrates suggests that the catalytic system is highly resilient to impurities and functional group interference, a key indicator of a robust manufacturing process capable of handling real-world feedstock variability.
How to Synthesize 2-Phenyl-4H-benzoquinol-4-one Efficiently
To implement this advanced synthesis in a laboratory or pilot plant setting, operators must adhere to the optimized parameters defined in the patent to ensure maximum conversion and purity. The process involves dissolving the 2-iodoaniline and terminal alkyne substrates in anhydrous acetonitrile, followed by the sequential addition of the palladium catalyst, the bidentate phosphine ligand, and the solid chromium hexacarbonyl source. The critical step is the introduction of the mixed base system, where triethylamine and piperazine are added in a carefully controlled molar ratio to initiate the CO release. Detailed standardized synthesis steps are provided in the guide below.
- Prepare the reaction mixture by combining 2-iodoaniline derivatives, terminal alkynes, chromium hexacarbonyl, palladium acetate catalyst, and Xantphos ligand in acetonitrile solvent.
- Add a mixed base system consisting of triethylamine and piperazine in a specific molar ratio to activate the solid carbonyl source.
- Heat the reaction mixture to 50-60°C under an air atmosphere for 10-12 hours to facilitate the carbonylation cyclization, then isolate the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from gaseous CO methods to this solid-source protocol offers tangible strategic benefits that extend far beyond the laboratory bench. The most immediate impact is on capital expenditure and operational safety costs. By eliminating the need for high-pressure autoclaves and specialized gas handling infrastructure, facilities can utilize standard stainless steel reactors, significantly lowering the barrier to entry for production. Moreover, the use of a solid reagent simplifies logistics; chromium hexacarbonyl can be shipped and stored using standard chemical warehousing protocols, unlike compressed gas cylinders which require stringent regulatory compliance and dedicated storage zones. This shift reduces the administrative burden and insurance costs associated with hazardous material management, allowing for a more agile and responsive supply chain.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the simplification of the reaction setup and the efficiency of the reagent usage. Traditional carbonylation often suffers from poor atom economy due to the difficulty in controlling gas flow, leading to wasted reagents and the need for extensive purification to remove metal carbonyl byproducts. In this novel method, the solid source releases CO stoichiometrically matched to the reaction demand, minimizing waste. Additionally, the elimination of expensive noble metal scavengers often required to remove residual catalysts from high-pressure runs further drives down the cost of goods sold (COGS). The mild reaction temperature of 50-60°C also translates to substantial energy savings compared to processes requiring high heat or cryogenic cooling, contributing to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for any pharmaceutical intermediate, and this method enhances reliability by decoupling production from the volatility of industrial gas supplies. Gaseous CO is often a byproduct of other industrial processes and can be subject to supply disruptions or regional shortages. In contrast, chromium hexacarbonyl is a commercially available bulk chemical with a stable global supply chain. The robustness of the reaction conditions, which tolerate air and moisture better than strict inert atmosphere methods, also reduces the risk of batch failures due to minor environmental fluctuations. This resilience ensures that production schedules can be met consistently, reducing the risk of downstream delays for API manufacturers who depend on timely delivery of these key heterocyclic building blocks.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this technology is inherently designed for scalability. The absence of high-pressure gas removes the primary scaling bottleneck, allowing for straightforward translation from liter-scale batches to multi-ton production without exponential increases in safety engineering controls. From an environmental perspective, the "one-pot" nature of the reaction reduces solvent consumption and waste generation associated with multi-step sequences. The use of a solid carbonyl source prevents the direct emission of carbon monoxide into the workplace atmosphere, aligning with increasingly stringent occupational health and safety regulations. This compliance advantage future-proofs the manufacturing site against tightening environmental laws, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solid-source carbonylation technology. These answers are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the practical aspects of adopting this method for industrial synthesis. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is chromium hexacarbonyl preferred over gaseous CO for quinolone synthesis?
A: Chromium hexacarbonyl serves as a stable, solid CO source that eliminates the safety hazards associated with high-pressure toxic carbon monoxide gas. It allows the reaction to proceed under ambient pressure and air atmosphere, significantly simplifying equipment requirements and operational safety protocols.
Q: What is the role of the triethylamine and piperazine mixed base system?
A: The synergistic combination of triethylamine and piperazine is critical for efficiently activating chromium hexacarbonyl to release CO in situ. This specific base pairing matches the kinetics of the palladium-catalyzed cross-coupling reaction, ensuring high yields and preventing the accumulation of unreacted intermediates.
Q: Can this method accommodate diverse substrate substituents?
A: Yes, the protocol demonstrates excellent substrate tolerance. It successfully accommodates various substituents on both the 2-iodoaniline ring (such as methyl, chloro, or fluoro groups) and the terminal alkyne (including phenyl, alkyl-substituted phenyl, and heterocycles like thiophene), yielding target quinolones with high purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinolone Supplier
The technological advancements described in patent CN110204489B represent a significant opportunity for the pharmaceutical industry to produce high-quality quinolone intermediates more safely and economically. At NINGBO INNO PHARMCHEM, we recognize the potential of this solid-carbonyl methodology and have integrated similar state-of-the-art carbonylation capabilities into our CDMO service offerings. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to full-scale manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of quinolone intermediate meets the exacting standards required for drug substance synthesis.
We invite potential partners to leverage our technical expertise to optimize their supply chains for these critical heterocyclic compounds. Whether you require custom synthesis of specific quinolone derivatives or scale-up of existing routes, our engineering team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs. We encourage you to contact our technical procurement team today to request specific COA data for our catalog items or to discuss route feasibility assessments for your proprietary molecules, ensuring a secure and efficient supply of high-purity pharmaceutical intermediates for your global operations.
