Scalable One-Pot Synthesis of High-Purity Diphenyl Disulfide Intermediates for Pharmaceutical Applications
Scalable One-Pot Synthesis of High-Purity Diphenyl Disulfide Intermediates for Pharmaceutical Applications
The landscape of fine chemical manufacturing is constantly evolving, driven by the urgent need for safer, more efficient, and environmentally sustainable synthetic routes. A significant breakthrough in this domain is detailed in patent CN111763163B, which discloses a novel preparation method for diphenyl disulfide compounds. This technology represents a paradigm shift away from hazardous traditional methodologies, utilizing a sophisticated one-pot halogen-magnesium exchange strategy. By leveraging isopropylmagnesium halide Grignard reagents and dichlorodisulfide, this process enables the rapid construction of carbon-sulfur bonds under mild conditions. For R&D directors and process chemists, this patent offers a compelling alternative that addresses critical pain points regarding toxicity, operational complexity, and waste generation. The ability to synthesize these vital intermediates with high efficiency and minimal environmental impact positions this technology as a cornerstone for modern pharmaceutical and agrochemical supply chains.
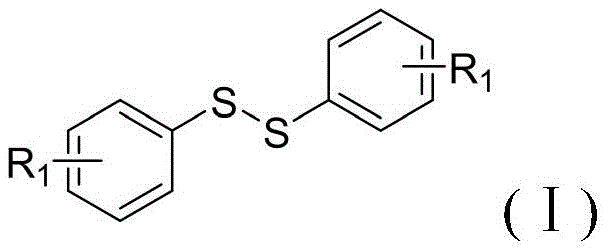
Diphenyl disulfides serve as indispensable building blocks in the synthesis of complex active pharmaceutical ingredients (APIs), agrochemicals, and functional materials. The structural versatility allowed by the substituent R1, which can range from alkyl groups to base-sensitive cyano or halogen moieties, underscores the broad applicability of this synthetic platform. As depicted in the general structure, the symmetrical or asymmetrical nature of the disulfide bridge can be precisely controlled, offering chemists a robust toolkit for molecular design. The implementation of this patented method not only streamlines the production of these compounds but also aligns with the rigorous quality standards demanded by global regulatory bodies. By adopting this advanced synthetic route, manufacturers can secure a reliable diphenyl disulfide supplier status while simultaneously enhancing their internal process safety profiles and reducing their overall carbon footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of diphenyl disulfide derivatives has been plagued by significant safety and environmental challenges associated with the use of thiophenols as starting materials. Thiophenols are notorious for their extremely unpleasant, penetrating odors which can cause severe nausea and headaches even at trace concentrations, posing a substantial occupational health hazard. Furthermore, these thiol compounds are chemically unstable, highly toxic, and possess explosive characteristics that necessitate specialized storage and handling infrastructure, drastically inflating operational costs. Conventional oxidative coupling routes using thiophenols often require harsh reaction conditions, strong oxidants, and transition metal catalysts, which can lead to difficult-to-remove metal impurities in the final product. The lengthy synthetic sequences and the generation of large volumes of hazardous waste streams make these traditional processes increasingly untenable in the context of modern green chemistry initiatives and strict environmental regulations.
The Novel Approach
In stark contrast, the methodology outlined in patent CN111763163B introduces a transformative one-pot synthesis that completely bypasses the use of free thiophenols. By employing substituted halobenzenes and isopropylmagnesium halides, the process initiates a controlled halogen-magnesium exchange to generate the reactive phenyl Grignard species in situ. This intermediate is then immediately trapped by dichlorodisulfide, facilitating a rapid and efficient C-S bond coupling without the need for external catalysts. This approach not only eliminates the handling of malodorous and toxic thiols but also significantly shortens the synthetic route, thereby reducing the time and resources required for production. The simplicity of the workup procedure, involving standard aqueous quenching and extraction, further enhances the practicality of this method for large-scale manufacturing, making it an ideal solution for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Halogen-Magnesium Exchange and S-S Bond Formation
The core of this innovative synthesis lies in the precise execution of the halogen-magnesium exchange reaction, a fundamental transformation in organometallic chemistry. The process begins with the interaction between a substituted halobenzene and isopropylmagnesium halide in an anhydrous organic solvent such as tetrahydrofuran (THF) or diethyl ether. Maintaining the reaction temperature within the critical range of -78°C to -20°C is paramount to ensuring complete exchange while suppressing competing side reactions such as Wurtz-type coupling or nucleophilic attack on sensitive functional groups. The kinetic control afforded by these low temperatures allows for the generation of a high concentration of the substituted phenyl Grignard reagent, which acts as a potent nucleophile. This species is then poised to attack the electrophilic sulfur centers in dichlorodisulfide, driving the formation of the disulfide linkage with high regioselectivity and minimal byproduct formation.
Following the formation of the Grignard reagent, the addition of dichlorodisulfide triggers the crucial coupling step that constructs the target disulfide framework. The reaction mechanism likely proceeds through a nucleophilic substitution at the sulfur atom, displacing a chloride ion and forming a transient sulfenyl intermediate which rapidly reacts with a second equivalent of the Grignard species or undergoes disproportionation to yield the stable disulfide product. One of the most remarkable aspects of this mechanism is its tolerance for various functional groups; for instance, cyano groups, which are typically sensitive to strong nucleophiles, are preserved intact under these optimized conditions. This functional group compatibility is a major advantage for R&D teams designing complex molecules, as it removes the need for extensive protecting group strategies. The subsequent quenching with saturated ammonium chloride effectively neutralizes any remaining organometallic species, allowing for a straightforward isolation of the high-purity product via simple extraction and concentration.
How to Synthesize Diphenyl Disulfide Efficiently
The practical implementation of this synthesis requires careful attention to reaction parameters to maximize yield and safety. The patented procedure outlines a robust protocol where substituted halobenzenes are treated with isopropylmagnesium halide at cryogenic temperatures, followed by the introduction of the sulfur source. This sequence ensures that the highly reactive Grignard intermediate is consumed immediately, minimizing decomposition pathways. The detailed standardized synthesis steps provided below offer a clear roadmap for laboratory and pilot-scale execution, ensuring reproducibility and consistency across different batches. By adhering to these guidelines, manufacturers can achieve the reported yields of 80-91% and purities exceeding 95%, meeting the stringent requirements for downstream applications.
- Perform halogen-magnesium exchange between substituted halobenzene and isopropylmagnesium halide in THF at -78°C to -20°C.
- Add dichlorodisulfide (S2Cl2) to the generated Grignard reagent solution to effect C-S bond formation.
- Quench the reaction with saturated ammonium chloride, extract with ethyl acetate or ether, and concentrate to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology translates into tangible strategic benefits that extend far beyond the laboratory bench. The elimination of thiophenol from the supply chain removes a significant logistical bottleneck, as sourcing and transporting hazardous, malodorous thiols often incur premium costs and regulatory burdens. By switching to stable, commodity-grade aryl halides and Grignard reagents, companies can leverage existing supply networks and reduce raw material volatility. Furthermore, the simplified one-pot nature of the reaction reduces the number of unit operations required, leading to lower energy consumption and reduced demand for reactor time. This operational efficiency directly contributes to substantial cost savings in production, allowing organizations to maintain competitive pricing margins while adhering to strict budgetary constraints.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the removal of expensive transition metal catalysts and the avoidance of complex purification steps required to remove metal residues. Traditional cross-coupling methods often rely on palladium or copper catalysts, which are not only costly but also require extensive downstream processing to meet residual metal specifications for pharmaceutical grades. By utilizing a catalyst-free Grignard pathway, this method inherently produces cleaner crude products, significantly reducing the load on purification units and lowering the overall cost of goods sold. Additionally, the high atom economy and excellent yields minimize raw material waste, further enhancing the financial viability of the process for commercial scale-up of complex sulfur-containing intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the availability and stability of raw materials. Aryl halides and isopropylmagnesium halides are widely produced commodity chemicals with robust global supply chains, unlike specialized thiols which may have limited suppliers and long lead times. This shift in raw material strategy mitigates the risk of supply disruptions and price spikes, ensuring a steady flow of materials for continuous production. Moreover, the improved safety profile of the process reduces the likelihood of shutdowns due to safety incidents or environmental compliance issues, thereby guaranteeing consistent delivery schedules for customers. This reliability is essential for reducing lead time for high-purity disulfides, enabling faster time-to-market for new drug candidates.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to multi-ton production often reveals hidden challenges, but this one-pot method is inherently designed for scalability. The exothermic nature of the Grignard formation is manageable within standard industrial reactors equipped with cooling capabilities, and the absence of hazardous gases or explosive intermediates simplifies the engineering controls required. From an environmental perspective, the process generates significantly less hazardous waste compared to oxidative coupling methods, aligning with corporate sustainability goals and reducing disposal costs. The use of common solvents like THF and ether, which can be efficiently recovered and recycled, further minimizes the environmental footprint, making this technology a responsible choice for modern chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this diphenyl disulfide synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process capabilities and limitations. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this method into their existing manufacturing portfolios.
Q: Why is this Grignard-based route superior to traditional thiophenol oxidation?
A: Traditional methods rely on thiophenol, which is highly toxic, malodorous, and prone to explosion. This patented Grignard route utilizes stable aryl halides and avoids hazardous thiol handling, significantly improving operator safety and environmental compliance.
Q: What is the typical yield and purity achievable with this method?
A: According to patent CN111763163B, the method consistently achieves yields between 80% and 91% with product purity exceeding 95%, eliminating the need for complex purification steps often required in metal-catalyzed couplings.
Q: Can this process tolerate sensitive functional groups like nitriles?
A: Yes, the controlled low-temperature halogen-magnesium exchange conditions allow for the tolerance of base-sensitive groups such as cyano groups, which might decompose under harsher nucleophilic substitution conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diphenyl Disulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in patent CN111763163B and are fully equipped to leverage this technology for our global partners. As a premier CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to full-scale manufacturing. Our state-of-the-art facilities are designed to handle sensitive organometallic reactions with the utmost precision, supported by rigorous QC labs that enforce stringent purity specifications on every batch. We are committed to delivering high-quality diphenyl disulfide intermediates that meet the exacting standards of the pharmaceutical and agrochemical industries.
We invite you to collaborate with us to optimize your supply chain and achieve your cost targets through this advanced manufacturing approach. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your next project.
