Scalable Room-Temperature Synthesis of (RP)-Menthylphenylphosphine Oxide for Advanced Chiral Catalysis
Scalable Room-Temperature Synthesis of (RP)-Menthylphenylphosphine Oxide for Advanced Chiral Catalysis
The landscape of asymmetric synthesis is continually evolving, driven by the demand for more efficient and accessible chiral building blocks. A pivotal advancement in this domain is detailed in patent CN103665038A, which discloses a novel and robust methodology for the preparation of (RP)-menthylphenylphosphine oxide. This compound serves as a critical precursor for phosphorus-chiral ligands, which are indispensable in the manufacture of high-value pharmaceuticals and agrochemicals. Unlike traditional approaches that often rely on cumbersome resolution processes or extreme cryogenic conditions, this invention leverages the inherent stereochemical stability provided by the bulky menthyl group. By enabling the synthesis to proceed under ambient or mild reflux conditions, the technology offers a transformative pathway for industrial scale-up. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic and operational advantages of this route is essential for optimizing supply chains and reducing manufacturing costs in chiral ligand production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of phosphorus-chiral compounds has been fraught with significant technical and economic challenges. Conventional strategies typically involve the chemical resolution of racemic dihydrocarbyl phosphine oxides using tartaric acid derivatives, a process that necessitates multiple recrystallization steps and the subsequent removal of resolving agents. Alternatively, existing synthetic routes, such as those described by Xu et al., require the substitution of menthoxy groups on chiral phosphonites using lithium or Grignard reagents at extremely low temperatures, specifically around -78°C. These cryogenic requirements impose severe constraints on industrial applicability, demanding specialized cooling infrastructure and resulting in high energy expenditures. Furthermore, the stereoselectivity of reactions forming chiral phosphorus atoms has traditionally been low, leading to poor yields and complex purification workflows that hinder the commercial viability of phosphorus-chiral ligands compared to their carbon-chiral counterparts like BINAP.
The Novel Approach
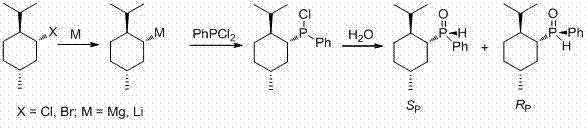
The methodology outlined in patent CN103665038A represents a paradigm shift by eliminating the need for cryogenic reactors and complex resolution agents. This innovative approach utilizes a direct reaction between menthyl magnesium halides or menthyl lithium solutions and phenylphosphine dichloride. Crucially, this transformation can be conducted at room temperature or under mild reflux conditions, drastically simplifying the operational parameters. The presence of the voluminous menthyl group not only facilitates the formation of the phosphorus-carbon bond but also stabilizes the configuration of the phosphorus atom, thereby enhancing asymmetric induction. This streamlined process allows for the direct isolation of the desired (RP)-isomer through straightforward recrystallization, bypassing the need for expensive chiral auxiliaries. For manufacturers focused on cost reduction in chiral ligand manufacturing, this route offers a compelling alternative that merges high stereocontrol with operational simplicity.
Mechanistic Insights into Grignard-Mediated Phosphorylation
The core of this synthetic strategy lies in the generation of a highly reactive organometallic species followed by a nucleophilic attack on the phosphorus center. The process initiates with the formation of menthyl magnesium halide or menthyl lithium from menthyl chloride or bromide in anhydrous ether or hydrocarbon solvents. This organometallic reagent then reacts with phenylphosphine dichloride, where the menthyl anion displaces a chloride ion to form phenyl menthyl phosphine chloride. The reaction kinetics are favorable enough to proceed without the extreme thermal management required by previous methods. Subsequent hydrolysis converts the phosphorus-chlorine bond into a phosphorus-hydroxyl bond, which rapidly isomerizes to the stable phosphine oxide form. The stereochemical outcome is governed by the chiral environment created by the menthyl moiety, which exerts significant steric influence over the phosphorus atom, favoring the formation of specific diastereomers.
Impurity control is inherently managed through the physical properties of the diastereomeric mixture generated during hydrolysis. The reaction produces a mixture of RP and SP configurations, but the distinct solubility differences between these isomers allow for effective purification. While low-temperature hydrolysis may slightly favor the RP isomer ratio, the patent demonstrates that room temperature hydrolysis is sufficient when coupled with optimized recrystallization protocols. The use of solvents such as petroleum ether, hexane, or ethanol enables the selective precipitation of the target (RP)-menthylphenylphosphine oxide. This mechanism ensures that the final product achieves an optical purity greater than 99%, meeting the stringent specifications required for high-purity pharmaceutical intermediates. The robustness of this mechanism against minor fluctuations in temperature further underscores its suitability for large-scale commercial operations.
How to Synthesize (RP)-Menthylphenylphosphine Oxide Efficiently
Implementing this synthesis requires careful attention to solvent dryness and reagent stoichiometry to maximize yield and purity. The process begins with the preparation of the Grignard or Lithium reagent under an inert atmosphere, followed by the controlled addition of phenylphosphine dichloride. Detailed standard operating procedures regarding addition rates, stirring times, and specific solvent volumes are critical for reproducibility. The following guide outlines the standardized synthesis steps derived from the patent examples, ensuring that technical teams can replicate the high-quality results demonstrated in the intellectual property.
- Prepare menthyl magnesium halide or menthyl lithium solution by reacting menthyl chloride or bromide with magnesium or lithium in anhydrous ether or hydrocarbon solvents.
- React the prepared organometallic reagent with phenylphosphine dichloride under reflux conditions to form phenyl menthyl phosphine chloride.
- Hydrolyze the intermediate with water or aqueous reagents, followed by extraction, drying, and recrystallization to isolate the (RP)-isomer with >99% optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the transition from cryogenic to ambient temperature processing offers profound logistical and financial benefits. The elimination of -78°C cooling requirements removes the dependency on specialized low-temperature reactors and excessive energy consumption, directly translating to reduced capital expenditure and lower operational costs. Furthermore, the simplified workup procedure, which avoids the use of complex chiral resolution agents, streamlines the downstream processing stages. This reduction in unit operations enhances the overall throughput of the manufacturing facility, allowing for faster batch turnover and improved responsiveness to market demand. For procurement managers, these efficiencies signify a more stable and cost-effective source of critical chiral intermediates.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of energy-intensive cryogenic steps and expensive resolving agents. By conducting the reaction at room temperature or under reflux, the facility saves significantly on cooling utilities and equipment maintenance. Additionally, the high crude yields reported, ranging from 70% to 95%, minimize raw material waste. The ability to purify the product via simple recrystallization rather than chromatography or multi-step resolution further reduces solvent consumption and labor costs, resulting in substantial overall cost savings for the production of complex chiral phosphine oxides.
- Enhanced Supply Chain Reliability: The use of readily available starting materials such as menthyl chloride, magnesium, and phenylphosphine dichloride ensures a resilient supply chain. These commodities are widely produced and do not suffer from the scarcity issues often associated with specialized chiral catalysts. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by equipment failure or environmental fluctuations. This reliability is crucial for maintaining continuous supply lines for downstream API manufacturers, reducing lead time for high-purity pharmaceutical intermediates and ensuring consistent availability for global clients.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard chemical engineering unit operations such as reflux and crystallization. The absence of hazardous cryogenic fluids and the reduction in solvent usage during purification contribute to a smaller environmental footprint. Waste streams are easier to manage as they primarily consist of common organic solvents and inorganic salts, facilitating compliance with increasingly stringent environmental regulations. This scalability supports the commercial scale-up of complex chiral ligands from kilogram to multi-ton quantities without compromising quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of (RP)-menthylphenylphosphine oxide. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and advantages of this technology for potential partners and technical stakeholders.
Q: What are the temperature advantages of this synthesis method compared to prior art?
A: Unlike conventional methods requiring cryogenic conditions at -78°C, this patented process operates effectively at room temperature or under mild reflux, significantly reducing energy consumption and equipment complexity.
Q: How is high optical purity achieved without complex resolution agents?
A: The method utilizes the steric bulk of the menthyl group to stabilize the phosphorus configuration, allowing for direct separation of the (RP)-isomer through simple recrystallization rather than multi-step chemical resolution.
Q: What is the typical yield and purity of the final product?
A: The process yields crude mixtures with 70% to 95% efficiency, and subsequent recrystallization delivers the target (RP)-menthylphenylphosphine oxide with an optical purity exceeding 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (RP)-Menthylphenylphosphine Oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-performance chiral intermediates in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Our capability to implement the room-temperature synthesis described in patent CN103665038A allows us to offer a competitive advantage in both pricing and delivery reliability for clients requiring phosphorus-chiral building blocks.
We invite you to collaborate with us to optimize your supply chain for chiral catalysis. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your R&D and commercial production goals efficiently.
