Advanced Synthesis of 2-Cyanopyrrole Compounds for Scalable Pharmaceutical Intermediate Production
Introduction to Next-Generation 2-Cyanopyrrole Synthesis
The pharmaceutical industry continuously seeks robust and scalable pathways for constructing nitrogen-containing heterocycles, particularly the pyrrole scaffold, which serves as a critical backbone in numerous bioactive molecules. Patent CN110903231B introduces a significant advancement in this domain by detailing a novel synthetic method for 2-cyanopyrrole compounds. These compounds are not merely academic curiosities; they are valuable organic synthesis intermediates capable of conversion into diverse active pharmaceutical ingredients (APIs), including HDAC inhibitors and anti-inflammatory agents. The disclosed technology leverages trifluoromethanesulfonic acid (TfOH) as a potent organocatalyst, enabling the reaction between N-(4-methylbenzenesulfonyl)cinnamamine derivatives and tert-butyl isonitrile. This approach represents a paradigm shift from traditional transition-metal-dependent methodologies, offering a cleaner, more atom-economical route that aligns perfectly with modern green chemistry principles and the stringent purity requirements of global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2-cyanopyrrole core has relied heavily on transition metal catalysis or hazardous cyanide sources, presenting substantial challenges for industrial scale-up. For instance, early methodologies described by Chatani utilized cobalt complexes to facilitate the reaction between alkynes and trimethylsilyl cyanide.
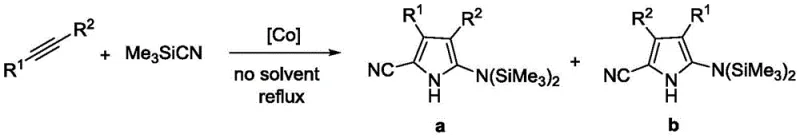
While effective in laboratory settings, cobalt catalysts introduce significant downstream processing burdens, specifically the rigorous removal of trace heavy metals to meet ppm-level specifications required for drug substances. Furthermore, alternative routes reported by Jeffrey employed cuprous cyanide (CuCN) as the cyanide source.
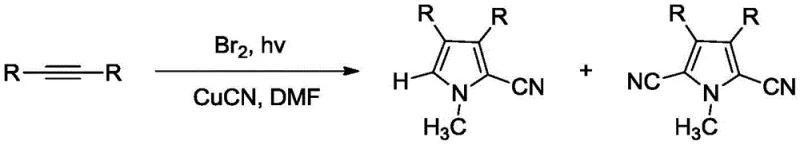
The use of CuCN is increasingly problematic due to its high toxicity and the generation of stoichiometric copper waste, complicating environmental compliance and waste disposal protocols. Additionally, palladium-catalyzed variants often suffer from high catalyst costs and sensitivity to substrate electronics, limiting their versatility across diverse chemical libraries needed for drug discovery campaigns.
The Novel Approach
In stark contrast to these legacy methods, the technology protected under CN110903231B eliminates the need for expensive transition metals entirely. By employing trifluoromethanesulfonic acid as a Brønsted acid catalyst, the reaction proceeds through a distinct mechanistic pathway that activates the imine functionality of the cinnamamine substrate. This organocatalytic strategy allows for the use of tert-butyl isonitrile, a stable and manageable liquid reagent, as the sole source of the cyano group. The reaction conditions are remarkably mild, operating effectively in chlorobenzene at temperatures between 80°C and 90°C. This thermal profile is significantly more energy-efficient than high-temperature pyrolysis or cryogenic conditions often required for sensitive organometallic intermediates. Moreover, the substrate scope is impressive, tolerating various electronic environments on the aromatic ring, including electron-withdrawing halogens and electron-donating methoxy or tert-butyl groups, thereby providing a versatile platform for generating diverse pharmaceutical intermediates without the need for extensive protecting group manipulations.
Mechanistic Insights into TfOH-Catalyzed Cyclization
The core innovation of this synthesis lies in the efficient activation of the N-sulfonyl cinnamamine by the strong acid catalyst. Trifluoromethanesulfonic acid protonates the imine nitrogen, increasing the electrophilicity of the adjacent carbon and facilitating the nucleophilic attack by the isonitrile carbon. This initiates a cascade that ultimately forms the five-membered pyrrole ring with the cyano group positioned at the 2-position. The presence of 2,3-dichloro-4,5-dicyano-p-benzoquinone (DDQ) serves as a crucial oxidant, driving the aromatization of the intermediate dihydropyrrole species to the final stable pyrrole product. This oxidative cyclization is highly selective, minimizing the formation of polymeric byproducts that often plague acid-catalyzed reactions of unsaturated systems.
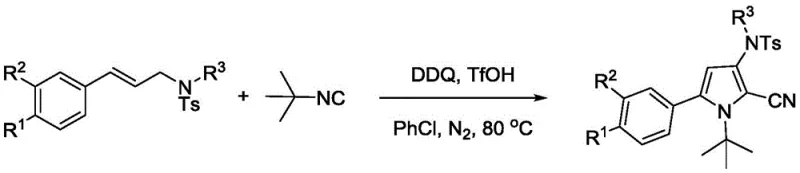
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed routes. The absence of metal-ligand complexes reduces the risk of metal-induced side reactions such as homocoupling or beta-hydride elimination. The primary impurities are typically unreacted starting materials or hydrolysis products, which are easily removed during the aqueous workup and subsequent chromatographic purification. The use of a sulfonyl protecting group on the nitrogen further enhances stability and directs the regioselectivity of the cyclization, ensuring that the cyano group is installed exclusively at the desired position. This high level of regiocontrol is critical for maintaining the biological activity of downstream derivatives, as positional isomers can exhibit vastly different pharmacological profiles.
How to Synthesize 2-Cyanopyrrole Derivatives Efficiently
Implementing this synthesis in a production environment requires strict adherence to the optimized parameters outlined in the patent to ensure consistent quality and yield. The process begins with the precise weighing of reagents under an inert atmosphere to prevent moisture interference, which could deactivate the acid catalyst or hydrolyze the isonitrile. The reaction mixture is then heated to the specified range, and progress is monitored via thin-layer chromatography to determine the exact endpoint, preventing over-reaction or degradation. Following the reaction, a standard extractive workup followed by column chromatography yields the high-purity product suitable for further medicinal chemistry elaboration. For detailed operational parameters and safety guidelines, please refer to the standardized protocol below.
- Under inert atmosphere, mix N-(4-methylbenzenesulfonyl)cinnamamine, tert-butyl isonitrile, trifluoromethanesulfonic acid, and DDQ in chlorobenzene solvent.
- Heat the reaction mixture to 80-90°C and stir until the starting materials are completely consumed, typically monitored by TLC.
- Cool to room temperature, extract with ethyl acetate, wash with brine, dry, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical elegance. The shift from transition metal catalysis to organocatalysis fundamentally alters the cost structure and risk profile of the manufacturing process. By eliminating precious metals like palladium or cobalt, the direct material costs are significantly reduced, and the supply chain becomes less vulnerable to the geopolitical volatility often associated with rare earth and platinum group metal markets. Furthermore, the simplified purification process translates to shorter cycle times and higher throughput in multipurpose manufacturing plants.
- Cost Reduction in Manufacturing: The economic impact of removing heavy metal catalysts cannot be overstated. Traditional methods require expensive scavenging resins or complex recrystallization steps to reduce metal residues to acceptable limits, adding substantial cost per kilogram. This new method bypasses those requirements entirely. Additionally, tert-butyl isonitrile is a commercially available commodity chemical, avoiding the need for custom synthesis of specialized cyanating agents. The use of chlorobenzene, a standard industrial solvent, further ensures that solvent recovery and recycling infrastructure already present in most facilities can be utilized without capital expenditure. These factors combine to deliver a substantially lower cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: Reliability is paramount in API supply chains. The reliance on simple, robust reagents like triflic acid and DDQ ensures that production is not halted by shortages of exotic ligands or catalysts. The reaction demonstrates broad substrate adaptability, meaning a single manufacturing line can produce a wide library of analogues by simply swapping the cinnamamine starting material. This flexibility allows for rapid response to changing clinical demands without the need for lengthy process re-validation associated with changing catalyst systems. The mild reaction conditions also reduce equipment wear and tear, extending the lifespan of reactor vessels and lowering maintenance downtime.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden hazards, but this protocol is inherently safer. The absence of pyrophoric reagents or highly toxic cyanide salts like CuCN simplifies safety protocols and reduces the burden on environmental health and safety (EHS) teams. Waste streams are easier to treat, as they do not contain heavy metal sludge requiring specialized hazardous waste disposal. The moderate temperatures and atmospheric pressure operation facilitate straightforward scale-up from pilot plant to commercial tonnage, ensuring that supply continuity can be maintained as drug candidates progress from Phase I trials to market launch without the need for disruptive process redesigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 2-cyanopyrrole synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and licensees.
Q: What is the cyanide source in this novel synthesis method?
A: The method utilizes tert-butyl isonitrile as the cyano group source, which reacts under trifluoromethanesulfonic acid catalysis to form the 2-cyanopyrrole skeleton.
Q: What are the typical reaction conditions for this process?
A: The reaction proceeds in chlorobenzene solvent at mild temperatures between 80°C and 90°C, utilizing trifluoromethanesulfonic acid as a catalyst and DDQ as an oxidant.
Q: How does this method compare to traditional cobalt-catalyzed routes?
A: Unlike traditional methods requiring expensive transition metal catalysts like cobalt or toxic cuprous cyanide, this protocol uses readily available organic reagents and mild acid catalysis, significantly simplifying purification and reducing heavy metal contamination risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Cyanopyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from gram-scale optimization to full-scale manufacturing. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including residual solvent analysis and trace metal testing, guaranteeing that every batch of 2-cyanopyrrole intermediate meets the highest global standards. We understand the critical nature of timeline adherence in drug development and have optimized our workflows to minimize lead times without compromising on quality or safety.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data for our reference standards and comprehensive route feasibility assessments. Let us help you secure a sustainable and cost-effective supply of high-purity pharmaceutical intermediates for your pipeline.
