Revolutionizing Alpha-Alkoxy-Alpha-Nitrosoacetate Production with Efficient Palladium-Catalyzed Multicomponent Reactions
Revolutionizing Alpha-Alkoxy-Alpha-Nitrosoacetate Production with Efficient Palladium-Catalyzed Multicomponent Reactions
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for more efficient, atom-economical, and scalable processes. A significant breakthrough in this domain is documented in Chinese Patent CN108658812B, which discloses a novel method for synthesizing alpha-alkoxy-alpha-nitrosoacetate derivatives. These compounds are critical building blocks in organic synthesis, particularly valued for their potential applications in developing antitumor agents, including treatments for breast cancer, liver cancer, and lymphoma. The patent outlines a sophisticated yet operationally simple one-step reaction that couples diazo compounds, benzyl alcohols, and nitrosobenzenes. By leveraging allyl palladium chloride as a catalyst and incorporating molecular sieves as an additive, this methodology achieves high selectivity and yield under mild conditions. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this technology represents a paradigm shift towards greener and more cost-effective manufacturing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the alpha-alkoxy-alpha-nitrosoacetate skeleton has been fraught with challenges that hindered its widespread adoption in commercial settings. Traditional synthetic routes often suffered from low substrate activity and a distinct lack of effective catalysts capable of controlling selectivity. This resulted in processes that were not only costly due to low yields but also operationally繁琐 (complicated), requiring multiple steps and harsh reaction conditions. The difficulty in controlling regioselectivity often led to complex impurity profiles, necessitating extensive and expensive purification protocols. For supply chain heads, these inefficiencies translated into longer lead times and higher risks of batch-to-batch variability. Furthermore, the reliance on stoichiometric reagents rather than catalytic systems contributed to poor atom economy, generating substantial chemical waste that complicated environmental compliance and disposal logistics.
The Novel Approach
In stark contrast, the methodology presented in CN108658812B introduces a highly efficient three-component reaction that addresses these historical pain points directly. By utilizing a palladium-catalyzed system, the invention enables the rapid assembly of the target molecule from relatively simple and readily available starting materials. The reaction proceeds at room temperature, typically between -10°C and 40°C, eliminating the need for energy-intensive heating or cryogenic cooling. The use of 4A molecular sieves as a water-absorbing agent is a critical innovation that drives the reaction equilibrium forward, ensuring high conversion rates without the need for excessive reagent loads. This approach not only simplifies the operational workflow but also enhances the overall safety profile of the synthesis, making it an attractive option for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Palladium-Catalyzed Three-Component Coupling
The core of this technological advancement lies in the unique mechanistic pathway facilitated by the allyl palladium chloride catalyst. The reaction initiates with the interaction of the diazo compound with the palladium center, leading to the formation of a reactive metal-carbene species. This intermediate subsequently undergoes an insertion reaction with the benzyl alcohol, generating a hydroxyl ylide intermediate in situ. Unlike traditional methods where such intermediates might decompose or react non-selectively, the presence of the nitrosobenzene component allows for a highly selective capture of this ylide. This cascade process effectively constructs the quaternary carbon center bearing both the alkoxy and nitroso groups in a single operational step. The precision of this catalytic cycle ensures that the desired alpha-alkoxy-alpha-nitrosoacetate framework is formed with high regioselectivity, minimizing the formation of structural isomers that often plague multicomponent reactions.
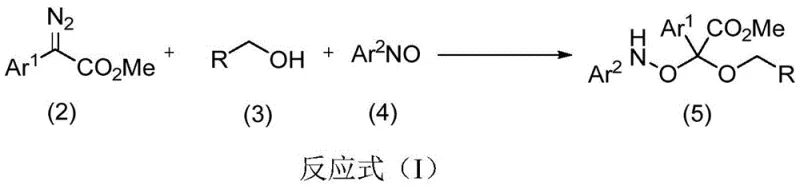
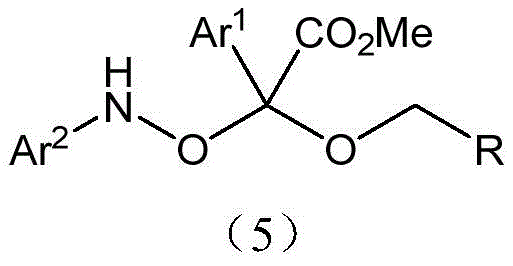
Furthermore, the impurity control mechanism inherent in this design is robust. The use of molecular sieves plays a dual role: physically removing water generated during the process and preventing the hydrolysis of sensitive intermediates. This is crucial for maintaining the integrity of the nitroso functionality, which can be susceptible to degradation under moist conditions. The low catalyst loading, specified at a molar ratio of 0.05 relative to the substrates, indicates a highly active catalytic turnover, reducing the residual metal burden in the final product. For quality control teams, this translates to a cleaner crude product profile, easing the burden on downstream purification units. The ability to tune the electronic properties of the aryl groups (Ar1, Ar2) and the R group allows for the synthesis of a diverse library of derivatives, providing medicinal chemists with a versatile toolkit for structure-activity relationship (SAR) studies without compromising on process efficiency.
How to Synthesize Alpha-Alkoxy-Alpha-Nitrosoacetate Efficiently
Implementing this synthesis route requires careful attention to the order of addition and the quality of reagents to maximize yield and purity. The process begins by dissolving nitrosobenzene, benzyl alcohol, the palladium catalyst, and the molecular sieves in a suitable organic solvent, preferably a chlorinated alkane like dichloromethane. This mixture forms the base into which the diazo compound solution is introduced. The controlled dropwise addition of the diazo compound over a period of one hour is critical to manage the exotherm and maintain the concentration of the reactive carbene species at an optimal level. Following the addition, a brief stirring period of approximately ten minutes ensures complete consumption of the starting materials. The detailed standardized synthesis steps, including specific molar ratios and purification techniques, are outlined below for technical reference.
- Prepare a mixed solution of nitrosobenzene, benzyl alcohol, allyl palladium chloride catalyst, and 4A molecular sieves in an organic solvent such as dichloromethane.
- Dissolve the diazo compound separately in the same organic solvent and add it dropwise to the reaction mixture over a period of one hour at room temperature.
- Stir the reaction for an additional ten minutes after addition, then purify the crude product via column chromatography using ethyl acetate and petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this patented synthesis method offers tangible strategic benefits beyond mere technical novelty. The shift from multi-step, low-yield processes to a streamlined one-step catalytic reaction fundamentally alters the cost structure of producing these high-value intermediates. By reducing the number of unit operations, manufacturers can significantly lower labor costs, energy consumption, and solvent usage. The simplicity of the workup procedure, which often involves standard column chromatography or recrystallization, further reduces the time required for batch release. This efficiency gain allows for a more responsive supply chain, capable of adapting quickly to fluctuating market demands for antitumor drug precursors. Additionally, the use of commercially available starting materials mitigates the risk of raw material shortages, ensuring a stable and continuous supply for downstream API production.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the elimination of expensive transition metal removal steps and the reduction of solvent waste. Since the catalyst loading is minimal (0.05 equivalents) and the reaction achieves high atom economy, the overall material cost per kilogram of product is drastically reduced. The avoidance of harsh reagents and extreme temperatures also lowers the capital expenditure required for specialized reactor equipment, allowing for production in standard glass-lined or stainless steel vessels. These factors combine to create a leaner manufacturing process that delivers substantial cost savings without compromising on the quality or purity specifications required for pharmaceutical applications.
- Enhanced Supply Chain Reliability: Reliability in the supply of complex intermediates is often compromised by lengthy synthesis times and fragile process windows. This palladium-catalyzed method operates at room temperature with a short reaction time of 0 to 1 hour, significantly compressing the production cycle. The robustness of the reaction against varying electronic substituents on the aromatic rings means that a single platform technology can be used to produce a wide range of derivatives. This flexibility reduces the need for multiple dedicated production lines, simplifying inventory management and reducing the risk of bottlenecks. For global supply chains, this translates to shorter lead times and a higher degree of certainty in meeting delivery schedules for critical oncology drug ingredients.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden complexities, but this method is inherently designed for scalability. The exothermic nature of the diazo decomposition is managed through controlled addition, a technique that scales linearly from grams to tons. Furthermore, the high selectivity of the reaction minimizes the generation of hazardous by-products, aligning with modern green chemistry principles. The reduced solvent intensity and the ability to recycle molecular sieves contribute to a lower environmental footprint. For companies facing increasing regulatory pressure regarding waste disposal and emissions, adopting this cleaner synthesis route provides a competitive advantage in maintaining environmental compliance while expanding production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific beneficial effects and background technical details provided in the patent documentation, aiming to clarify the operational feasibility and strategic value for potential partners. Understanding these nuances is essential for R&D teams evaluating the technology for integration into their existing pipelines and for procurement officers assessing the long-term viability of the supply source.
Q: What are the primary advantages of this palladium-catalyzed method over traditional synthesis routes?
A: This method offers superior atom economy and high regioselectivity by utilizing a one-step three-component reaction. It eliminates the need for harsh conditions and complex multi-step sequences, significantly simplifying the operational workflow while maintaining high yields.
Q: What specific catalyst and additives are required for optimal performance?
A: The process utilizes allyl palladium chloride as the catalyst at a low loading ratio of 0.05 equivalents. Crucially, 4A molecular sieves are employed as a water-absorbing additive to drive the reaction equilibrium and ensure high purity of the final alpha-alkoxy-alpha-nitrosoacetate derivative.
Q: Can this synthesis be adapted for large-scale pharmaceutical intermediate production?
A: Yes, the reaction operates effectively at room temperature (-10°C to 40°C) with short reaction times (0-1 hour), making it highly suitable for scale-up. The use of common solvents like dichloromethane and straightforward purification via column chromatography supports robust commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Alkoxy-Alpha-Nitrosoacetate Supplier
As the demand for advanced oncology intermediates continues to grow, partnering with a manufacturer that possesses deep technical expertise is crucial for success. NINGBO INNO PHARMCHEM stands at the forefront of this field, leveraging cutting-edge catalytic technologies like the one described in CN108658812B to deliver high-purity pharmaceutical intermediates. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the pre-clinical or commercial stage. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alpha-alkoxy-alpha-nitrosoacetate derivative meets the highest industry standards for safety and efficacy.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how our efficient synthesis routes can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you accelerate your drug development timeline with reliable, high-quality intermediates produced through state-of-the-art palladium-catalyzed chemistry.
