Advanced Synthesis of Methyl 2-Fluoro-4-Bromo-6-Methylbenzoate for Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-efficient pathways for complex aromatic intermediates. A recent technological breakthrough documented in patent CN115650851A introduces a superior preparation method for Methyl 2-fluoro-4-bromo-6-methylbenzoate, a critical building block in the synthesis of advanced therapeutic agents. This innovation addresses long-standing inefficiencies in traditional manufacturing by shifting the synthetic starting point from expensive carboxylic acids to readily available aniline derivatives. For R&D Directors and Procurement Managers, this represents a significant opportunity to optimize bill-of-materials costs while maintaining rigorous quality standards. The protocol leverages a sequence of well-understood yet precisely optimized reactions, including regioselective bromination, diazotization, and Grignard carboxylation, to achieve high purity and yield without relying on hazardous cyanide chemistry.
The strategic value of this patent lies not only in its chemical elegance but also in its industrial applicability. By utilizing 2-fluoro-6-methylaniline as the foundational feedstock, the process bypasses the premium pricing associated with pre-functionalized benzoic acid starting materials. This shift fundamentally alters the cost structure of the supply chain, offering a competitive edge for manufacturers aiming to reduce production expenses. Furthermore, the elimination of toxic cyanide reagents simplifies environmental compliance and waste management protocols, aligning with modern green chemistry initiatives. As global demand for fluorinated intermediates continues to rise, adopting such optimized synthetic routes becomes essential for maintaining supply continuity and market responsiveness.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
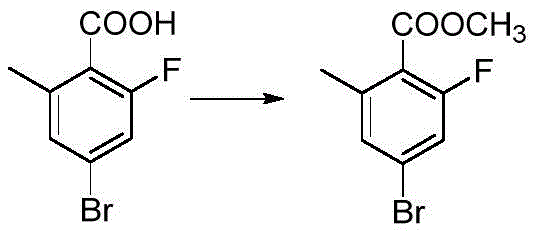
Historically, the synthesis of Methyl 2-fluoro-4-bromo-6-methylbenzoate has relied on routes that commence with 2-fluoro-4-bromo-6-methylbenzoic acid, as disclosed in prior art such as WO2021/7477. While chemically feasible, this approach suffers from severe economic drawbacks due to the exorbitant cost of the starting carboxylic acid. Sourcing this specific halogenated acid often involves complex multi-step preparations or limited supplier availability, creating bottlenecks in the supply chain and inflating the final unit price of the intermediate. Additionally, alternative pathways that attempt to build the aromatic ring from scratch or utilize cyanation strategies introduce significant safety hazards. The use of sodium cyanide or cuprous cyanide necessitates specialized containment infrastructure, rigorous personnel training, and costly waste treatment procedures to neutralize toxic effluents, all of which erode profit margins and increase operational risk.
The Novel Approach
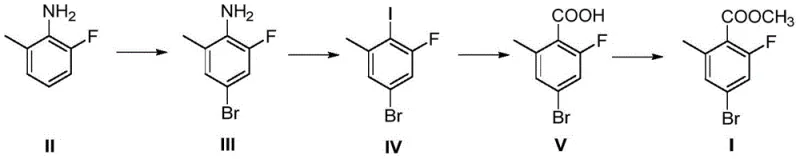
The methodology outlined in CN115650851A circumvents these challenges by initiating the synthesis with 2-fluoro-6-methylaniline, a commodity chemical that is abundant and economically priced. This strategic pivot allows for the late-stage introduction of the carboxyl group via Grignard carboxylation, a transformation that is both atom-economical and scalable. The process flow is streamlined into four distinct operational units, each optimized for maximum conversion and minimal byproduct formation. By avoiding the direct purchase of the expensive acid precursor, manufacturers can achieve substantial cost reductions in pharmaceutical intermediate manufacturing. Moreover, the final esterification step proceeds with exceptional efficiency, often reaching near-quantitative yields, ensuring that the material throughput is maximized throughout the production campaign.
Mechanistic Insights into Sandmeyer Reaction and Grignard Carboxylation
The core of this synthetic innovation relies on the precise execution of a Sandmeyer reaction to install the iodine functionality, which subsequently serves as the handle for Grignard reagent formation. In the second step, the bromo-aniline intermediate is converted into a diazonium salt using sodium nitrite under acidic conditions. This reactive species is then treated with potassium iodide and copper powder to effect the substitution. The choice of iodine over direct carboxylation at this stage is critical; the carbon-iodine bond is sufficiently reactive to facilitate magnesium insertion under mild conditions, whereas the carbon-bromine bond might require more forcing conditions that could compromise the integrity of other functional groups. This mechanistic nuance ensures high selectivity and prevents the formation of homocoupling byproducts that often plague transition metal-catalyzed cross-couplings.
Following the formation of the aryl iodide, the generation of the Grignard reagent is conducted in ethereal solvents such as tetrahydrofuran (THF) or diethyl ether. The subsequent exposure to carbon dioxide gas allows for the nucleophilic attack of the organomagnesium species onto the electrophilic carbon of CO2, forming the carboxylate salt which is then acidified to yield the benzoic acid derivative. This carboxylation strategy is particularly advantageous for impurity control, as gaseous CO2 is a clean reagent that does not introduce heavy metal contaminants or complex organic residues. The final esterification is catalyzed by concentrated sulfuric acid in methanol, driving the equilibrium towards the ester product through the removal of water. This sequence guarantees a high-purity profile, with gas chromatography data indicating purity levels exceeding 98%, meeting the stringent requirements of regulatory bodies for pharmaceutical applications.
How to Synthesize Methyl 2-Fluoro-4-Bromo-6-Methylbenzoate Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly temperature control during the diazotization and Grignard formation steps. The protocol dictates specific molar ratios and solvent choices to ensure reproducibility at scale. For instance, the bromination step utilizes N-bromosuccinimide (NBS) in DMF at controlled temperatures to prevent poly-bromination, while the Grignard step requires anhydrous conditions to prevent reagent quenching. The detailed standardized operating procedures, including exact addition rates, quenching protocols, and crystallization conditions, are essential for technology transfer and process validation. The following guide outlines the critical operational milestones required to replicate the high yields reported in the patent data.
- Bromination of 2-fluoro-6-methylaniline using N-bromosuccinimide (NBS) in DMF to form the bromo-aniline intermediate.
- Diazotization followed by Sandmeyer reaction with potassium iodide and copper powder to install the iodine substituent.
- Formation of Grignard reagent using magnesium, followed by carboxylation with carbon dioxide to yield the benzoic acid derivative.
- Acid-catalyzed esterification using methanol and concentrated sulfuric acid to produce the final methyl ester target.
Commercial Advantages for Procurement and Supply Chain Teams
For Procurement Managers and Supply Chain Heads, the adoption of this patented route offers tangible benefits that extend beyond simple chemistry. The primary advantage is the drastic reduction in raw material expenditure. By substituting a high-value specialty acid with a low-cost commodity aniline, the variable cost per kilogram of the final product is significantly lowered. This cost structure provides greater flexibility in pricing negotiations with downstream API manufacturers and enhances the overall competitiveness of the supply chain. Furthermore, the reliance on standard, non-proprietary reagents such as magnesium, iodine, and methanol ensures that sourcing risks are minimized, as these materials are available from multiple global vendors, reducing the likelihood of supply disruptions due to single-source dependencies.
- Cost Reduction in Manufacturing: The elimination of expensive starting materials directly impacts the bottom line. Since the process avoids the use of pre-functionalized benzoic acids, the procurement budget can be reallocated to other critical areas. Additionally, the high yield of the final esterification step minimizes material loss, ensuring that every gram of input translates effectively into saleable output. The avoidance of cyanide reagents also removes the substantial costs associated with hazardous waste disposal and regulatory compliance monitoring, further contributing to overall cost efficiency without compromising on safety standards.
- Enhanced Supply Chain Reliability: Utilizing 2-fluoro-6-methylaniline as the starting material leverages a robust global supply network. Unlike niche intermediates that may suffer from production outages or geopolitical supply constraints, commodity anilines are produced in large volumes by major chemical manufacturers. This abundance ensures consistent availability and stable pricing, allowing for long-term supply agreements and reliable production planning. The simplified reagent list also reduces the complexity of inventory management, streamlining the logistics of raw material intake and storage.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, utilizing reaction conditions that are easily managed in standard stainless steel reactors. The absence of toxic cyanides simplifies the environmental permitting process and reduces the liability associated with chemical handling. Waste streams are easier to treat, and the use of common solvents like methanol and THF facilitates recycling and recovery programs. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on yield expectations, safety profiles, and scalability potential. Understanding these details is crucial for making informed decisions about process adoption and supplier qualification.
Q: Why is this synthesis route more cost-effective than conventional methods?
A: Conventional methods often start from expensive 2-fluoro-4-bromo-6-methylbenzoic acid. This patented route utilizes 2-fluoro-6-methylaniline, a significantly cheaper commodity chemical, reducing raw material costs substantially.
Q: Does this process avoid the use of highly toxic cyanide reagents?
A: Yes. Unlike alternative routes that may require sodium cyanide or cuprous cyanide for nitrile formation, this method employs a Sandmeyer reaction with iodide, eliminating the handling risks and waste disposal costs associated with cyanides.
Q: What is the overall yield and purity profile of this method?
A: The patent data indicates a total yield of approximately 71%, with individual step yields ranging from 83% to 100%. The final product purity exceeds 98% as confirmed by gas chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 2-Fluoro-4-Bromo-6-Methylbenzoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes in the modern pharmaceutical landscape. Our team of expert chemists has thoroughly analyzed the methodology presented in CN115650851A and is fully equipped to execute this process at commercial scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are backed by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of Methyl 2-fluoro-4-bromo-6-methylbenzoate meets the highest industry standards for impurity profiles and physical properties.
We invite you to collaborate with us to optimize your supply chain for this key intermediate. By leveraging our technical expertise and manufacturing capabilities, you can secure a stable source of high-quality material while achieving significant cost efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patented technology can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
