Advanced Aryl Trifluoromethoxylation Technology for Commercial Scale Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries are constantly seeking efficient pathways to introduce fluorine-containing functional groups, particularly the trifluoromethoxy group, due to its unique ability to enhance metabolic stability and lipophilicity in bioactive molecules. Patent CN108530240B presents a groundbreaking methodology for the synthesis of aryl trifluoromethoxy compounds directly from arylamine precursors. This technology addresses a critical gap in organofluorine chemistry by enabling a mild, one-pot transformation that bypasses the need for complex pre-functionalized substrates. By leveraging aryl diazonium salts as key intermediates, this process offers a streamlined route that is both operationally simple and chemically robust. For global procurement teams and R&D directors, this represents a significant opportunity to optimize the supply chain for high-value fluorinated intermediates. The method described herein not only improves yield but also simplifies the purification process, making it an attractive candidate for large-scale manufacturing of complex pharmaceutical building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a trifluoromethoxy group onto an aromatic ring has been a challenging synthetic task, often requiring harsh reaction conditions or expensive, unstable reagents. Traditional approaches, such as those utilizing arylboronic acids or aryltin compounds, necessitate the prior synthesis of these specific organometallic precursors, adding multiple steps to the overall process and increasing waste generation. Furthermore, earlier methodologies reported by research groups often relied on strong oxidants, photocatalysts, or extreme temperatures to drive the reaction, which poses significant safety risks and operational difficulties in an industrial setting. The instability of certain trifluoromethoxy sources under these aggressive conditions frequently leads to decomposition and low product purity, complicating downstream processing. These limitations have historically constrained the widespread adoption of trifluoromethoxylated motifs in drug discovery pipelines, driving up costs and extending lead times for new candidate molecules.
The Novel Approach
In stark contrast, the technology disclosed in patent CN108530240B utilizes a direct dediazoniation strategy that transforms readily available arylamines into valuable trifluoromethoxy products with remarkable efficiency. By employing silver trifluoromethoxide as the trifluoromethoxy source in an acetonitrile solvent, the reaction proceeds under mild thermal conditions, typically ranging from -40°C to 35°C. This approach eliminates the need for pre-functionalized boronic or stannane substrates, effectively shortening the synthetic route and reducing raw material costs. The one-pot nature of the reaction minimizes handling of unstable intermediates and reduces solvent consumption, aligning perfectly with green chemistry principles. As illustrated in the reaction scheme below, the conversion is direct and high-yielding, particularly when electron-withdrawing groups are present on the aromatic ring.
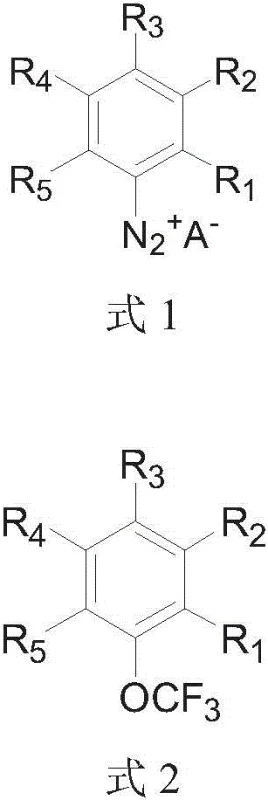
Mechanistic Insights into Silver-Mediated Dediazoniation Trifluoromethoxylation
The core of this innovative process lies in the generation and subsequent reaction of the aryl diazonium salt intermediate. The mechanism involves the initial formation of the diazonium species from the parent arylamine, which serves as an excellent leaving group for nucleophilic substitution. Unlike traditional nucleophilic aromatic substitutions that require strong electron-withdrawing groups ortho or para to the leaving group, this silver-mediated pathway is facilitated by the unique reactivity of the silver trifluoromethoxide complex. The silver center likely coordinates with the diazonium nitrogen, promoting the release of nitrogen gas and generating a highly reactive aryl-silver intermediate or a radical species that rapidly captures the trifluoromethoxy group. This mechanistic pathway allows for the functionalization of a broad range of substrates, including those with neutral or moderately electron-donating substituents, which were previously difficult to trifluoromethoxylate efficiently.
Impurity control is a critical aspect of this chemistry, particularly given the potential instability of diazonium salts at elevated temperatures. The patent specifies a precise temperature profile, initiating the reaction at cryogenic temperatures (-40°C to -20°C) to ensure the stable formation of the initial complex before allowing a controlled warm-up to room temperature. This thermal management strategy suppresses side reactions such as hydrolysis to phenols or homocoupling, which are common pitfalls in diazonium chemistry. Furthermore, the choice of the tetrafluoroborate anion for the diazonium salt significantly enhances stability and solubility in organic solvents like acetonitrile, leading to cleaner reaction profiles and higher isolated yields. This level of control over the reaction environment ensures that the final product meets the stringent purity specifications required for pharmaceutical applications.
How to Synthesize Aryl Trifluoromethoxy Compounds Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing high-purity aryl trifluoromethoxy compounds suitable for commercial use. The process begins with the preparation of the aryl diazonium tetrafluoroborate salt from the corresponding arylamine, a standard procedure that can be easily scaled. Following isolation or in-situ generation, the diazonium salt is reacted with silver trifluoromethoxide in dry acetonitrile under an inert atmosphere. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the aryl diazonium salt by reacting the corresponding arylamine with tetrafluoroboric acid and an nitrite source at low temperature.
- Mix the isolated aryl diazonium salt with silver trifluoromethoxide in acetonitrile solvent at temperatures between -40°C and -20°C.
- Allow the reaction mixture to naturally warm to room temperature (0-35°C) and stir for 5 to 17 hours to complete the trifluoromethoxylation.
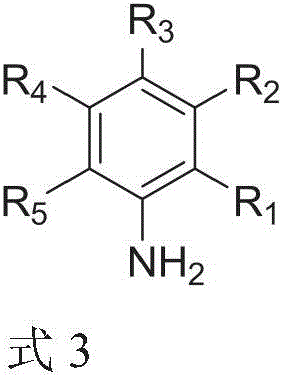
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this trifluoromethoxylation technology offers substantial strategic benefits beyond mere chemical efficiency. The primary advantage lies in the significant cost reduction in pharmaceutical intermediates manufacturing achieved by shortening the synthetic sequence. By starting from cheap and abundant arylamines rather than expensive organometallic reagents, companies can drastically lower their raw material expenditure. Additionally, the elimination of transition metal catalysts that require complex removal steps simplifies the purification process, reducing the burden on quality control labs and accelerating batch release times. The mild reaction conditions also translate to lower energy consumption and reduced wear on reactor equipment, contributing to a more sustainable and cost-effective production model.
- Cost Reduction in Manufacturing: The process utilizes silver trifluoromethoxide, which, while a specialized reagent, is used in stoichiometric amounts that are economically viable given the high value of the final product. More importantly, the avoidance of multi-step precursor synthesis and the high atom economy of the one-pot reaction lead to substantial cost savings. The simplified workup procedure, involving standard extraction and chromatography, further reduces operational expenses associated with solvent recovery and waste disposal. This economic efficiency makes the technology highly competitive for the production of bulk fluorinated intermediates.
- Enhanced Supply Chain Reliability: Relying on arylamines as starting materials ensures a robust and stable supply chain, as these compounds are commodity chemicals produced by numerous global suppliers. This diversification of sourcing options mitigates the risk of supply disruptions that often plague specialized organometallic reagents. Furthermore, the stability of the diazonium tetrafluoroborate salts allows for potential storage or transport if necessary, adding flexibility to production scheduling. This reliability is crucial for maintaining continuous manufacturing operations and meeting tight delivery deadlines for downstream API synthesis.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable, operating at near-ambient pressures and moderate temperatures that are compatible with standard glass-lined or stainless steel reactors. The use of acetonitrile, a common industrial solvent, facilitates easy integration into existing manufacturing infrastructure without the need for specialized containment systems required for highly toxic or volatile solvents. Moreover, the high selectivity of the reaction minimizes the formation of hazardous byproducts, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethoxylation technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a reliable basis for feasibility assessments. Understanding these nuances is essential for R&D teams planning to integrate this chemistry into their process development workflows.
Q: What are the advantages of using aryl diazonium salts for trifluoromethoxylation compared to boronic acids?
A: Using aryl diazonium salts allows for a direct one-pot reaction from readily available arylamines, avoiding the multi-step synthesis required for boronic acid precursors and eliminating the need for harsh oxidants or photocatalysts.
Q: How does the choice of counter-ion affect the reaction yield?
A: The patent data indicates that tetrafluoroborate anions significantly improve product yield compared to chloride or hydrogen sulfate anions, likely due to better stability and solubility profiles of the diazonium intermediate.
Q: Is this process scalable for industrial production of fluorinated intermediates?
A: Yes, the process utilizes mild temperatures (-40°C to 35°C) and common solvents like acetonitrile, making it highly suitable for commercial scale-up without requiring specialized high-pressure or high-temperature equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Trifluoromethoxy Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced fluorination technologies like the one described in patent CN108530240B for the next generation of pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. We are committed to delivering high-purity aryl trifluoromethoxy compounds that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in handling sensitive fluorinated intermediates guarantees consistent quality and supply continuity for your critical projects.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your specific application needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your target molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you accelerate your development timeline and optimize your manufacturing costs effectively.
