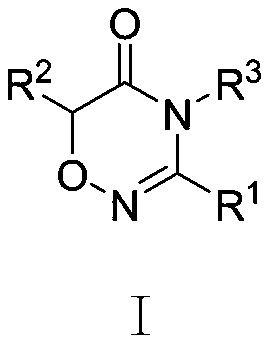
Scalable Metal-Free Synthesis of 4H-1,2,4-Oxadiazine-5(6H)-one Derivatives for Global Supply Chains
The chemical landscape for heterocyclic intermediates is constantly evolving, driven by the need for greener and more cost-effective manufacturing processes. Patent CN107629020B introduces a significant breakthrough in the synthesis of 4H-1,2,4-oxadiazine-5(6H)-one derivatives, a core scaffold increasingly vital for next-generation agrochemicals and pharmaceutical agents. This technology leverages a novel dipole-dipolar cycloaddition strategy that bypasses the limitations of traditional catalytic systems, offering a robust pathway for industrial scale-up. By utilizing substituted alpha-halogenated amides and chlorooxime compounds as key building blocks, the method achieves high regioselectivity and yield without relying on scarce or toxic transition metals. For global supply chain leaders, this represents a pivotal shift towards more sustainable and economically viable production of complex heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the oxadiazinone ring system has relied on sophisticated and often costly catalytic systems that pose significant challenges for large-scale manufacturing. Prior art, such as the work by Zhu Hongjun's group, utilized NIS and BF3·Et2O to promote cyclization, which introduces expensive Lewis acids and iodine sources that complicate downstream purification. Similarly, methods reported by Zhao's group involving elemental iodine or Loiseleur's use of DAST (diethylaminosulfur trifluoride) require hazardous reagents that demand specialized handling and waste disposal protocols. These conventional routes often suffer from narrow substrate scope, meaning they fail to accommodate diverse functional groups without significant optimization. Furthermore, the reliance on multi-step transformations, as seen in Humphrey's approach using 2-hydroxyisobutyronitrile, increases the overall process mass intensity and reduces the final throughput. The accumulation of heavy metal residues from catalysts like palladium or copper in other generic heterocycle syntheses also necessitates expensive scavenging steps to meet strict regulatory purity standards.
The Novel Approach
In stark contrast, the methodology disclosed in CN107629020B streamlines the synthesis into a direct, one-pot reaction that dramatically simplifies the operational workflow. By reacting substituted alpha-halogenated amides with substituted chlorooxime compounds in the presence of a simple inorganic or organic base, the process eliminates the need for any transition metal catalysts entirely. This metal-free approach not only reduces the raw material bill of materials but also removes the burden of metal removal validation, which is a critical bottleneck in API intermediate production. The reaction conditions are remarkably mild, operating effectively between 25°C and 100°C, which allows for the use of standard glass-lined or stainless steel reactors without requiring exotic high-pressure equipment. The broad adaptability of this method is evidenced by its tolerance for various substituents, including alkyl, alkoxy, and halophenyl groups, ensuring versatility for different derivative targets. Ultimately, this novel route transforms a complex multi-step challenge into a straightforward, high-yielding transformation suitable for continuous manufacturing environments.
Mechanistic Insights into Base-Catalyzed Dipolar Cycloaddition
The core of this synthetic innovation lies in the efficient generation of a reactive nitrile oxide or similar dipolar species in situ, which subsequently undergoes cycloaddition with the amide functionality. Under the alkaline conditions provided by bases such as sodium carbonate, potassium carbonate, or triethylamine, the chlorooxime compound is activated to form the necessary electrophilic or nucleophilic species required for ring closure. The alpha-halogenated amide serves as a versatile precursor, where the halogen atom facilitates the elimination or substitution events necessary to form the N-O bond of the oxadiazinone ring. This mechanism avoids the high-energy intermediates typically associated with thermal cyclizations, thereby preserving sensitive functional groups on the aromatic rings. The precise control over the molar ratios, specifically maintaining a base-to-amide ratio between 2.0 and 4.0, ensures complete conversion while minimizing side reactions such as hydrolysis of the amide bond. Kinetic studies suggest that the reaction proceeds through a concerted pathway that favors the formation of the 1,2,4-oxadiazine isomer over other potential regioisomers, guaranteeing high structural fidelity.
From an impurity control perspective, the absence of transition metals fundamentally alters the impurity profile of the final product, shifting the focus from metal scavenging to organic byproduct management. Traditional metal-catalyzed routes often generate difficult-to-remove metal-organic complexes that can persist through crystallization steps, whereas this base-mediated process generates primarily inorganic salts like sodium chloride or potassium bromide as byproducts. These inorganic salts are easily removed during the aqueous workup phase described in the patent, where washing with water or saturated salt solutions effectively partitions the organic product from the reaction matrix. The high selectivity of the cyclization minimizes the formation of oligomeric byproducts or open-chain intermediates that could complicate purification. Consequently, the crude product obtained after solvent removal is of sufficient quality that simple column chromatography or recrystallization yields the target compound with high purity. This clean reaction profile is essential for reducing the number of purification cycles, directly impacting the overall cost of goods and production timeline.
How to Synthesize 4H-1,2,4-Oxadiazine-5(6H)-one Efficiently
Implementing this synthesis protocol requires careful attention to reagent stoichiometry and solvent selection to maximize yield and reproducibility across different batches. The process begins by charging a reaction vessel with the substituted alpha-halogenated amide and the substituted chlorooxime compound, ensuring that the molar ratio of the oxime to the amide is maintained between 1.0 and 2.0 to drive the reaction to completion. A suitable organic solvent, such as acetonitrile, tetrahydrofuran, or toluene, is added to dissolve the reactants, creating a homogeneous reaction mixture that facilitates efficient heat and mass transfer. The addition of the base is a critical step, with options ranging from mild carbonates like Na2CO3 and K2CO3 to stronger alkoxides, depending on the electronic nature of the substrates involved. Once the reagents are combined, the mixture is stirred at a controlled temperature between 25°C and 100°C, allowing the cyclization to proceed over a defined period until monitoring indicates full consumption of the starting materials.
- Charge a reaction vessel with substituted alpha-halogenated amide and substituted chlorooxime compound, then add a suitable organic solvent such as acetonitrile or THF.
- Introduce a base such as sodium carbonate or potassium carbonate at a molar ratio of 2.0 to 4.0 relative to the amide substrate.
- Stir the mixture at a temperature between 25°C and 100°C, followed by aqueous workup, extraction, and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis route offers substantial strategic benefits that extend beyond simple chemical efficiency. The elimination of precious metal catalysts removes a significant source of cost volatility and supply risk, as the prices of metals like palladium or rhodium can fluctuate wildly based on geopolitical factors. Furthermore, the use of commodity chemicals such as alpha-haloamides and chlorooximes ensures a stable and diversified supply base, reducing the risk of single-source bottlenecks that can halt production lines. The mild reaction conditions also translate to lower energy consumption and reduced wear on manufacturing equipment, contributing to a lower total cost of ownership for the production facility. By simplifying the purification process and removing the need for specialized metal scavengers, manufacturers can achieve faster batch turnover times and higher overall equipment effectiveness. These factors combine to create a more resilient and cost-competitive supply chain for high-value heterocyclic intermediates.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the complete removal of expensive transition metal catalysts and ligands, which often account for a disproportionate share of raw material costs in fine chemical synthesis. Without the need for these costly inputs, the direct material cost per kilogram of the final product is significantly lowered, improving margin potential for downstream applications. Additionally, the simplified workup procedure eliminates the expense of purchasing and disposing of metal scavenging resins, which are both costly to buy and expensive to incinerate as hazardous waste. The high yields reported in the patent examples, often exceeding 80% for various substrates, mean that less raw material is wasted, further enhancing the economic efficiency of the process. This cumulative effect results in a drastically simplified cost structure that allows for more competitive pricing in the global market.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved because the key starting materials, such as alpha-halogenated amides and chlorooximes, are widely available from multiple chemical suppliers globally. Unlike specialized catalysts that may have long lead times or limited production capacity, these commodity reagents can be procured in bulk quantities with short delivery windows. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the rate of batch failures and rejections. This stability allows supply chain planners to maintain leaner inventory levels while still ensuring continuity of supply for critical customers. The ability to switch between different bases or solvents without compromising the reaction outcome provides additional flexibility to navigate regional supply disruptions.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of exothermic hazards associated with strong oxidizers or pyrophoric catalysts. The reaction can be safely conducted in standard reactors at atmospheric pressure, minimizing the need for capital-intensive high-pressure infrastructure. From an environmental perspective, the metal-free nature of the synthesis significantly reduces the heavy metal load in wastewater streams, simplifying compliance with stringent environmental regulations like REACH or TSCA. The use of common organic solvents that can be recovered and recycled further enhances the green credentials of the process, aligning with corporate sustainability goals. This combination of safety, scalability, and environmental friendliness makes the technology highly attractive for long-term commercial investment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, based on the detailed disclosures within the patent documentation. Understanding these specifics helps R&D and procurement teams evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the practical realities of scaling this chemistry while maintaining high quality standards.
Q: What are the primary advantages of this synthesis method over traditional routes?
A: This method eliminates the need for expensive transition metal catalysts or harsh reagents like DAST, utilizing cheap bases and readily available alpha-haloamides instead, which significantly reduces raw material costs and simplifies waste treatment.
Q: What is the typical reaction temperature range for this cyclization?
A: The reaction proceeds efficiently under mild thermal conditions ranging from 25°C to 100°C, allowing for flexible energy management and safer operation compared to high-temperature processes.
Q: Does this process generate heavy metal impurities?
A: No, the protocol is entirely metal-free, relying on organic bases and halogenated precursors, which ensures the final product is free from transition metal contamination, a critical factor for pharmaceutical and agrochemical compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4H-1,2,4-Oxadiazine-5(6H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for producing high-quality 4H-1,2,4-oxadiazine-5(6H)-one derivatives. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for agrochemical and pharmaceutical applications. We are committed to leveraging this innovative patent technology to deliver cost-effective solutions that enhance your supply chain resilience.
We invite you to contact our technical procurement team to discuss how we can tailor this synthesis method to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free process for your specific derivative. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that will demonstrate the viability of partnering with us for your next generation of heterocyclic intermediates.
