Scalable Green Synthesis of 2'-Aminobenzothiazole-Arylmethyl-2-Naphthol Derivatives for Pharmaceutical Applications
Scalable Green Synthesis of 2'-Aminobenzothiazole-Arylmethyl-2-Naphthol Derivatives for Pharmaceutical Applications
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally benign pathways for constructing complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN103360339A introduces a transformative approach to synthesizing 2'-aminobenzothiazole-arylmethyl-2-naphthol derivatives, a class of compounds renowned for their broad-spectrum biological activities including antiviral, antibacterial, and anticancer properties. This technology leverages a novel bissulfonate acidic ionic liquid catalyst within an aqueous medium, effectively addressing the long-standing challenges of catalyst recovery, reaction efficiency, and environmental impact associated with traditional synthetic routes. By utilizing water as the sole solvent and a highly active dual-acid catalyst, this method achieves exceptional yields within remarkably short reaction windows, positioning it as a superior candidate for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aminothiazole-naphthol hybrids has relied on catalytic systems that suffer from significant operational inefficiencies and environmental drawbacks. Early methodologies, such as those employing lithium chloride (LiCl) in aqueous media, while conceptually green, were plagued by excessively long reaction times and the requirement for stoichiometric or near-stoichiometric amounts of expensive catalysts, which complicated downstream purification. Furthermore, subsequent advancements using mono-sulfonic acidic ionic liquids under solvent-free conditions, although an improvement, still faced critical hurdles regarding catalyst stability and loss during recycling. These conventional catalysts often exhibited weak acidity, necessitating higher loadings (typically around 10 mol%) and resulting in substantial material loss after just a few cycles, thereby inflating the cost of goods and generating unnecessary chemical waste that burdens the supply chain.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a specialized bissulfonate acidic ionic liquid that fundamentally alters the reaction kinetics and thermodynamics. This novel catalytic system operates efficiently in water, a non-toxic and inexpensive solvent, at moderate temperatures ranging from 80°C to 95°C. The reaction proceeds with remarkable speed, typically completing within a mere 3 to 10 minutes, which drastically enhances throughput capabilities for manufacturing facilities. The general reaction scheme involves the condensation of an aromatic aldehyde, 2-aminobenzothiazole, and β-naphthol in a precise 1:1:1 molar ratio, facilitated by the unique dual-acid functionality of the catalyst.
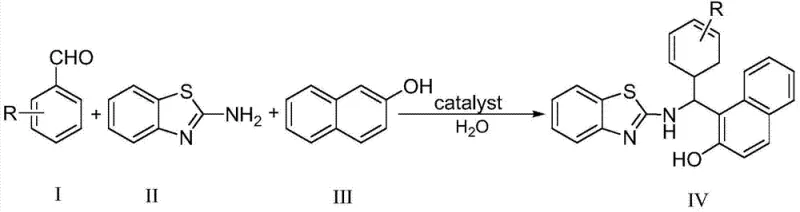
This streamlined process not only simplifies the operational workflow but also ensures high product purity through a straightforward filtration and recrystallization protocol, eliminating the need for complex chromatographic separations often required by older methods.
Mechanistic Insights into Bissulfonate Acidic Ionic Liquid Catalysis
The superior performance of this synthesis route is rooted in the unique structural attributes of the bissulfonate acidic ionic liquid catalyst. Unlike mono-functional acidic ionic liquids, this catalyst features two sulfonic acid (-SO3H) groups attached to the cationic framework, creating a high density of Brønsted acid sites. This bifunctional acidity allows for simultaneous activation of multiple reaction centers, specifically enhancing the electrophilicity of the carbonyl carbon in the aromatic aldehyde. The strong proton-donating capability facilitates the rapid formation of the imine intermediate between the aldehyde and the amine group of the 2-aminobenzothiazole, which is the rate-determining step in many similar condensations.
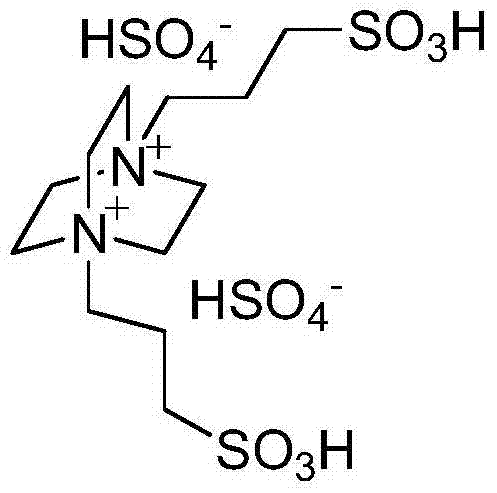
Furthermore, the ionic nature of the catalyst ensures excellent solubility and dispersion within the polar aqueous reaction medium, maximizing the contact area between the catalyst and the organic substrates. This homogeneous-like behavior in a heterogeneous setup promotes efficient mass transfer and minimizes side reactions such as polymerization or oxidation, which are common pitfalls in acid-catalyzed condensations. The robustness of the ionic liquid structure also prevents hydrolysis under the reaction conditions, ensuring that the catalyst remains intact and active throughout the process, which is critical for maintaining consistent impurity profiles in high-purity pharmaceutical intermediates.
How to Synthesize 2'-Aminobenzothiazole-Arylmethyl-2-Naphthol Efficiently
The practical implementation of this green synthesis protocol is designed for ease of adoption in both laboratory and pilot-scale environments. The process requires standard glassware or stainless steel reactors equipped with heating and stirring capabilities, making it accessible for immediate technology transfer. The procedure involves charging the reactor with the three key starting materials—aromatic aldehyde, 2-aminobenzothiazole, and β-naphthol—along with the calculated amount of water and the catalyst. Following a brief heating period where the temperature is maintained between 80°C and 95°C, the reaction mixture is monitored until conversion is complete, indicated by the disappearance of starting materials. The detailed standardized synthesis steps, including specific workup procedures and quality control checkpoints, are outlined below to ensure reproducibility and safety.
- Mix aromatic aldehyde, 2-aminobenzothiazole, and beta-naphthol in a 1: 1:1 molar ratio with water solvent.
- Add 5-8 mol% bissulfonate acidic ionic liquid catalyst and heat to 80-95°C for 3-10 minutes.
- Cool to room temperature, filter the precipitate, and recrystallize from 95% ethanol to obtain pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this patented synthesis method offers compelling strategic advantages that extend beyond simple chemical efficiency. The elimination of volatile organic solvents in favor of water significantly reduces the regulatory burden associated with solvent handling, storage, and disposal, leading to substantial cost savings in waste management and compliance. Additionally, the dramatic reduction in reaction time from hours to minutes translates directly into increased asset utilization, allowing manufacturing plants to produce larger volumes of high-purity intermediates without expanding their physical footprint or capital expenditure on new equipment.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the low catalyst loading and its exceptional recyclability. By requiring only 5 to 8 mol% of the catalyst compared to higher loadings in legacy methods, the direct material cost per kilogram of product is significantly lowered. Moreover, the ability to recycle the catalyst directly from the filtrate without complex regeneration steps minimizes raw material consumption and reduces the frequency of catalyst procurement, creating a more predictable and stable cost structure for long-term production contracts.
- Enhanced Supply Chain Reliability: The reliance on water as a solvent and commercially available starting materials mitigates risks associated with the supply of specialized or hazardous reagents. Water is universally available and inexpensive, removing a potential bottleneck from the supply chain. Furthermore, the robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or mixing, ensuring consistent batch-to-batch quality and reliable delivery schedules for downstream API manufacturers who depend on timely intermediate supply.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with modern green chemistry principles and increasingly stringent global regulations. The absence of toxic organic solvents reduces the risk of fire and exposure hazards for plant personnel. The simplified workup, involving only filtration and recrystallization, generates minimal aqueous waste that is easier to treat than solvent-laden effluent. This environmental compatibility facilitates smoother regulatory approvals and enhances the sustainability profile of the final pharmaceutical products, a key metric for modern ESG-focused supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green catalytic technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters and partners.
Q: What are the advantages of using bissulfonate acidic ionic liquids over traditional catalysts?
A: Unlike traditional Lewis acids or weaker ionic liquids, bissulfonate acidic ionic liquids offer higher acid density and catalytic activity, allowing for significantly reduced reaction times (3-10 minutes) and lower catalyst loading (5-8%), while maintaining excellent recyclability in aqueous media.
Q: Is the catalyst reusable in this synthesis process?
A: Yes, the catalyst demonstrates high stability and can be recycled directly from the filtrate without additional treatment. Experimental data indicates minimal loss in catalytic activity even after multiple cycles, ensuring consistent yield and reducing waste generation.
Q: What represents the primary environmental benefit of this method?
A: The primary environmental benefit is the replacement of volatile organic solvents with water. This eliminates the need for complex solvent recovery systems, reduces VOC emissions, and simplifies the post-reaction workup to a simple filtration and recrystallization step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2'-Aminobenzothiazole-Arylmethyl-2-Naphthol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our technical team has thoroughly evaluated the potential of the bissulfonate ionic liquid catalysis described in CN103360339A and is fully prepared to leverage this technology for the custom manufacturing of high-value heterocyclic intermediates. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2'-aminobenzothiazole-arylmethyl-2-naphthol delivered meets the highest standards required for drug substance synthesis.
We invite forward-thinking R&D and procurement leaders to collaborate with us to optimize their supply chains using this innovative green chemistry approach. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations that demonstrate how we can drive value and reliability into your pharmaceutical intermediate sourcing strategy.
