Advanced Chiral Phosphite Ligands from Methyl Glucoside for Scalable Asymmetric Catalysis
Advanced Chiral Phosphite Ligands from Methyl Glucoside for Scalable Asymmetric Catalysis
The landscape of asymmetric synthesis is constantly evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and fine chemical sectors. A significant breakthrough in this domain is documented in Chinese patent CN102532225A, which discloses a novel class of chiral phosphite ester ligands derived from methyl glucoside. These ligands represent a strategic advancement in ligand design, utilizing a rigid carbohydrate backbone to induce chirality in metal-catalyzed transformations. Specifically, the technology enables the efficient asymmetric 1,4-conjugate addition of organozinc reagents to α,β-unsaturated enones, a critical reaction for constructing chiral carbon centers found in numerous bioactive molecules. The resulting ligand/copper complexes have demonstrated impressive catalytic performance, achieving yields as high as 92% and enantioselectivities up to 85% under optimized conditions. For R&D directors and process chemists seeking robust alternatives to expensive, proprietary ligand systems, this methyl glucoside-derived scaffold offers a compelling balance of synthetic accessibility and catalytic efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric 1,4-conjugate addition of organometallic reagents to enones has relied heavily on sophisticated phosphoramidite or phosphite ligands based on bulky biaryl scaffolds, such as BINOL or TADDOL derivatives. While effective, these conventional ligands often suffer from significant drawbacks regarding their preparation and cost. The synthesis of these traditional ligands frequently involves multi-step sequences requiring harsh reaction conditions, expensive chiral starting materials, and rigorous purification protocols to remove trace metal impurities that could poison downstream catalytic cycles. Furthermore, the steric bulk required for high enantioselectivity in these systems can sometimes limit substrate scope, necessitating the customization of the ligand for every new substrate class. From a supply chain perspective, the reliance on scarce or costly chiral pool precursors for these traditional ligands introduces volatility into the procurement process, potentially leading to extended lead times and inflated costs for the final active pharmaceutical ingredients (APIs).
The Novel Approach
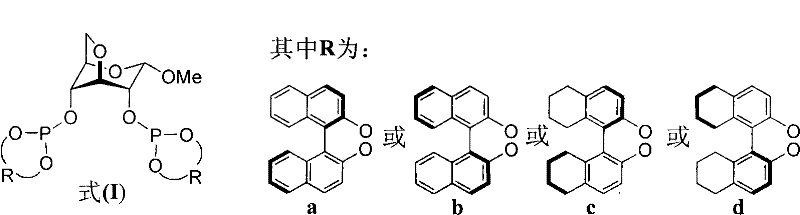
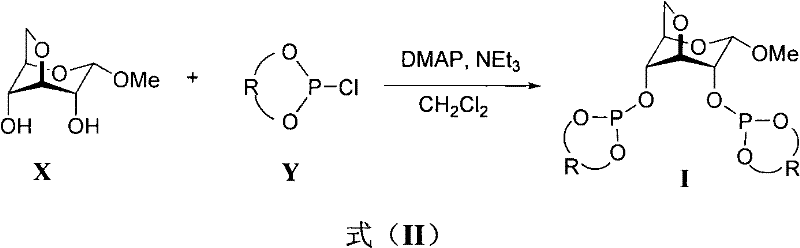
In stark contrast, the methodology presented in patent CN102532225A leverages the abundant and inexpensive chiral pool of carbohydrates, specifically utilizing a 3,6-anhydro-α-D-methyl glucoside skeleton as the chiral backbone. This approach fundamentally simplifies the ligand architecture while maintaining the necessary rigidity to transmit chiral information to the metal center. The synthesis involves a straightforward condensation reaction between the glucoside diol and various chiral chlorophosphites, as illustrated in the reaction scheme below. This modular design allows for the rapid generation of a ligand library by simply varying the 'R' group, which can be derived from binaphthyl or tetrahydronaphthyl motifs. The simplicity of this route not only reduces the number of synthetic steps but also eliminates the need for complex protecting group strategies often seen in carbohydrate chemistry. Consequently, this novel approach provides a cost-effective and scalable pathway to high-performance ligands that can be readily adapted for industrial applications.
Mechanistic Insights into Cu-Catalyzed Asymmetric Conjugate Addition
The efficacy of these methyl glucoside-derived phosphite ligands lies in their ability to form stable, chiral complexes with copper salts, which serve as the active catalytic species. Upon mixing the ligand with a copper source such as Cu(OTf)₂ or (CuOTf)₂·C₆H₆ in solvents like THF or toluene, a coordinatively unsaturated copper-phosphite complex is generated in situ. This complex activates the organozinc reagent, facilitating the transmetallation process that is critical for the conjugate addition mechanism. The rigid bicyclic structure of the anhydro-methyl glucoside core imposes a specific geometric constraint on the copper center, creating a well-defined chiral pocket. This pocket discriminates between the enantiotopic faces of the α,β-unsaturated enone substrate during the nucleophilic attack, thereby controlling the stereochemical outcome of the reaction. The electronic properties of the phosphite moieties, tuned by the aromatic 'R' groups, further modulate the Lewis acidity of the copper center, optimizing the reaction rate and selectivity.
Impurity control is another critical aspect where this ligand system excels. The use of well-defined, crystalline ligands helps minimize the formation of non-selective background reactions that often plague asymmetric catalysis. In traditional systems, free ligands or ill-defined copper aggregates can lead to racemic product formation, complicating downstream purification. However, the strong chelation of the bidentate phosphite ligand to the copper center ensures that the majority of the catalytic activity proceeds through the desired chiral pathway. Moreover, the reaction conditions specified in the patent, such as the use of dry dichloromethane and controlled temperatures ranging from -40°C to 20°C, are designed to suppress side reactions like 1,2-addition or polymerization of the enone. This high level of chemoselectivity translates directly to cleaner reaction profiles, reducing the burden on purification teams and ensuring that the final beta-substituted ketone products meet stringent purity specifications required for pharmaceutical intermediates.
How to Synthesize Chiral Phosphite Ligand Efficiently
The preparation of these high-value ligands is remarkably straightforward, making it accessible for both laboratory-scale optimization and pilot-plant production. The process relies on standard Schlenk techniques to maintain an inert atmosphere, preventing the hydrolysis of the sensitive phosphite ester bonds. By carefully controlling the stoichiometry of the base and the addition rate of the chlorophosphite, manufacturers can maximize the conversion of the starting glucoside material. The following guide outlines the generalized procedure derived from the patent embodiments, serving as a foundational protocol for process development teams aiming to implement this technology.
- Under a nitrogen atmosphere, dissolve chiral 3,6-anhydro-alpha-D-methyl glucoside skeleton and chiral chlorophosphite in dichloromethane solvent.
- Add 4-Dimethylaminopyridine (DMAP) and triethylamine as bases, maintaining the reaction temperature between -15°C and 30°C for 1 to 3 hours.
- Remove solvent under reduced pressure, add toluene to precipitate insolubles, filter, concentrate the filtrate, and purify via rapid column chromatography to obtain the white solid ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this methyl glucoside-based ligand technology presents a multitude of strategic benefits that extend beyond mere technical performance. The primary advantage stems from the drastic simplification of the supply chain for the ligand itself. Unlike complex biaryl ligands that rely on specialized, low-volume suppliers, the core scaffold is derived from methyl glucoside, a commodity chemical produced in massive quantities for the food and pharmaceutical industries. This abundance ensures a stable, long-term supply of the raw material, insulating the manufacturing process from the volatility often associated with niche chiral reagents. Furthermore, the synthetic route to the ligand avoids the use of precious metals or exotic reagents, relying instead on common organic bases and solvents that are easily sourced globally.
- Cost Reduction in Manufacturing: The economic implications of switching to this ligand system are profound. By eliminating the need for expensive, multi-step ligand syntheses, the overall cost of goods sold (COGS) for the chiral intermediate can be significantly lowered. The high atom economy of the condensation reaction and the ability to recover solvents like toluene and dichloromethane further contribute to process efficiency. Additionally, the high yields reported (up to 92%) mean that less starting material is wasted, directly improving the mass balance of the production process. This efficiency allows for a more competitive pricing structure for the final API intermediate, providing a distinct market advantage.
- Enhanced Supply Chain Reliability: Reliability is paramount in the pharmaceutical supply chain, and this technology enhances it by decoupling production from scarce resources. The robustness of the ligand synthesis, which tolerates standard workup procedures like filtration and concentration, ensures consistent batch-to-batch quality. This consistency reduces the risk of production delays caused by out-of-specification reagents. Moreover, the ligand's stability as a white solid facilitates easier storage and transportation compared to oilier or more sensitive liquid ligands, reducing logistical complexities and potential degradation during transit.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process aligns well with modern green chemistry principles. The reaction operates at mild temperatures and does not generate hazardous heavy metal waste streams, as the copper loading is catalytic and can be effectively managed. The use of standard organic solvents allows for established recycling protocols, minimizing the environmental footprint of the manufacturing site. The scalability of the reaction, demonstrated by the straightforward exothermic control and simple phase separations, means that transitioning from gram-scale R&D to ton-scale commercial production can be achieved with minimal process re-engineering, accelerating time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ligand technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of the operational parameters and expected outcomes for potential adopters of this methodology.
Q: What are the primary advantages of this methyl glucoside-derived ligand over traditional phosphoramidites?
A: The ligand offers a simpler synthesis route from cheap, readily available methyl glucoside scaffolds compared to complex binaphthyl-based systems, while still achieving high enantioselectivity (up to 85% ee) and yields (up to 92%) in copper-catalyzed conjugate additions.
Q: Can this catalyst system be scaled for industrial production of beta-substituted ketones?
A: Yes, the protocol utilizes standard organic solvents like dichloromethane and toluene, operates at manageable temperatures (-15°C to 30°C), and uses stable copper salts, making it highly suitable for commercial scale-up in pharmaceutical intermediate manufacturing.
Q: What is the substrate scope for the asymmetric 1,4-conjugate addition using this ligand?
A: The patent demonstrates effectiveness across various cyclic enones including 2-cyclopentenone, 2-cyclohexenone, and 2-cycloheptenone, reacting with organozinc reagents to produce optically active beta-substituted ketones with varying degrees of stereocontrol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phosphite Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in accelerating drug discovery and development. Our team of expert chemists has extensively evaluated the potential of methyl glucoside-derived ligands and is fully equipped to support your projects from early-stage route scouting to commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench to plant is seamless. Our state-of-the-art facilities include rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of ligand or intermediate meets the highest international standards.
We invite you to collaborate with us to leverage this cost-effective and high-performance catalytic system for your next project. Whether you require custom synthesis of specific ligand variants or full-scale production of the resulting chiral ketones, our technical procurement team is ready to assist. Please contact us today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this technology can optimize your supply chain and reduce overall manufacturing costs.
