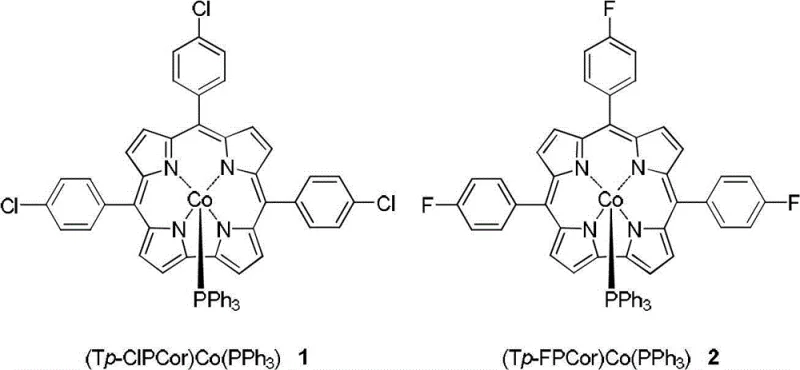
Advanced Synthesis of Halogenated Pentacoordinate Corrole Cobalt Complexes for Catalytic Applications
The chemical landscape for advanced functional materials is constantly evolving, driven by the need for more efficient catalytic systems and stable sensor components. Patent CN102942597A introduces a significant breakthrough in the field of metal-organic macrocycle synthesis, specifically detailing the preparation of novel five-coordinated corrole cobalt coordination compounds containing chlorine or fluorine substituents. Unlike traditional methods that often struggle with complex purification or unstable intermediates, this invention provides a robust pathway to synthesize tris(4-chlorophenyl)corrole cobalt triphenylphosphine and tris(4-fluorophenyl)corrole cobalt triphenylphosphine. These compounds represent a critical class of materials with wide-ranging application prospects in homogeneous catalysis, electrochemical sensors, solar cell technologies, and even pharmaceutical research. By leveraging a direct coordination strategy between metal-free corroles, cobalt acetate, and triphenylphosphine, the patent outlines a process that is not only chemically elegant but also industrially viable, addressing key pain points regarding solvent toxicity and workup difficulty that have historically plagued this sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of metal corrole complexes has heavily relied on methodologies adapted from porphyrin chemistry, which often present substantial operational challenges for large-scale manufacturing. Conventional routes frequently necessitate the use of high-boiling polar aprotic solvents such as N,N-dimethylformamide (DMF), pyridine, or dimethyl sulfoxide (DMSO) to facilitate the insertion of the metal ion into the corrole macrocycle. While effective on a milligram scale in academic settings, these solvents create severe bottlenecks in commercial production due to their high boiling points, which make removal via distillation energy-intensive and time-consuming. Furthermore, residual traces of these solvents can be notoriously difficult to eliminate completely, potentially contaminating the final product and compromising its performance in sensitive applications like organic electronics or biological sensing. Additionally, older template-directed synthesis methods involving single pyrrole cyclization often suffer from low regioselectivity and complex impurity profiles, requiring extensive and costly purification steps that reduce overall process efficiency and yield.
The Novel Approach
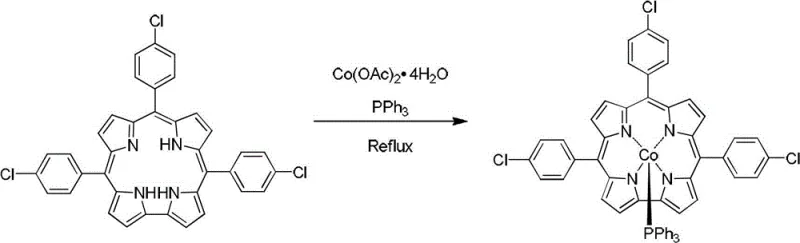
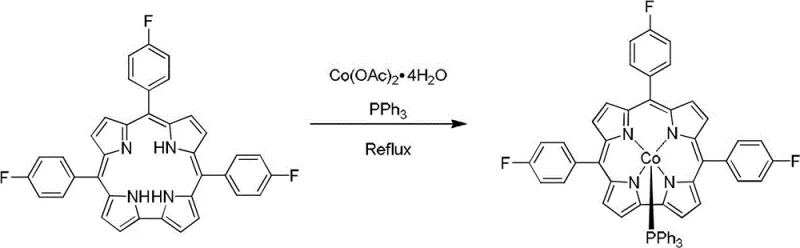
The methodology disclosed in patent CN102942597A offers a transformative alternative by utilizing methanol as the primary reaction medium, a strategic choice that drastically simplifies the entire workflow. In this novel approach, the metal-free corrole precursors are dissolved in methanol along with cobalt acetate tetrahydrate and triphenylphosphine, creating a homogeneous reaction mixture that proceeds smoothly under reflux conditions. The use of methanol, a low-boiling and environmentally friendlier solvent compared to DMF, allows for rapid and energy-efficient removal via rotary evaporation once the reaction is complete. This shift not only reduces the thermal load on the equipment but also minimizes the risk of thermal degradation of the sensitive corrole macrocycle during workup. The reaction is driven to completion by the strong coordination affinity between the cobalt center and the axial triphenylphosphine ligand, resulting in stable pentacoordinate complexes that precipitate or can be easily isolated. This streamlined protocol eliminates the need for harsh reaction conditions and complex extraction procedures, marking a significant step forward in the practical synthesis of functionalized corrole materials.
Mechanistic Insights into Co-Corrole Coordination Chemistry
The core of this synthesis lies in the precise coordination chemistry occurring at the cobalt center within the contracted corrole macrocycle. Unlike porphyrins, corroles possess a smaller central cavity and a formal charge of 3-, which naturally stabilizes higher oxidation states of transition metals, typically Co(III). In this specific reaction mechanism, the cobalt acetate serves as the metal source, dissociating in the methanol medium to provide Co(II) or Co(III) species that insert into the corrole ring. The simultaneous presence of triphenylphosphine (PPh3) is critical; it acts as a strong sigma-donor axial ligand that coordinates to the cobalt ion perpendicular to the macrocyclic plane. This axial coordination saturates the coordination sphere of the cobalt, forming a stable five-coordinate geometry that prevents the formation of mu-oxo dimers, a common decomposition pathway for four-coordinate metallo-corroles in the presence of oxygen. The electron-withdrawing nature of the chloro or fluoro substituents on the meso-phenyl rings further modulates the electronic density of the macrocycle, enhancing the oxidative stability of the complex and tuning its redox potentials for specific electrochemical applications.
From a quality control and impurity management perspective, this reaction system offers a unique and highly advantageous visual feedback mechanism. The starting metal-free corrole materials typically exhibit a deep墨 green (dark green) coloration in methanol solution, characteristic of their free-base electronic structure. As the cobalt insertion and axial ligation proceed, the electronic environment of the macrocycle changes fundamentally, shifting the absorption spectrum and causing the solution to turn a vivid bright red. This distinct chromatic transition serves as an intrinsic process analytical technology (PAT) tool, allowing chemists to monitor reaction progress in real-time without the need for immediate thin-layer chromatography (TLC) or spectroscopic analysis. This capability ensures that the reaction is stopped precisely at the point of maximum conversion, minimizing the formation of side products such as demetalated species or over-oxidized byproducts. Consequently, the crude reaction mixture is cleaner, which directly translates to higher recovery rates during the subsequent silica gel column chromatography purification step, ensuring a final product with high chemical purity suitable for demanding research and industrial uses.
How to Synthesize Tris(4-halophenyl)corrole Cobalt Complexes Efficiently
The synthesis protocol described in the patent is designed for reproducibility and scalability, making it an ideal candidate for technology transfer from the laboratory to pilot plant operations. The process relies on standard chemical engineering unit operations such as dissolution, reflux heating, solvent evaporation, and chromatographic separation, all of which are well-understood and easily implemented in GMP-compliant facilities. The molar ratios are optimized to ensure complete consumption of the valuable corrole precursor, using a slight excess of the cobalt salt and phosphine ligand to drive the equilibrium towards the product. For R&D teams looking to replicate or adapt this chemistry, the key lies in maintaining strict control over the reflux temperature and ensuring thorough mixing to prevent localized concentration gradients that could lead to incomplete metallation. The following guide outlines the standardized operational parameters derived from the patent examples, providing a clear roadmap for producing these high-value coordination compounds.
- Dissolve the metal-free corrole precursor (tris(4-chlorophenyl)corrole or tris(4-fluorophenyl)corrole) in methanol, then add cobalt acetate tetrahydrate and triphenylphosphine in a 1: 5:5 molar ratio.
- Heat the reaction mixture to reflux under magnetic stirring for 1 to 6 hours until the solution color changes from dark green to bright red, indicating complete coordination.
- Evaporate the solvent to dryness and purify the resulting residue via silica gel column chromatography using a dichloromethane and n-hexane mixture to isolate the target red solid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this methanol-based synthesis route presents compelling economic and logistical benefits that extend far beyond simple yield improvements. The primary advantage lies in the drastic simplification of the solvent recovery and waste management infrastructure. By replacing high-boiling, toxic solvents like DMF with methanol, manufacturers can significantly reduce the energy costs associated with solvent distillation and recycling, while also lowering the regulatory burden related to hazardous waste disposal. Methanol is a commodity chemical with a stable global supply chain, ensuring consistent raw material availability and price stability, which is crucial for long-term production planning. Furthermore, the elimination of difficult-to-remove solvents reduces the risk of batch rejection due to residual solvent limits, thereby improving overall production throughput and reducing the cost of quality failures. This process optimization directly contributes to a more resilient and cost-effective supply chain for high-purity corrole derivatives.
- Cost Reduction in Manufacturing: The switch to a methanol-based system fundamentally alters the cost structure of producing these specialized cobalt complexes. Since methanol has a much lower boiling point than traditional solvents like DMF or DMSO, the energy required for solvent removal via rotary evaporation or distillation is substantially lower, leading to direct utility savings. Additionally, the simplified workup procedure reduces the labor hours required for post-reaction processing, as there is no need for extensive aqueous washes or multiple extraction steps often required to remove polar aprotic solvents. The high stability of the pentacoordinate product also minimizes material loss during purification, ensuring that the expensive corrole starting material is converted into saleable product with maximum efficiency. These factors combine to deliver a manufacturing process that is leaner and more economically sustainable without compromising on the quality of the final active ingredient.
- Enhanced Supply Chain Reliability: The reliance on widely available and inexpensive reagents such as cobalt acetate tetrahydrate and triphenylphosphine mitigates the risk of supply disruptions that can occur with exotic or specialized catalysts. Cobalt acetate is a bulk chemical produced at scale for various industries, ensuring a steady flow of raw materials even during market fluctuations. The robustness of the reaction conditions, which tolerate standard reflux temperatures and do not require inert atmosphere gloveboxes for the entire duration, allows for production in standard multipurpose chemical reactors. This flexibility enables manufacturers to schedule production runs more efficiently and respond quickly to changes in demand. Moreover, the stability of the final red solid products ensures they can be stored and transported under ambient conditions without special packaging or temperature control, further simplifying logistics and reducing shipping costs for global distribution.
- Scalability and Environmental Compliance: Scaling this synthesis from gram to kilogram quantities is straightforward due to the absence of exothermic hazards or sensitive intermediates that typically complicate scale-up. The reaction is homogeneous and proceeds cleanly, meaning that heat transfer and mass transfer issues are minimal even in larger vessels. From an environmental perspective, the use of methanol aligns better with green chemistry principles compared to chlorinated or high-boiling amide solvents, facilitating easier compliance with increasingly stringent environmental regulations. The waste stream generated is less toxic and easier to treat, reducing the liability and cost associated with environmental health and safety (EHS) management. This makes the process not only commercially attractive but also socially responsible, appealing to end-users who prioritize sustainability in their sourcing decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these pentacoordinate corrole cobalt complexes. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating these materials into your specific product development pipelines or manufacturing processes. We encourage technical teams to review these points carefully to assess the fit for their specific requirements.
Q: What are the advantages of using methanol over DMF in this synthesis?
A: Using methanol significantly simplifies post-reaction processing compared to high-boiling solvents like N,N-dimethylformamide (DMF) or dimethyl sulfoxide (DMSO). Methanol is easily removed via rotary evaporation, reducing energy consumption and processing time while eliminating the risk of solvent entrapment in the final crystal lattice.
Q: How is the reaction progress monitored without advanced instrumentation?
A: The reaction features a distinct visual indicator where the starting metal-free corrole solution is dark green, while the formed pentacoordinate cobalt complex is bright red. This sharp color transition allows operators to visually track the reaction endpoint effectively, ensuring consistent batch quality without constant spectroscopic monitoring.
Q: Are these complexes stable enough for commercial storage and transport?
A: Yes, the introduction of the axial triphenylphosphine ligand stabilizes the cobalt center, resulting in pentacoordinate complexes that are chemically robust and thermally stable. This stability facilitates easier purification via column chromatography and ensures the material maintains its integrity during long-term storage and global logistics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Corrole Cobalt Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced coordination compounds play in driving innovation across the pharmaceutical, agrochemical, and electronic materials sectors. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the early stages of R&D or full-scale manufacturing. We are committed to delivering high-purity corrole cobalt complexes that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in handling air-sensitive and moisture-sensitive organometallic species guarantees that every batch we deliver maintains the structural integrity and performance characteristics defined in the patent literature, providing you with a reliable foundation for your downstream applications.
We invite you to collaborate with us to optimize your supply chain for these specialized catalysts and intermediates. By partnering with NINGBO INNO PHARMCHEM, you gain access to our deep technical knowledge and flexible manufacturing capabilities. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized synthesis methods can enhance your project's efficiency and profitability. Let us be your trusted partner in bringing high-performance corrole technologies to the market.
