Advanced Altimezole Manufacturing: Eliminating Palladium Catalysts for Cost-Effective Veterinary API Production
The pharmaceutical and veterinary sectors are constantly seeking robust synthetic pathways that balance high purity with economic viability. Patent CN111925330A, published in late 2020, introduces a transformative preparation method for Altimezole, a critical alpha-2 adrenoceptor antagonist used primarily as a veterinary sedative reversal agent. This technology addresses significant bottlenecks in existing manufacturing processes by replacing hazardous and expensive reduction steps with a streamlined Huang Minlon reduction protocol. For R&D directors and supply chain managers, this patent represents a pivotal shift towards safer, more scalable production of high-purity veterinary intermediates. The disclosed method not only simplifies the operational workflow but also drastically reduces the dependency on precious metal catalysts, positioning it as a superior alternative for industrial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
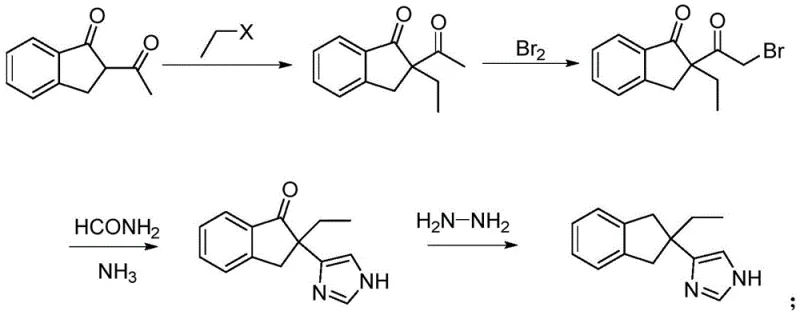
Historically, the synthesis of Altimezole and its analogues has been plagued by complex post-treatment procedures and reliance on unstable reagents. Traditional routes often utilize o-benzyl dibromide and acetylacetone as starting materials, leading to the formation of 2,2-diacetylindane intermediates that are notoriously difficult to purify, resulting in suboptimal yields. Furthermore, earlier methodologies frequently necessitated the use of lithium catalysts for bromination steps, which demand strictly anhydrous and oxygen-free environments due to their extreme reactivity, posing significant safety risks in large-scale reactors. Another prevalent conventional approach involves a two-step reduction process utilizing sodium borohydride followed by palladium-on-carbon hydrogenation under pressure. This not only inflates production costs due to the price of palladium but also introduces safety hazards associated with high-pressure hydrogenation equipment, making the process less attractive for continuous manufacturing environments.
The Novel Approach
The innovative strategy outlined in the patent circumvents these historical challenges through a cleverly designed four-step sequence that prioritizes operational simplicity and safety. By initiating the synthesis with the alkylation of 2-acetyl indanone using haloethane under mild base catalysis, the process avoids the need for cryogenic lithium chemistry. The subsequent bromination is carefully controlled at low temperatures to prevent over-bromination, ensuring high selectivity for the mono-bromo intermediate. Most critically, the final reduction step employs hydrazine hydrate in a Huang Minlon reduction, effectively collapsing the traditional two-step reduction into a single operation. This eliminates the requirement for expensive palladium catalysts and high-pressure hydrogenation vessels, thereby streamlining the workflow and significantly lowering the barrier to entry for commercial production facilities.
Mechanistic Insights into Base-Catalyzed Alkylation and Cyclization
The success of this synthetic route relies heavily on the precise manipulation of electronic effects within the indanone scaffold. In the initial alkylation step, the presence of two adjacent carbonyl groups renders the hydrogen at the 2-position highly acidic and active. Under the influence of a strong base such as potassium tert-butoxide, this proton is readily abstracted to form a stable enolate, which then undergoes nucleophilic substitution with haloethane. This mechanism ensures high conversion rates without generating significant by-products, provided that the reaction is conducted under an inert atmosphere to prevent moisture interference. The choice of solvent, preferably 1,4-dioxane or DMF, plays a crucial role in solubilizing the ionic intermediates while maintaining a reaction temperature between 5°C and 35°C, which is mild enough to prevent thermal degradation yet sufficient to drive the alkylation to completion within 5 to 6 hours.
Impurity control is further refined in the subsequent cyclization step where the alpha-bromo ketone reacts with formamide. The patent highlights a critical mechanistic nuance: the introduction of ammonia gas during this stage. Mechanistically, the ammonia serves to lower the activation energy required for the conversion of the initial oxazole intermediate into the desired imidazole ring. Without the ammonia atmosphere, higher temperatures would be required, leading to the carbonization of organic reactants and the formation of tarry by-products that are difficult to separate. By maintaining a continuous flow of ammonia at 120°C to 130°C, the reaction proceeds rapidly and cleanly. This careful control of the reaction environment ensures that the final Altimezole product achieves a purity exceeding 97%, with the total reaction yield surpassing 60%, a substantial improvement over legacy methods that often struggle to exceed 40-50% overall efficiency due to cumulative losses in multi-step purifications.
How to Synthesize Altimezole Efficiently
The synthesis of Altimezole via this patented route offers a clear pathway for laboratories aiming to produce high-quality veterinary intermediates with minimal environmental impact. The process leverages widely available raw materials such as 2-acetyl indanone and formamide, avoiding the supply chain volatility associated with specialized organometallic reagents. The operational protocol emphasizes strict temperature control during the bromination phase and the utilization of microwave heating in the final reduction step to enhance energy efficiency. For process chemists looking to implement this technology, the detailed standard operating procedures regarding molar ratios, solvent volumes, and workup techniques are essential for replicating the high yields reported in the patent examples. The following guide outlines the critical operational parameters required to achieve optimal results.
- React 2-acetyl indanone with haloethane under base catalysis (e.g., potassium tert-butoxide) at 5-35°C to obtain 2-acetyl-2-ethyl indanone.
- Perform controlled bromination of 2-acetyl-2-ethyl indanone at low temperatures (-10 to 5°C) to generate 2-(2-bromoacetyl)-2-ethyl indanone while minimizing dibromo byproducts.
- Cyclize the bromo-intermediate with formamide in an ammonia atmosphere at 120-130°C to form the imidazole ring structure.
- Execute a one-step Huang Minlon reduction using hydrazine hydrate and base under microwave heating to yield high-purity Altimezole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates directly into enhanced operational resilience and cost efficiency. The removal of palladium-on-carbon from the process flow is a significant strategic advantage, as it decouples production costs from the fluctuating market prices of precious metals. Furthermore, the elimination of high-pressure hydrogenation steps reduces the regulatory burden and insurance costs associated with operating hazardous high-pressure reactors. The simplified workflow, characterized by fewer unit operations and easier post-treatment protocols such as simple extraction and recrystallization, allows for faster batch turnover times. This agility enables manufacturers to respond more swiftly to market demands for veterinary pharmaceuticals, ensuring a steady supply of critical medications like Antisedan without the delays typically caused by complex purification bottlenecks.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally stronger due to the substitution of expensive reagents with commodity chemicals. By replacing the palladium-catalyzed hydrogenation with a hydrazine-based reduction, manufacturers eliminate the capital expenditure associated with recovering and recycling noble metals. Additionally, the high selectivity of the base-catalyzed alkylation minimizes the formation of difficult-to-remove impurities, which reduces the consumption of solvents and adsorbents during the purification phase. This leaner chemical usage profile leads to substantial savings in raw material procurement and waste disposal costs, making the overall cost of goods sold significantly more competitive in the global veterinary API market.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of robust, commercially available starting materials that do not require specialized storage conditions like strict inert atmospheres or cryogenic temperatures. The stability of the intermediate products, particularly the 2-acetyl-2-ethyl indanone, allows for potential stockpiling or campaign production, providing a buffer against upstream supply disruptions. Moreover, the avoidance of lithium catalysts, which are sensitive to transport and storage conditions, simplifies logistics and reduces the risk of batch failures due to reagent degradation. This reliability ensures that downstream formulation partners receive consistent quality intermediates, fostering stronger long-term partnerships between suppliers and pharmaceutical companies.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route offers distinct advantages for large-scale production. The reaction conditions are generally mild, operating at atmospheric pressure and moderate temperatures, which simplifies the engineering requirements for scaling up from pilot plants to multi-ton commercial reactors. The reduction in hazardous waste generation, specifically the absence of heavy metal residues from palladium catalysts, simplifies wastewater treatment and aligns with increasingly stringent environmental regulations. The process generates fewer three wastes (waste water, waste gas, waste residue), facilitating easier compliance with green chemistry standards and reducing the environmental footprint of the manufacturing facility, which is a key metric for modern sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Altimezole synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the practical benefits of adopting this novel route over traditional methods.
Q: Why is the ammonia atmosphere critical in the imidazole formation step?
A: The continuous introduction of ammonia gas during the reaction of the alpha-haloketone with formamide significantly lowers the required reaction temperature for oxazole-to-imidazole conversion. This prevents high-temperature carbonization of reactants, improves the reaction rate, and ultimately enhances the overall yield of the intermediate.
Q: How does this new route improve upon traditional Altimezole synthesis methods?
A: Traditional methods often rely on expensive palladium-on-carbon hydrogenation under pressure or dangerous lithium catalysts requiring absolute anhydrous conditions. This novel route replaces the multi-step reduction with a single-step Huang Minlon reduction using hydrazine hydrate, eliminating the need for precious metal catalysts and high-pressure equipment while simplifying post-treatment.
Q: What represents the primary cost-saving advantage for procurement teams?
A: The primary economic benefit stems from the elimination of palladium carbon, a costly noble metal catalyst, and sodium borohydride. Additionally, the process achieves a total reaction yield exceeding 60% with product purity over 97%, reducing raw material waste and purification costs associated with complex impurity profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Altimezole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the modern veterinary pharmaceutical landscape. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemical transformations like the Huang Minlon reduction are executed with precision and safety. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Altimezole intermediate meets the highest international standards for veterinary drug manufacturing. We are committed to delivering consistent quality and supply continuity for our global partners.
We invite pharmaceutical companies and veterinary drug manufacturers to collaborate with us to leverage this advanced synthesis technology for their supply chains. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you optimize your Altimezole sourcing strategy for maximum efficiency and reliability.
