Advanced Manufacturing of ABT-888 Core Intermediate via Streamlined Acetic Acid Cyclization
Introduction to Patent CN106432195B and Technological Breakthroughs
The pharmaceutical landscape for poly(ADP-ribose) polymerase (PARP) inhibitors has been significantly advanced by the innovations detailed in Chinese Patent CN106432195B, which outlines a superior method for preparing (R)-2-(2-methylpyrrolidin-2-yl)-1H-benzimidazole-4-carboxamide, widely known as the core intermediate for ABT-888 (Veliparib). This specific compound serves as a critical building block in the synthesis of potent anticancer agents currently undergoing Phase III clinical trials for triple-negative breast cancer and non-small cell lung cancer. The patent addresses longstanding inefficiencies in the manufacturing of this complex heterocyclic structure by introducing a streamlined synthetic route that replaces harsh reaction conditions with milder, more selective reagents. By shifting away from traditional strong acid methodologies, this technology not only enhances the chemical integrity of the final product but also aligns with modern green chemistry principles, offering a robust solution for reliable pharmaceutical intermediate supplier networks seeking to optimize their production pipelines for high-value oncology drugs.
The significance of this patent extends beyond mere academic interest, representing a tangible leap forward in process chemistry that directly impacts the scalability and economic viability of ABT-888 production. Traditional synthesis pathways often suffer from low overall yields and complex purification requirements due to the sensitivity of the benzimidazole-4-carboxamide moiety to hydrolytic degradation. The disclosed invention mitigates these risks by utilizing acetic acid as a dual-purpose reagent for both cyclization and protecting group removal, a strategy that preserves the delicate amide functionality while ensuring high stereochemical fidelity. For R&D directors and process chemists, this approach offers a compelling alternative to legacy methods, promising higher purity profiles exceeding 99% HPLC purity and chiral purity greater than 99.5%, which are essential specifications for regulatory approval in global markets. This technological refinement underscores the importance of continuous process improvement in the competitive field of specialty chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described in earlier patent applications like CN101155797A, typically rely on multi-step sequences that introduce significant operational complexity and cost burdens to the manufacturing process. As illustrated in the reaction scheme below, conventional routes often necessitate the use of methyl iodide for methylation, followed by separate hydrolysis and coupling steps, culminating in a cyclization that requires strong acids like trifluoroacetic acid (TFA) or hydrochloric acid. These harsh conditions frequently lead to the unwanted hydrolysis of the carboxamide group, generating difficult-to-remove impurities that compromise the overall yield and necessitate extensive downstream purification efforts. Furthermore, many existing processes depend on palladium-catalyzed hydrogenation to remove benzyl protecting groups, which introduces the risk of heavy metal contamination and requires specialized high-pressure equipment, thereby increasing capital expenditure and safety protocols for production facilities.
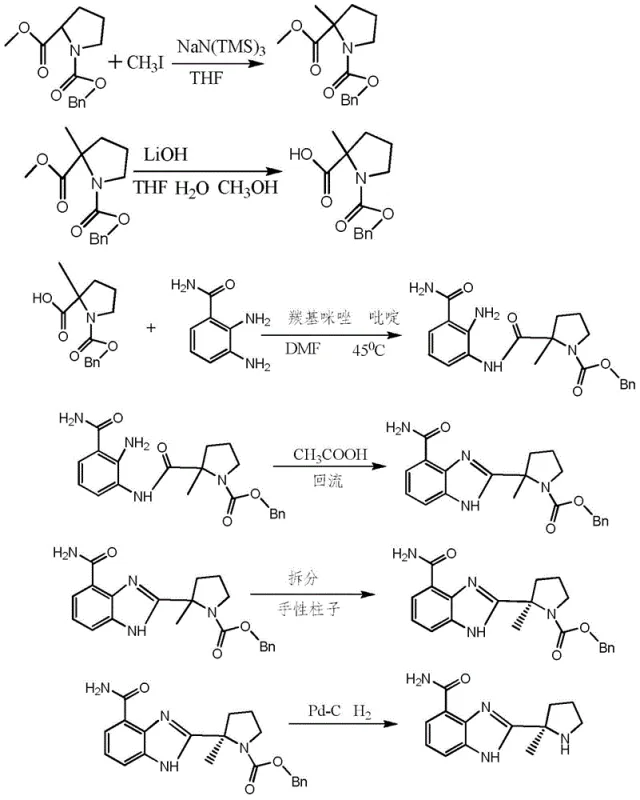
In addition to chemical inefficiencies, conventional methods often rely on chiral column chromatography to achieve the necessary enantiomeric excess, a technique that is notoriously difficult to scale and economically prohibitive for commercial production. The reliance on such separation technologies creates bottlenecks in supply chains, limiting the throughput of reliable agrochemical intermediate supplier and pharmaceutical partners alike. The accumulation of waste streams from strong acid usage and the need for solvent-intensive purification steps further exacerbate the environmental footprint of these legacy processes. For procurement managers, these factors translate into volatile pricing and extended lead times, as the complexity of the synthesis makes the supply chain vulnerable to disruptions in raw material availability or equipment maintenance. Consequently, there is a pressing industry demand for a more robust, scalable, and environmentally benign synthetic strategy that can deliver high-purity intermediates without the baggage of obsolete processing techniques.
The Novel Approach
The innovative methodology presented in Patent CN106432195B fundamentally reimagines the synthesis of this key intermediate by consolidating multiple transformation steps into a single, efficient operation. Instead of subjecting the molecule to sequential deprotection and cyclization under aggressive conditions, the novel approach utilizes a pre-formed chiral intermediate, R-(2-methyl-1-BOC-pyrrolidine)-2-carboxylic acid, which is coupled with 2,3-diaminobenzamide to form a stable amide precursor. The true brilliance of this route lies in the subsequent treatment with acetic acid, which acts as a mild yet effective medium to simultaneously remove the tert-butoxycarbonyl (BOC) protecting group and induce intramolecular cyclization to form the benzimidazole ring. This one-pot transformation eliminates the need for intermediate isolation and drastically reduces the exposure of the sensitive carboxamide group to hydrolytic conditions, resulting in a marked improvement in both yield and product quality. By avoiding the use of TFA and palladium catalysts, the process simplifies the workflow and enhances the safety profile of the manufacturing operation.
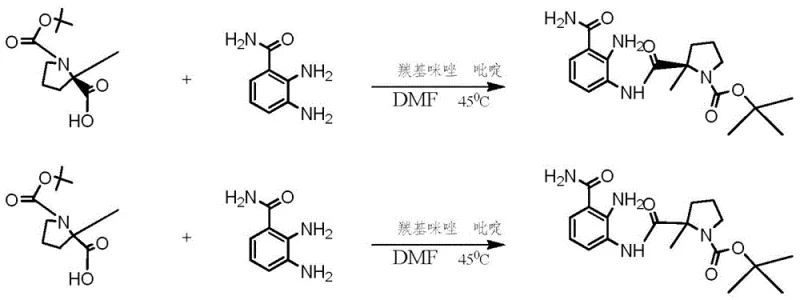
Furthermore, this novel approach offers flexibility in achieving the desired chiral purity without resorting to expensive chromatographic techniques. The patent discloses that if the starting material possesses sufficient optical purity, the final product can be obtained with greater than 99.5% enantiomeric excess directly. In cases where racemic or lower purity materials are used, the process incorporates a cost-effective chemical resolution step using chiral tartaric acid derivatives, such as L-(-)-di-p-toluoyltartaric acid, which can be performed in standard alcohol solvents. This adaptability allows manufacturers to optimize their cost structures based on the availability and price of starting materials, providing a strategic advantage in cost reduction in API manufacturing. The ability to recycle the acetic acid solvent further contributes to the economic and environmental sustainability of the process, making it an attractive option for large-scale commercial production where waste minimization is a key performance indicator.
Mechanistic Insights into Acetic Acid-Mediated Cyclization and Deprotection
The core chemical innovation of this patent revolves around the mechanistic role of acetic acid in facilitating the concurrent deprotection of the BOC group and the cyclization of the diamine intermediate. In traditional strong acid environments, the protonation of the carbonyl oxygen in the carboxamide side chain makes it highly susceptible to nucleophilic attack by water, leading to hydrolysis and the formation of carboxylic acid impurities. Acetic acid, being a weaker acid with a pKa of approximately 4.76, provides a sufficiently acidic environment to cleave the acid-labile BOC group via the formation of a tert-butyl cation intermediate, yet it lacks the aggressive protonating power that triggers rapid amide hydrolysis. This delicate balance allows the free amine generated from BOC removal to immediately attack the adjacent amide carbonyl, closing the ring to form the benzimidazole system before hydrolytic degradation can occur. This kinetic control is crucial for maintaining the structural integrity of the 4-carboxamide substituent, which is essential for the biological activity of the final PARP inhibitor.
From an impurity control perspective, this mechanism significantly simplifies the purification profile of the reaction mixture. By minimizing the formation of hydrolyzed byproducts, the crude product obtained after neutralization and precipitation is of exceptionally high quality, often requiring only a simple recrystallization from methanol to meet stringent purity specifications. The absence of palladium residues, which are common in hydrogenation-based deprotection methods, removes the need for specialized scavenging resins or activated carbon treatments, further streamlining the downstream processing. For quality control laboratories, this translates to faster release times and reduced analytical burden, as the impurity spectrum is cleaner and more predictable. The robustness of this chemical transformation ensures consistent batch-to-batch reproducibility, a critical factor for regulatory compliance and the validation of commercial manufacturing processes in the highly regulated pharmaceutical industry.
How to Synthesize (R)-2-(2-methylpyrrolidin-2-yl)-1H-benzimidazole-4-carboxamide Efficiently
The implementation of this streamlined synthesis route requires careful attention to reaction parameters, particularly the concentration of acetic acid and the reflux duration, to maximize yield and purity. The patent provides several embodiments demonstrating that acetic acid concentrations ranging from 50% to 100% can be effectively utilized, with reaction times varying between 2 to 8 hours depending on the specific solvent composition. The process begins with the coupling of the chiral pyrrolidine acid with the diamine, followed by the critical cyclization step where the choice of acid concentration can be tuned to balance reaction rate and solubility. Detailed standard operating procedures for scaling this reaction from laboratory to pilot plant are essential to ensure thermal management and efficient solvent recovery. For a comprehensive guide on the specific stoichiometry, temperature profiles, and workup procedures validated in the patent examples, please refer to the standardized synthesis steps provided below.
- Couple R-(2-methyl-1-BOC-pyrrolidine)-2-carboxylic acid with 2,3-diaminobenzamide to form the protected amide intermediate.
- Reflux the intermediate in 50-100% acetic acid solution for 2 to 8 hours to effect simultaneous cyclization and BOC deprotection.
- Neutralize the reaction mixture with alkali, precipitate the product, and optionally perform chiral resolution using tartaric acid derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of the synthetic methodology described in Patent CN106432195B offers profound commercial advantages that resonate deeply with procurement managers and supply chain leaders focused on efficiency and cost containment. By eliminating the need for expensive and hazardous reagents such as trifluoroacetic acid and palladium on carbon, the process inherently reduces the raw material costs associated with each production batch. The removal of the hydrogenation step not only lowers capital equipment requirements but also mitigates the safety risks associated with high-pressure hydrogen gas, leading to lower insurance and operational overheads. Furthermore, the ability to recycle the acetic acid solvent creates a closed-loop system that minimizes waste disposal costs and environmental compliance burdens, contributing to substantial cost savings over the lifecycle of the product. These efficiencies allow suppliers to offer more competitive pricing structures while maintaining healthy margins, a critical factor in the price-sensitive generic pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and strong mineral acids removes the necessity for complex purification steps designed to remove trace metal impurities, which are often costly and time-consuming. This simplification of the downstream processing workflow directly translates to reduced labor hours and lower consumption of auxiliary materials like scavengers and filtration media. Additionally, the avoidance of chiral column chromatography, a technique with low throughput and high solvent consumption, dramatically lowers the variable costs per kilogram of product. The overall process intensification means that the same production capacity can yield significantly more product in less time, optimizing asset utilization and driving down the unit cost of goods sold for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like acetic acid and readily available chiral resolving agents ensures a stable and resilient supply chain that is less susceptible to geopolitical disruptions or vendor shortages. Unlike specialized catalysts or custom-synthesized reagents that may have long lead times, the inputs for this process are globally sourced and abundant, guaranteeing continuity of supply even during market fluctuations. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the risk of batch failures and production delays. This reliability is paramount for securing long-term contracts with major pharmaceutical companies that require guaranteed delivery schedules for their clinical and commercial programs.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, which involves straightforward neutralization and precipitation, makes this process highly amenable to scale-up from kilogram to multi-ton quantities without significant engineering challenges. The reduction in hazardous waste generation, particularly the absence of heavy metal waste streams and halogenated solvent usage, aligns perfectly with increasingly stringent environmental regulations and corporate sustainability goals. Facilities can operate with smaller waste treatment footprints and lower emissions, enhancing their social license to operate and reducing the regulatory friction often associated with expanding production capacity. This environmental stewardship not only future-proofs the manufacturing site but also appeals to eco-conscious stakeholders and investors in the modern chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route, providing clarity for stakeholders evaluating its potential for integration into their supply chains. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is essential for assessing the feasibility of adopting this technology for commercial production of ABT-888 intermediates.
Q: Why is acetic acid preferred over trifluoroacetic acid (TFA) for this cyclization?
A: Acetic acid is a weaker acid that prevents the hydrolysis of the sensitive carboxamide group, which often occurs with strong acids like TFA or HCl, thereby significantly improving product purity and yield.
Q: Is chiral column chromatography required for this process?
A: No, the process eliminates the need for expensive chiral column separation by utilizing high-purity starting materials or employing cost-effective chemical resolution with tartaric acid derivatives.
Q: How does this method impact heavy metal contamination risks?
A: This route omits the palladium-carbon catalytic hydrogenation step found in conventional methods, thereby removing the risk of residual palladium contamination and simplifying purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable ABT-888 Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the development of next-generation oncology therapeutics. Our team of expert process chemists has thoroughly analyzed the innovations presented in Patent CN106432195B and is fully equipped to translate this laboratory-scale success into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot batches to full-scale manufacturing is seamless and compliant with cGMP standards. Our state-of-the-art facilities are designed to handle the specific requirements of this acetic acid-mediated cyclization, complete with rigorous QC labs capable of verifying stringent purity specifications and chiral integrity to meet the exacting demands of global regulatory agencies.
We invite pharmaceutical partners and procurement specialists to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Our technical procurement team is ready to provide specific COA data from trial batches and comprehensive route feasibility assessments to demonstrate how this optimized process can enhance your supply chain efficiency. By collaborating with NINGBO INNO PHARMCHEM, you gain access to a reliable partner committed to delivering high-quality intermediates that accelerate your drug development timelines while optimizing your overall cost structure. Contact us today to discuss how we can support your ABT-888 production needs with precision, reliability, and scientific excellence.
