Revolutionizing 2-Aryloxy Ethanol Production via Cobalt-Catalyzed C-H Activation for Commercial Scale-Up
Revolutionizing 2-Aryloxy Ethanol Production via Cobalt-Catalyzed C-H Activation for Commercial Scale-Up
The landscape of fine chemical synthesis is undergoing a paradigm shift towards sustainability and atom economy, driven by the urgent need to replace hazardous reagents with greener alternatives. Patent CN111004176A introduces a groundbreaking methodology for the synthesis of 2-aryloxy ethanol compounds, a structural motif ubiquitous in bioactive molecules and pharmaceutical intermediates. This innovation leverages a cobalt-catalyzed dehydrogenative cross-coupling strategy, utilizing N-(naphthalen-1-yl)picolinamide derivatives and industrial-grade ethylene glycol as primary feedstocks. By bypassing the traditional reliance on toxic ethylene oxide or halogenated precursors, this technology offers a compelling value proposition for R&D directors seeking robust synthetic routes and procurement managers aiming for significant cost reduction in API manufacturing. The method not only enhances operational safety by eliminating volatile gases but also improves the overall economic feasibility through the use of earth-abundant base metal catalysts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-aryloxy ethanol compounds has been dominated by nucleophilic substitution reactions involving phenols and highly reactive, dangerous reagents. As illustrated in the general reaction scheme below, the classical approach typically employs ethylene oxide, a flammable and toxic gas, or 2-haloethanols, which pose significant environmental and health risks.
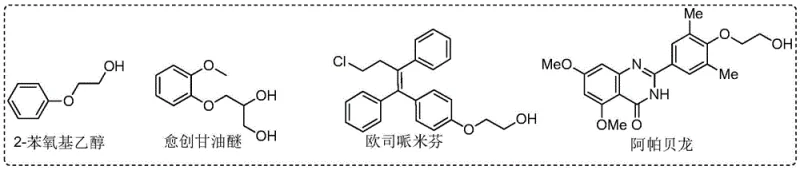
Furthermore, while recent advancements have attempted to mitigate these risks, such as the method reported by the Wu group using ethylene carbonate, significant drawbacks remain. Although ethylene carbonate is a greener alternative, the reaction requires harsh conditions, specifically temperatures as high as 170°C in DMF, which limits its applicability to thermally sensitive substrates and increases energy consumption. Additionally, transition metal-catalyzed coupling of halobenzenes with glycols, while effective, generates stoichiometric amounts of metal salt waste and relies on pre-halogenated aromatic hydrocarbons, thereby reducing atom economy and increasing the burden on waste treatment facilities.
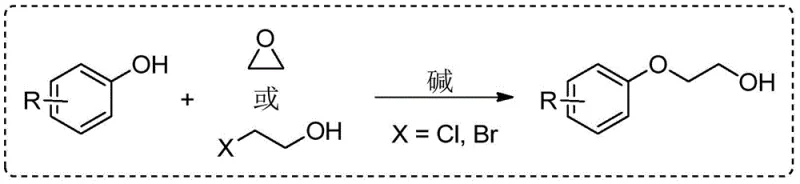
The Novel Approach
In stark contrast to these legacy technologies, the novel approach detailed in CN111004176A utilizes a direct C-H bond functionalization strategy. This method transforms inert carbon-hydrogen bonds into valuable carbon-oxygen bonds without the need for pre-functionalized halides or toxic epoxides. The core innovation lies in the use of a picolinamide directing group on the naphthalene scaffold, which facilitates regioselective ortho-C-H activation. By employing cheap and readily available cobalt salts as catalysts and manganese salts as oxidants, the process achieves high efficiency under relatively mild thermal conditions (90-110°C). This shift from stoichiometric hazardous reagents to catalytic C-H activation represents a fundamental upgrade in synthetic design, aligning perfectly with modern green chemistry principles and offering a scalable pathway for the reliable pharmaceutical intermediate supplier market.
Mechanistic Insights into Cobalt-Catalyzed Dehydrogenative Cross-Coupling
The mechanistic elegance of this transformation relies on the synergistic interaction between the cobalt catalyst, the oxidant, and the picolinamide directing group. The reaction initiates with the coordination of the cobalt species to the nitrogen atom of the picolinamide moiety, which directs the metal center to the proximal C-H bond on the naphthalene ring. This coordination facilitates the cleavage of the C-H bond, forming a stable cyclometallated cobalt intermediate. Subsequently, the ethylene glycol interacts with this organometallic species, likely undergoing deprotonation by the base (KOAc) to form a cobalt-alkoxide complex. The presence of the oxidant, such as Mn(OAc)2, is crucial for regenerating the active high-valent cobalt species and facilitating the reductive elimination step that forms the final C-O bond, releasing the 2-aryloxy ethanol product and restoring the catalyst for the next cycle.
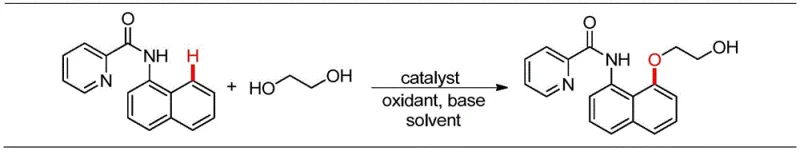
From an impurity control perspective, this mechanism offers distinct advantages over traditional nucleophilic substitutions. Since the reaction does not involve highly reactive electrophiles like ethylene oxide, the formation of poly-ether byproducts or oligomerization side reactions is significantly suppressed. The use of a specific directing group ensures high regioselectivity, minimizing the formation of meta- or para-substituted isomers which are often difficult to separate. Furthermore, the choice of fluorobenzene as a solvent provides a stable medium that solubilizes the organic substrates while remaining inert under the oxidative conditions, thereby ensuring a clean reaction profile that simplifies downstream purification and enhances the purity of the final high-purity pharmaceutical intermediates.
How to Synthesize 8-Hydroxyethoxy-N-(naphthalen-1-yl)picolinamide Efficiently
The practical implementation of this synthesis involves a straightforward one-pot procedure that is amenable to standard laboratory and pilot plant equipment. The process begins with the preparation of the N-(naphthalen-1-yl)picolinamide substrate, followed by the key coupling step with ethylene glycol. Detailed operational parameters, including precise stoichiometry and workup procedures, are critical for maximizing yield and reproducibility. For a comprehensive understanding of the standardized synthetic steps required to achieve the reported 70-72% yields, please refer to the technical guide below.
- Prepare the reaction mixture by combining N-(naphthalen-1-yl)picolinamide substrate, cobalt catalyst (e.g., Co(OAc)2), oxidant (e.g., Mn(OAc)2), and base (e.g., KOAc) in a suitable solvent like fluorobenzene.
- Add ethylene glycol or diethylene glycol to the sealed reaction vessel and heat the mixture to a temperature range of 90-110°C for 12-24 hours under stirring.
- Upon completion, perform post-processing including extraction with ethyl acetate, drying over anhydrous magnesium sulfate, and purification via column chromatography to isolate the pure 2-aryloxy ethanol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cobalt-catalyzed technology translates into tangible strategic benefits beyond mere chemical novelty. The transition from precious metal catalysts or hazardous gaseous reagents to base metal systems fundamentally alters the cost structure and risk profile of the supply chain. By eliminating the need for specialized high-pressure equipment required for ethylene oxide handling and avoiding the volatility of halogenated feedstocks, manufacturers can achieve substantial cost savings in facility maintenance and safety compliance. Moreover, the reliance on commodity chemicals like ethylene glycol ensures a stable and continuous supply of raw materials, insulating production schedules from the fluctuations often seen with specialty reagents.
- Cost Reduction in Manufacturing: The replacement of expensive palladium or rhodium catalysts with inexpensive cobalt acetate drastically lowers the direct material cost per kilogram of product. Additionally, the avoidance of toxic ethylene oxide removes the necessity for complex scrubbing systems and specialized containment infrastructure, leading to reduced capital expenditure (CAPEX) and operational expenditure (OPEX). The high atom economy of the C-H activation process means less waste is generated per unit of product, further driving down waste disposal costs and improving the overall process mass intensity (PMI).
- Enhanced Supply Chain Reliability: Ethylene glycol is a globally produced commodity chemical with a robust supply network, unlike specialized halogenated aromatics which may have limited suppliers. This abundance ensures that production can be scaled up rapidly without facing raw material bottlenecks. Furthermore, the mild reaction conditions (100°C) reduce the energy load on manufacturing plants compared to high-temperature alternatives, contributing to more predictable production timelines and reducing the risk of thermal runaway incidents that could disrupt supply continuity.
- Scalability and Environmental Compliance: The generation of benign byproducts and the absence of halogenated waste streams simplify the environmental permitting process for new manufacturing lines. The process is inherently safer, reducing the regulatory burden associated with handling carcinogenic or mutagenic reagents. This environmental friendliness not only aligns with corporate sustainability goals but also future-proofs the supply chain against increasingly stringent global environmental regulations, ensuring long-term viability for the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cobalt-catalyzed synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the method's versatility and operational requirements for potential partners.
Q: What are the primary advantages of this cobalt-catalyzed method over traditional phenol-based synthesis?
A: Unlike traditional methods that rely on toxic ethylene oxide or hazardous haloethanols, this cobalt-catalyzed approach utilizes inexpensive ethylene glycol and achieves C-H activation. This results in significantly higher atom economy, eliminates the need for pre-functionalized phenols, and operates at milder temperatures (100°C) compared to alternative high-temperature carbonate methods.
Q: Is the catalyst system cost-effective for large-scale manufacturing?
A: Yes, the process employs cobalt acetate (Co(OAc)2) and manganese acetate (Mn(OAc)2), which are substantially cheaper and more abundant than precious metals like palladium or rhodium. This transition to base metal catalysis drastically reduces raw material costs and simplifies the removal of heavy metal residues from the final pharmaceutical intermediate.
Q: What is the substrate scope regarding substituents on the naphthalene ring?
A: The method demonstrates versatility with various substituents (R groups) including hydrogen, halogens like chlorine or bromine, and alkyl groups. While electron-neutral and electron-rich substrates generally provide high yields (up to 72%), electron-deficient or sterically hindered substrates may require optimization, yet the protocol remains robust for diverse chemical scaffolds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aryloxy Ethanol Supplier
The technological breakthroughs encapsulated in CN111004176A represent a significant leap forward in the efficient production of valuable chemical building blocks. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of 2-aryloxy ethanol intermediate meets the exacting standards required by the global pharmaceutical industry.
We invite forward-thinking organizations to collaborate with us to leverage this advanced synthetic technology for their pipeline projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive landscape of fine chemical manufacturing.
