Advanced Nickel-Catalyzed Asymmetric Allylic Alkylation for Scalable Alpha-Acyllactam Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex molecular architectures, particularly those featuring quaternary carbon stereocenters which are prevalent in bioactive natural products and drug candidates. Patent CN113735669A introduces a groundbreaking approach to this challenge by detailing a nickel-catalyzed asymmetric allylic alkylation method. This technology enables the efficient construction of alpha-acyllactam compounds bearing quaternary carbon chiral centers, utilizing simple silane-protected allyl alcohol reagents. By leveraging low-equivalent nickel catalysts and cost-effective chiral ligands, this invention addresses critical bottlenecks in synthetic efficiency and operational simplicity, positioning itself as a vital tool for the scalable production of high-purity pharmaceutical intermediates.
Historically, the construction of quaternary carbon centers has relied heavily on noble metal catalysis, presenting significant economic and logistical hurdles for large-scale manufacturing. Conventional methods often employ palladium, rhodium, or iridium complexes, such as those reported by the Trost and Stoltz groups, which, while effective, suffer from high material costs and potential toxicity concerns associated with heavy metal residues. Furthermore, many of these established protocols require expensive ligands or harsh activation strategies, such as the use of strong bases like lithium tert-butoxide, which can limit functional group tolerance and complicate downstream processing. These limitations underscore the urgent need for alternative catalytic systems that maintain high stereocontrol without the prohibitive costs of second and third-row transition metals.
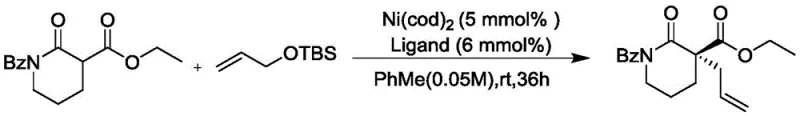
The novel approach disclosed in the patent circumvents these issues by utilizing nickel, an earth-abundant first-row transition metal, as the central catalytic element. This method achieves exceptional enantioselectivity and yield by employing a specific combination of Ni(cod)2 and chiral bisphosphine ligands, notably ligand L6. Unlike previous nickel-catalyzed attempts that were restricted to specific cyclic ketone substrates, this invention expands the scope to include a wide variety of alpha-acylated lactams. The use of silane-protected allyl alcohols serves as a stable and reactive allyl source, eliminating the need for unstable allyl halides or carbonates. This strategic shift not only reduces raw material costs but also simplifies the reaction workflow, making it highly attractive for industrial application where cost reduction in pharmaceutical intermediate manufacturing is paramount.
Mechanistically, the success of this transformation hinges on the precise interplay between the nickel center and the chiral ligand environment. The reaction likely proceeds through a standard oxidative addition of the allyl species to the Ni(0) center, followed by nucleophilic attack by the enolate generated from the alpha-acyllactam. The chiral ligand L6, identified through extensive screening as superior to other phosphine ligands like L1 or L5, creates a rigid chiral pocket that effectively discriminates between the enantiotopic faces of the pi-allyl nickel intermediate. This steric and electronic control is crucial for achieving the observed high enantiomeric excess (ee), with optimized conditions yielding products with up to 96% ee. The ability to fine-tune this selectivity through ligand modification provides R&D teams with a versatile platform for optimizing specific target molecules.
Furthermore, the impurity profile of the resulting compounds is tightly controlled by the mild reaction conditions and the specificity of the nickel catalyst. The use of toluene as a preferred solvent at temperatures ranging from 0°C to 5°C minimizes thermal degradation and side reactions such as polymerization or isomerization of the allyl group. The protocol specifies a catalyst loading of merely 5 mmol% for nickel and 6 mmol% for the ligand, which is significantly lower than the 10-12 mol% often required in comparable palladium or earlier nickel systems. This reduction in catalyst loading not only lowers the cost of goods but also simplifies the purification process, ensuring that the final API intermediates meet stringent purity specifications with minimal heavy metal contamination.
To facilitate the practical implementation of this technology, the patent outlines a clear pathway for synthesizing the target alpha-acyllactam quaternary carbon chiral center compounds. The process begins with the preparation of the necessary building blocks: the alpha-acylated lactam substrate and the silane-protected allyl alcohol. These precursors are then subjected to the nickel-catalyzed coupling under inert conditions. The following section details the standardized operational procedure derived from the patent examples, providing a reliable framework for reproducing these high-value chiral intermediates in a laboratory or pilot plant setting.
For procurement and supply chain leaders, the adoption of this nickel-catalyzed methodology offers compelling commercial advantages that extend beyond mere technical feasibility. The transition from noble metals to nickel represents a substantial opportunity for cost reduction in pharmaceutical intermediate manufacturing. Nickel salts and the associated ligands are inherently less expensive than their palladium or iridium counterparts, and the lower catalyst loading further amplifies these savings. Additionally, the use of simple, commercially available silane-protected reagents eliminates the need for complex, multi-step synthesis of specialized allyl donors, thereby streamlining the supply chain and reducing lead times for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of switching to a nickel-based catalytic system is profound. By replacing expensive noble metals with earth-abundant nickel, manufacturers can significantly lower the direct material costs associated with catalysis. The patent demonstrates that high yields (up to 94.1%) and excellent enantioselectivity can be achieved with catalyst loadings as low as 5 mol%, which drastically reduces the expense per kilogram of product. Furthermore, the simplified workup procedures implied by the clean reaction profiles reduce solvent consumption and waste disposal costs, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: Reliance on scarce precious metals like palladium and iridium introduces volatility into the supply chain, subject to geopolitical fluctuations and mining constraints. Nickel, being a widely produced industrial metal, offers a much more stable and secure supply base. The reagents used in this process, such as silane-protected allyl alcohols and benzoyl-protected lactams, are synthesized from readily available starting materials. This accessibility ensures consistent availability of raw materials, mitigating the risk of production delays and allowing for more accurate forecasting and inventory management for long-term commercial projects.
- Scalability and Environmental Compliance: The operational parameters defined in the patent, including moderate temperatures (0-5°C) and the use of common solvents like toluene, are highly conducive to scale-up. The reaction does not require cryogenic conditions or extreme pressures, which simplifies reactor design and operation. From an environmental perspective, the reduced use of heavy metals aligns with green chemistry principles and increasingly strict regulatory requirements regarding metal residues in pharmaceuticals. The ability to produce complex chiral intermediates with high efficiency and lower environmental impact positions this technology as a sustainable choice for modern chemical manufacturing.
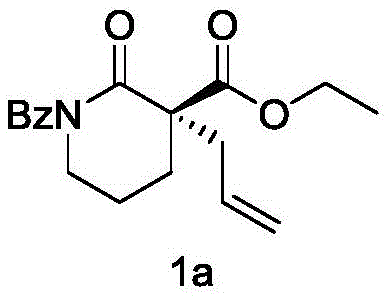
- Preparation of Substrates: Synthesize the alpha-acylated lactam compound (Compound 2) via benzoyl protection and alpha-acylation, and prepare the silane-protected allyl alcohol reagent (Compound 3) from allyl acid.
- Catalyst System Setup: In an inert atmosphere, mix Ni(cod)2 (5 mmol%) and chiral ligand L6 (6 mmol%) in toluene and stir at room temperature to form the active catalytic species.
- Asymmetric Alkylation Reaction: Add the lactam substrate and allyl reagent to the catalyst system and react at 0-5°C for 36 hours to yield the chiral quaternary carbon product with high enantioselectivity.
Frequently Asked Questions (FAQ)
Q: What is the primary advantage of using nickel over palladium in this allylic alkylation?
A: Nickel is a first-row transition metal that is significantly more abundant and cost-effective than noble metals like palladium or iridium. The patent demonstrates that nickel catalysts, specifically Ni(cod)2 paired with ligand L6, can achieve comparable or superior enantioselectivity (up to 96% ee) at lower catalyst loadings, reducing overall production costs.
Q: What types of lactam rings are compatible with this synthetic method?
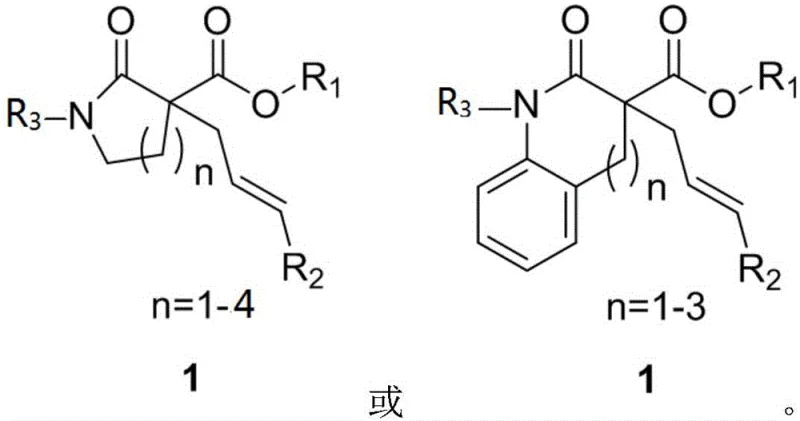
A: The method exhibits broad substrate scope, successfully accommodating five-to-eight-membered nitrogen heterocycles as well as six-to-eight-membered benzo-fused nitrogen heterocycles. This versatility allows for the construction of diverse molecular scaffolds essential for medicinal chemistry.
Q: Why is silane protection used for the allyl alcohol reagent?
A: Silane protection (using groups like TBS, TMS, or TBDPS) stabilizes the allyl alcohol reagent, preventing premature decomposition or side reactions. The patent data indicates that tert-butyldimethylsilyl (TBS) protection offers the optimal balance of stability and reactivity, leading to higher yields (90%) and excellent enantioselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Acyllactam Supplier
The technological advancements detailed in patent CN113735669A highlight the immense potential of nickel-catalyzed asymmetric synthesis in modern drug development. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative academic and patent literature into robust, commercial-grade processes. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch of alpha-acyllactam intermediates meets stringent purity specifications and enantiomeric excess requirements demanded by global regulatory bodies.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cost-effective nickel-catalyzed technology for your specific pipeline needs. Whether you require custom synthesis of novel lactam scaffolds or scale-up of existing routes, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring a seamless transition from bench-scale discovery to commercial supply.
