Advanced Rhodium-Catalyzed Synthesis of Pyrazolidone-Benzo 1,3-Oxazepine Intermediates for Pharmaceutical Applications
Introduction to Next-Generation Benzoxazepine Synthesis
The pharmaceutical industry continuously seeks efficient pathways to access complex heterocyclic scaffolds that serve as critical cores for bioactive molecules. Patent CN113185536A discloses a groundbreaking synthesis method for pyrazolidone-benzo 1,3-oxazepine compounds, a class of fused heterocycles known for their potent physiological activities including anticonvulsant, antitumor, and anxiolytic properties. This innovation addresses the longstanding challenges in constructing these nitrogen-oxygen containing frameworks by utilizing a direct series reaction between 1-aryl pyrazolidone compounds and diazo naphthalenone compounds. Unlike traditional approaches that rely on harsh conditions and poor atom economy, this rhodium-catalyzed protocol operates under mild thermal conditions with exceptional regioselectivity. For R&D directors and procurement specialists, this represents a significant opportunity to streamline the supply chain for high-purity pharmaceutical intermediates while reducing the environmental footprint associated with legacy synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of benzo 1,3-oxazepine rings has predominantly relied on the condensation reactions of bifunctional benzene derivatives, specifically di-halogenated or mono-halogenated benzenes, with various nucleophilic components. While these classical methods are chemically reliable, they suffer from severe economic and operational inefficiencies that hinder modern commercial scale-up of complex pharmaceutical intermediates. The starting materials, particularly poly-halogenated aromatics, often require multi-step preparation sequences, driving up raw material costs and extending lead times for high-purity intermediates. Furthermore, these stoichiometric processes generate substantial amounts of halogenated salt by-products, resulting in low atom economy and creating significant waste disposal challenges. The necessity for rigorous purification to remove trace halogen impurities further complicates the manufacturing process, often requiring specialized equipment and increasing the overall cost of goods sold for the final active pharmaceutical ingredient.
The Novel Approach
The methodology described in CN113185536A fundamentally reimagines this synthetic challenge by employing a transition metal-catalyzed cascade reaction that directly couples 1-aryl pyrazolidones with diazo naphthalenones. This approach bypasses the need for pre-functionalized halogenated precursors, utilizing instead simple, commercially available starting materials that are inexpensive and easy to source. The reaction proceeds through a sophisticated C-H activation mechanism facilitated by a pentamethylcyclopentadienyl rhodium catalyst, enabling the formation of multiple bonds in a single pot. This one-pot strategy not only simplifies the operational workflow but also drastically improves the atom economy of the transformation. By eliminating the generation of stoichiometric halogen waste and reducing the number of isolation steps, this novel route offers a greener, more sustainable alternative that aligns perfectly with modern green chemistry principles and cost reduction in API manufacturing initiatives.
Mechanistic Insights into Rhodium-Catalyzed Cyclization
The core of this technological breakthrough lies in the precise orchestration of a Rhodium(III)-catalyzed C-H activation and subsequent cyclization sequence. The reaction initiates with the coordination of the 1-aryl pyrazolidone substrate to the cationic Rhodium species generated in situ from the dimeric precursor [RhCp*Cl2]2. This coordination directs the metal center to activate the ortho-C-H bond of the aryl ring, forming a stable five-membered rhodacycle intermediate. Subsequently, the diazo naphthalenone component undergoes carbene transfer or insertion into the Rh-C bond, followed by migratory insertion and reductive elimination steps that forge the new C-C and C-O bonds essential for the oxazepine ring closure. The presence of silver acetate as an oxidant is critical for regenerating the active Rh(III) catalytic species from the reduced Rh(I) state, ensuring the catalytic cycle continues efficiently without the accumulation of inactive metal species.
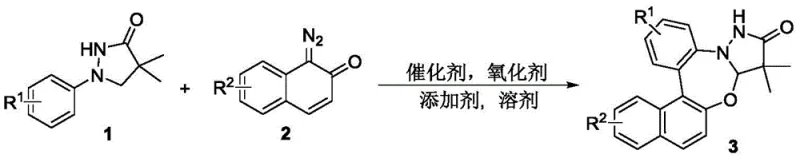
Crucially, the addition of carboxylic acid additives, such as 2,4,6-trimethylbenzoic acid (TMBzOH), plays a pivotal role in modulating the reactivity and selectivity of the transformation. These additives likely function as proton shuttles or ligands that stabilize key transition states, thereby suppressing side reactions and enhancing the yield of the desired fused heterocycle. The mechanistic pathway is highly sensitive to the electronic nature of the substrates, yet the system demonstrates remarkable tolerance to a wide range of functional groups including halogens, alkyls, and electron-withdrawing groups. This robustness ensures that the impurity profile of the final product remains clean, with minimal formation of regioisomers or oligomeric by-products. For quality control teams, this inherent selectivity translates to simpler purification protocols and higher confidence in the consistency of the batch-to-batch purity specifications required for regulatory compliance.
How to Synthesize Pyrazolidone-Benzo 1,3-Oxazepine Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, strict adherence to the optimized reaction parameters is essential. The standard protocol involves charging a reaction vessel with the 1-aryl pyrazolidone and diazo naphthalenone substrates in a molar ratio of approximately 1:1.5 to ensure complete consumption of the limiting reagent. The reaction is conducted in acetonitrile, which has been identified as the superior solvent for balancing solubility and reaction kinetics compared to chlorinated solvents or alcohols. The inclusion of 4Å molecular sieves is a critical detail often overlooked; these desiccants scavenge trace moisture that could otherwise decompose the sensitive diazo compound or deactivate the Lewis acidic catalyst, thereby significantly boosting the isolated yield.
- Combine 1-aryl pyrazolidone (1a) and diazo naphthalenone (2a) in acetonitrile solvent within a sealed reaction vessel under an argon atmosphere.
- Add the catalyst [RhCp*Cl2]2, oxidant AgOAc, and additive 2,4,6-trimethylbenzoic acid (TMBzOH) along with 4Å molecular sieves to the mixture.
- Heat the reaction mixture to 100 °C in an oil bath for 12 hours, then cool, extract, and purify via silica gel column chromatography to isolate the product.
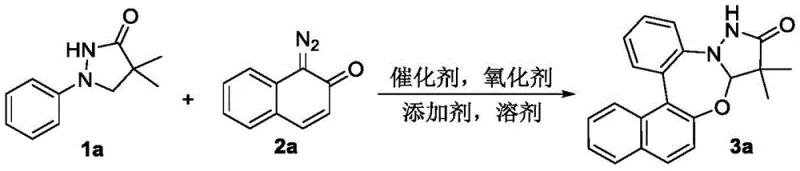
The detailed standardized synthesis steps for this high-efficiency route are provided in the structured guide below, which outlines the precise addition order, temperature ramping, and workup procedures necessary to achieve the reported 75% yield for the model compound 3a. Following the reaction completion at 100 °C over 12 hours, the mixture is cooled and subjected to a standard aqueous workup involving extraction with ethyl acetate. Final purification is achieved via silica gel column chromatography using a petroleum ether and ethyl acetate gradient, yielding the target pyrazolidone-benzo 1,3-oxazepine as a high-purity white solid suitable for downstream biological evaluation or further chemical modification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical elegance. The shift away from halogenated starting materials towards simple aryl pyrazolidones and diazo compounds significantly de-risks the supply chain by diversifying the vendor base for raw materials. Since these precursors are commodity chemicals rather than specialized custom synthons, their market availability is high, and price volatility is low. This stability ensures consistent production scheduling and reduces the likelihood of delays caused by raw material shortages, which is a common pain point in the manufacturing of complex heterocyclic intermediates. Furthermore, the simplified one-pot nature of the reaction reduces the total processing time and labor hours required per batch, directly contributing to lower operational expenditures.
- Cost Reduction in Manufacturing: The elimination of expensive halogenated precursors and the reduction in purification steps lead to substantial cost savings. By avoiding the generation of stoichiometric salt waste, the facility saves on waste treatment costs and solvent usage associated with extensive washing procedures. The high atom economy means that a greater proportion of the input mass is converted into valuable product, maximizing the return on investment for every kilogram of raw material purchased. Additionally, the use of a robust catalyst system allows for potentially lower catalyst loadings in optimized large-scale runs, further driving down the cost of goods.
- Enhanced Supply Chain Reliability: The reliance on broadly available organic building blocks ensures that the supply chain is resilient against geopolitical or logistical disruptions affecting niche chemical suppliers. The mild reaction conditions (60-120 °C) reduce the energy intensity of the process compared to high-temperature or cryogenic alternatives, lowering utility costs and minimizing the risk of thermal runaway incidents. This operational safety profile facilitates easier technology transfer between manufacturing sites, ensuring continuity of supply even if primary production facilities face unforeseen maintenance or capacity issues.
- Scalability and Environmental Compliance: The process aligns with stringent environmental regulations by minimizing hazardous waste generation. The absence of heavy halogen by-products simplifies effluent treatment, reducing the burden on environmental health and safety departments. The scalability of the reaction has been demonstrated through the successful synthesis of numerous derivatives with varying electronic properties, proving that the method is not limited to a single substrate. This versatility allows manufacturers to produce a library of analogues from a single platform technology, accelerating the development timeline for new drug candidates and improving time-to-market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Rhodium-catalyzed synthesis platform. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential licensees or manufacturing partners.
Q: What are the primary advantages of this Rhodium-catalyzed method over traditional halogenated benzene condensation?
A: This novel method utilizes readily available 1-aryl pyrazolidones and diazo naphthalenones, avoiding the costly and multi-step preparation of di-halogenated benzene precursors. It significantly improves atom economy by eliminating halogen waste and offers milder reaction conditions with high regioselectivity.
Q: What represents the optimal catalytic system for this transformation?
A: The patent identifies [RhCp*Cl2]2 as the preferred catalyst, used in conjunction with silver acetate (AgOAc) as the oxidant and 2,4,6-trimethylbenzoic acid (TMBzOH) as a crucial additive. This combination, particularly in acetonitrile solvent at 100 °C, yields up to 75% isolated yield for the model compound.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It employs simple operation steps, uses commercially accessible raw materials, and operates under relatively mild thermal conditions (60-120 °C). The high selectivity reduces downstream purification burdens, making it highly viable for cost reduction in API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazolidone-Benzo 1,3-Oxazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Rhodium-catalyzed technology in accelerating the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of pyrazolidone-benzo 1,3-oxazepine intermediate delivered meets the highest global regulatory standards. We understand that consistency and quality are paramount in the pharmaceutical supply chain, and our dedicated technical team is committed to maintaining the integrity of this advanced synthetic route.
We invite you to collaborate with us to leverage this innovative chemistry for your specific pipeline needs. Our experts can provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this method can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data for our reference standards and to discuss route feasibility assessments for your target molecules. Together, we can drive efficiency and innovation in the synthesis of these vital pharmaceutical intermediates.
