Revolutionizing Alpha-Bromo Aromatic Ketones Production via Aqueous Copper Catalysis
Revolutionizing Alpha-Bromo Aromatic Ketones Production via Aqueous Copper Catalysis
The chemical manufacturing landscape is undergoing a significant transformation driven by the urgent need for greener, more sustainable synthetic pathways, particularly in the production of high-value intermediates. Patent CN102503751B represents a pivotal advancement in this domain, introducing a robust method for synthesizing alpha-bromo aromatic ketones that fundamentally alters the traditional risk-reward profile of halogenation chemistry. This technology leverages a copper-nitrate-catalyzed oxidative system using molecular oxygen or air as the terminal oxidant, operating efficiently within an aqueous medium. For R&D directors and process chemists, this signifies a departure from hazardous volatile organic compounds (VOCs) toward a benign water-based protocol that maintains high reactivity while drastically minimizing environmental footprint. The strategic implementation of this methodology offers a compelling value proposition for supply chain leaders seeking to mitigate regulatory risks associated with solvent emissions and hazardous reagent handling.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of alpha-bromoaryl ketones has relied heavily on the direct addition of elemental bromine to aromatic ketones dissolved in organic solvents such as acetic acid, ether, dioxane, or chlorinated hydrocarbons. This conventional approach suffers from severe inherent drawbacks, primarily the poor atom economy where the reaction generates an equimolar amount of hydrogen bromide waste alongside the desired product, effectively capping bromine utilization at merely 50%. Furthermore, the reliance on liquid bromine introduces significant safety hazards due to its high volatility, corrosivity, and toxicity, necessitating expensive containment infrastructure and rigorous safety protocols. The use of organic solvents exacerbates these issues by creating substantial volumes of hazardous waste streams that require complex and costly recovery or incineration processes, thereby inflating the overall cost of goods sold and complicating environmental compliance audits for large-scale manufacturing facilities.
The Novel Approach
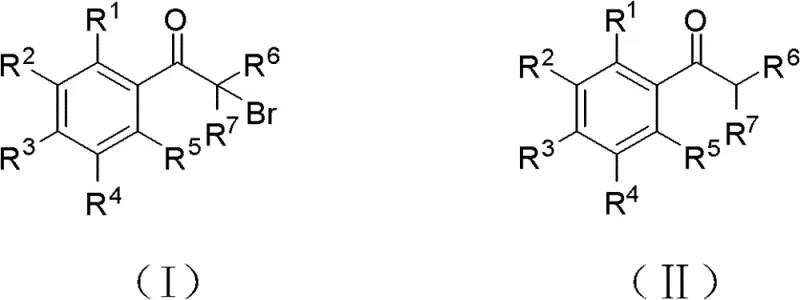
In stark contrast, the innovative methodology disclosed in the patent data utilizes hydrogen bromide as the brominating agent in conjunction with a copper nitrate catalyst and oxygen or air as the oxidant, all within a water solvent system. This paradigm shift eliminates the need for hazardous organic solvents during the reaction phase, replacing them with environmentally benign water which simplifies downstream processing and waste management. The reaction proceeds smoothly at temperatures ranging from 25°C to 100°C, demonstrating remarkable versatility across a broad spectrum of substrates including acetophenone derivatives and cyclic ketones. By employing air or oxygen, the process utilizes the cheapest and most abundant oxidants available, significantly reducing raw material costs compared to expensive stoichiometric oxidants like hydrogen peroxide or bromates. This approach not only enhances operational safety by avoiding liquid bromine but also ensures that the only byproduct is water, aligning perfectly with the principles of green chemistry and atom economy.
Mechanistic Insights into Copper-Catalyzed Oxidative Bromination
The core of this technological breakthrough lies in the intricate catalytic cycle mediated by copper species, which facilitates the activation of molecular oxygen to regenerate the active brominating species in situ. In this system, copper nitrate acts as a redox mediator, cycling between oxidation states to activate the hydrogen bromide and oxygen mixture, effectively generating an electrophilic bromine source without the need for handling elemental bromine directly. This mechanism allows for precise control over the bromination rate, minimizing over-bromination and side reactions that typically plague radical bromination processes. The presence of water as a solvent plays a crucial dual role; it acts as a heat sink to manage the exothermic nature of the bromination and facilitates the dissolution of the inorganic catalyst and acid, ensuring a homogeneous reaction environment that promotes consistent kinetics. For process chemists, understanding this mechanism is vital for optimizing catalyst loading, which the patent suggests can be as low as 0.001 molar equivalents, thereby reducing the burden of heavy metal removal in the final purification steps.
Impurity control is another critical aspect where this mechanistic understanding yields tangible benefits for pharmaceutical grade intermediates. Traditional methods often suffer from poly-bromination or ring bromination due to the high reactivity of free bromine, leading to complex impurity profiles that are difficult to separate. The controlled generation of the brominating species in this aqueous copper-catalyzed system favors mono-bromination at the alpha-position, significantly simplifying the impurity landscape. The patent data indicates that by adjusting the molar ratio of substrate to hydrogen bromide between 1:1.0 and 1:1.5, operators can fine-tune the reaction to maximize yield while suppressing side products. Furthermore, the use of auxiliary catalysts such as iron, potassium, or silver salts can be employed to further enhance selectivity and reaction rates for challenging substrates, providing a versatile toolkit for R&D teams to tailor the process for specific molecular architectures without compromising on purity or safety standards.
How to Synthesize Alpha-Bromo Aromatic Ketones Efficiently
Implementing this synthesis route requires careful attention to the specific reaction parameters outlined in the patent to ensure optimal yield and safety. The process is designed to be scalable, moving seamlessly from laboratory glassware to industrial reactors with minimal modification to the core chemistry. Operators must ensure that the hydrogen bromide is introduced either as a gas or, more conveniently, as an aqueous hydrobromic acid solution, typically at a concentration of 8 mol/L, to maintain the aqueous nature of the system. The reaction temperature should be maintained within the 50°C to 90°C range for most substrates to balance reaction rate and selectivity, although sensitive compounds may require lower temperatures near 25°C. For substrates with high melting points or poor water solubility, the patent allows for the addition of co-solvents like acetic acid or acetonitrile in minimal amounts (0-20 mL/g), ensuring that the green credentials of the process are largely preserved while maintaining reaction homogeneity.
- Prepare the reaction mixture by combining the aromatic ketone substrate, copper nitrate catalyst (0.001-0.2 molar equivalent), and aqueous hydrobromic acid in water.
- Introduce oxygen or air into the reactor at 1-10 atm pressure and heat the mixture to 25-100°C while stirring to initiate the oxidative bromination.
- Upon completion, cool the reaction, filter the precipitated solid product, wash with water to neutrality, and dry to obtain the pure alpha-bromo ketone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this aqueous oxidative bromination technology offers profound strategic advantages that extend far beyond simple chemical efficiency. The elimination of volatile organic solvents translates directly into substantial cost reductions related to solvent procurement, storage, and, most critically, waste disposal. Organic solvents represent a significant line item in the budget of any fine chemical manufacturing operation, and their associated disposal costs are often multiplicative due to environmental levies and treatment fees. By switching to water, the process effectively neutralizes these variable costs, stabilizing the cost base and protecting margins against fluctuations in the petrochemical market that drive solvent prices. Additionally, the use of air or oxygen as the oxidant removes the dependency on expensive, specialized oxidizing agents, further driving down the raw material cost per kilogram of the final product.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the material input list and waste management requirements. By replacing elemental bromine with hydrobromic acid and eliminating organic solvents, the facility reduces its exposure to hazardous material handling costs and insurance premiums associated with toxic reagents. The high atom economy means less raw material is wasted as byproduct, directly improving the mass balance and yield efficiency. Furthermore, the simplified workup procedure, often involving mere filtration and water washing for solid products, reduces energy consumption associated with distillation and solvent recovery, leading to a leaner, more cost-effective manufacturing operation that enhances overall competitiveness in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of commodity chemicals that are readily available in the global market. Hydrogen bromide, copper nitrate, and air are not subject to the same supply constraints or geopolitical volatility as specialized brominating reagents or high-purity organic solvents. This accessibility ensures continuous production capability even during market disruptions, reducing the risk of stockouts for critical pharmaceutical intermediates. The robustness of the reaction conditions, which tolerate atmospheric pressure and moderate temperatures, also means that the process can be executed in a wider range of manufacturing facilities without requiring specialized high-pressure or cryogenic infrastructure, thereby expanding the potential supplier base and reducing single-source dependency risks.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is inherently safer and more straightforward due to the absence of flammable organic vapors and highly corrosive liquid bromine. The aqueous system mitigates the risk of thermal runaway and fire, simplifying the safety engineering controls required for large-scale reactors. From a regulatory standpoint, the reduction in VOC emissions and hazardous waste generation aligns perfectly with increasingly stringent environmental regulations worldwide, such as REACH in Europe and EPA guidelines in the US. This proactive compliance posture minimizes the risk of regulatory fines or production shutdowns, ensuring long-term operational continuity and safeguarding the company's reputation as a responsible manufacturer of fine chemicals and pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of products manufactured via this route. The answers reflect the balance between theoretical efficiency and practical operational considerations observed in the provided examples.
Q: How does this aqueous method improve environmental compliance compared to traditional bromination?
A: Traditional methods utilize volatile organic solvents like acetic acid or dichloromethane and elemental bromine, generating significant hazardous waste. This patented process replaces organic solvents with water and uses hydrogen bromide with oxygen, resulting in water as the only byproduct, drastically reducing E-factors and wastewater treatment costs.
Q: What is the atom economy advantage of using HBr and Oxygen over elemental Bromine?
A: Conventional bromination with Br2 produces one equivalent of HBr waste for every equivalent of product, limiting atom utilization to 50%. This oxidative system utilizes HBr as the bromine source and Oxygen to regenerate the active species, theoretically allowing for higher atom efficiency and eliminating the stoichiometric acidic waste stream associated with direct bromination.
Q: Is the copper catalyst difficult to remove from the final pharmaceutical intermediate?
A: The process is designed for simplicity. For solid products, the protocol involves simple filtration and water washing, which effectively removes water-soluble copper salts. For liquid products or higher purity requirements, standard silica gel chromatography or crystallization techniques described in the patent ensure residual metal levels meet stringent pharmaceutical specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Bromo Aromatic Ketones Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is not just a regulatory requirement but a strategic imperative for modern pharmaceutical and agrochemical supply chains. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature are faithfully reproduced and optimized at an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities and residual metals, guaranteeing that every batch of alpha-bromo aromatic ketones meets the exacting standards required for downstream API synthesis. Our commitment to quality is matched by our dedication to sustainability, making us an ideal partner for companies looking to reduce their carbon footprint without compromising on product performance.
We invite you to engage with our technical procurement team to discuss how this advanced copper-catalyzed bromination technology can be integrated into your specific supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits tailored to your volume requirements. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven chemical expertise. Let us collaborate to build a more resilient, cost-effective, and sustainable future for your chemical manufacturing needs.
