Scalable One-Pot Synthesis of 3-Acyl Dihydroquinoline Derivatives for Advanced Pharmaceutical Applications
Scalable One-Pot Synthesis of 3-Acyl Dihydroquinoline Derivatives for Advanced Pharmaceutical Applications
The landscape of heterocyclic chemistry is continuously evolving, driven by the demand for more efficient and sustainable synthetic routes for bioactive scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN113234015B, which introduces a novel preparation method for 3-acyl dihydroquinoline derivatives. These compounds are not merely academic curiosities; they possess a wide array of potential biological activities, including anti-inflammatory, antifungal, antibacterial, and antitumor properties, making them highly valuable candidates for drug discovery programs. The patent outlines a robust copper-catalyzed multicomponent reaction that streamlines the construction of this complex core structure. By leveraging a one-pot strategy, this methodology addresses critical pain points in modern medicinal chemistry, such as step economy and waste generation. For R&D teams seeking reliable pharmaceutical intermediate suppliers, understanding the nuances of this technology is essential for securing a competitive edge in pipeline development.
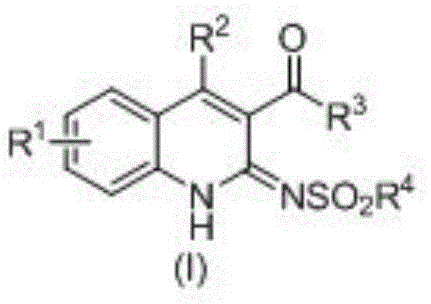
The structural versatility of these derivatives is evident in the general formula (I), where substituents R1 through R4 can be independently selected from a broad range of functional groups including alkyl, alkoxy, halogens, and phenyl rings. This modularity allows chemists to fine-tune the physicochemical properties of the final molecule to optimize bioavailability and target engagement. The ability to access such diverse chemical space through a unified synthetic platform is a hallmark of modern process chemistry. Furthermore, the high atom economy inherent in this transformation aligns perfectly with the principles of green chemistry, reducing the environmental footprint associated with traditional multi-step syntheses. As we delve deeper into the technical specifics, it becomes clear why this patent represents a pivotal shift in how these valuable intermediates are manufactured.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-acyl dihydroquinoline derivatives has been fraught with challenges that hinder both research throughput and commercial viability. Traditional routes often rely on the condensation of anthranoyl derivatives with beta-acyl compounds, a process that typically necessitates multiple synthetic steps to achieve the desired cyclization. These multi-step sequences invariably lead to cumulative yield losses, where the overall efficiency drops precipitously with each additional operation. Moreover, the starting materials required for these legacy methods are frequently difficult to obtain or require extensive pre-functionalization, driving up raw material costs and extending lead times. Side reactions are another persistent issue, often resulting in complex mixtures that are arduous to purify, thereby compromising the purity profile essential for pharmaceutical applications. The lack of a universal method with wide adaptability has long been a bottleneck, forcing process chemists to develop bespoke solutions for each specific analog, which is neither time-efficient nor cost-effective.
The Novel Approach
In stark contrast, the methodology disclosed in CN113234015B offers a paradigm shift through a direct, one-pot multicomponent reaction. This innovative approach combines an anthranilic acyl compound, a terminal alkynone, and a sulfonyl azide in a single vessel, facilitated by a copper catalyst and an organic ligand. The convergence of three distinct building blocks into the final heterocyclic scaffold eliminates the need for isolating unstable intermediates, significantly simplifying the operational workflow. The reaction proceeds with remarkable selectivity, predominantly yielding the target 3-acyl dihydroquinoline derivative with minimal byproduct formation. This high level of control is achieved through the careful optimization of reaction parameters, including solvent choice, temperature, and catalyst loading. For procurement managers focused on cost reduction in API manufacturing, this simplification translates directly into lower operational expenditures and reduced waste disposal costs. The robustness of this new route ensures that even complex analogs can be accessed with consistent quality, providing a stable foundation for supply chain planning.
Mechanistic Insights into Copper-Catalyzed Multicomponent Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to replicate or modify the process for specific analogs. The reaction initiates with a copper-catalyzed 1,3-dipolar cycloaddition between the terminal alkynone and the sulfonyl azide. This step generates a copper-triazole intermediate, which subsequently undergoes a ring-opening rearrangement to form a highly reactive ketene imine species. This transient intermediate, denoted as species A in the mechanistic scheme, serves as the electrophilic partner for the subsequent nucleophilic attack. The anthranilic acyl compound then engages in a nucleophilic addition with the ketene imine, forming a new carbon-nitrogen bond and establishing the core connectivity of the dihydroquinoline ring system. Finally, a dehydration condensation step aromatizes the system or stabilizes the double bond, delivering the final 3-acyl dihydroquinoline product. This cascade sequence is elegantly orchestrated by the copper center, which not only activates the alkyne but also facilitates the nitrogen extrusion and rearrangement steps.
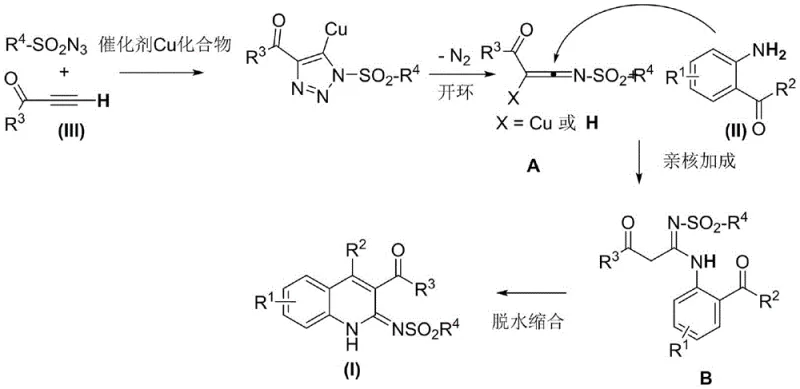
The control of impurities is intrinsically linked to the stability of the ketene imine intermediate and the kinetics of the nucleophilic addition. By maintaining optimal reaction temperatures between 25°C and 120°C, the process minimizes the decomposition of sensitive intermediates that could lead to polymeric byproducts or tar formation. The choice of ligand plays a pivotal role here; bulky and electron-rich ligands like TBTA stabilize the copper center, preventing premature catalyst deactivation and ensuring a steady flux of the reactive ketene imine. This controlled release mechanism prevents the accumulation of high concentrations of reactive species, which is a common cause of side reactions in multicomponent processes. Furthermore, the use of acetonitrile as the preferred solvent provides a polar environment that stabilizes the charged transition states without interfering with the catalytic cycle. For quality assurance teams, this mechanistic clarity means that process deviations can be quickly diagnosed and corrected, ensuring that the high-purity pharmaceutical intermediates produced meet stringent regulatory standards consistently.
How to Synthesize 3-Acyl Dihydroquinoline Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific protocols to maximize yield and purity. The general procedure involves charging a reaction vessel with the anthranilic acyl compound, the terminal alkynone, and the sulfonyl azide in a molar ratio typically ranging from 1:1:1 to 1:3:3. To this mixture, a catalytic amount of a copper salt, preferably cuprous iodide (CuI), and a stoichiometric amount of an organic ligand are added. Acetonitrile is the solvent of choice, utilized in a volume ratio of approximately 5 to 15 mL per mmol of substrate. The reaction mixture is then stirred at elevated temperatures, often between 60°C and 80°C, for a duration of 1 to 24 hours depending on the specific reactivity of the substrates. Upon completion, the workup is straightforward, involving extraction with ethyl acetate and water, drying over anhydrous sodium sulfate, and concentration. The crude product can be further purified via silica gel column chromatography or recrystallization to achieve analytical purity.
- Mix anthranilic acyl compound, terminal alkynone, sulfonyl azide, copper catalyst (e.g., CuI), and ligand (e.g., TBTA) in an organic solvent like acetonitrile.
- Stir the reaction mixture at temperatures between 25°C and 120°C for 1 to 24 hours to facilitate cycloaddition and rearrangement.
- Perform post-treatment via extraction with ethyl acetate and water, followed by drying, concentration, and purification via column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers profound advantages for supply chain resilience and cost management. The elimination of multi-step sequences drastically reduces the number of unit operations required, which in turn lowers the consumption of solvents, energy, and labor. This streamlined process flow enhances the overall throughput of the manufacturing facility, allowing for faster turnaround times on custom synthesis orders. For supply chain heads concerned with reducing lead time for high-purity pharmaceutical intermediates, the one-pot nature of this reaction is a game-changer. It mitigates the risk of bottlenecks associated with intermediate isolation and purification, ensuring a smoother flow of materials from raw input to finished goods. Additionally, the use of commodity chemicals like copper iodide and acetonitrile ensures that the supply of catalysts and solvents remains stable and unaffected by geopolitical fluctuations that often impact rare earth metals or exotic reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by its high atom economy and simplified workup. By avoiding the use of expensive transition metal catalysts like palladium or rhodium, and instead utilizing abundant copper salts, the direct material costs are significantly lowered. Furthermore, the high yields reported, often exceeding 90% and reaching up to 98% in optimized examples, mean that less raw material is wasted per kilogram of product produced. The simplified purification protocol, which often requires only crystallization or basic chromatography, reduces the consumption of silica gel and eluents, further driving down the cost of goods sold. These cumulative savings allow for more competitive pricing strategies without compromising on margin, making the final API or intermediate more attractive to downstream customers.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply security. The tolerance of the method to various functional groups means that a single set of operating parameters can be applied to a wide library of analogs, reducing the need for extensive process re-development for each new project. This standardization simplifies inventory management and operator training. Moreover, the starting materials—anthranilic acyl compounds, alkynones, and sulfonyl azides—are commercially available from multiple global suppliers, reducing the risk of single-source dependency. This diversification of the supply base ensures continuity of supply even in the face of market disruptions, providing peace of mind to procurement managers tasked with securing long-term contracts for critical drug substances.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram scale is facilitated by the exothermic nature of the cycloaddition and the stability of the intermediates. The process does not require cryogenic conditions or high-pressure equipment, allowing it to be run in standard glass-lined or stainless steel reactors found in most multipurpose plants. From an environmental standpoint, the high atom economy minimizes the generation of hazardous waste streams. The use of acetonitrile, while requiring proper recovery systems, is well-established in the industry with mature recycling technologies. The reduction in solvent usage per kilogram of product, coupled with the avoidance of toxic heavy metals, simplifies wastewater treatment and helps facilities meet increasingly stringent environmental regulations. This alignment with sustainability goals is increasingly becoming a prerequisite for doing business with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation. Understanding these nuances helps stakeholders make informed decisions about integrating this chemistry into their existing portfolios. Whether you are evaluating the feasibility of a new target or assessing the scalability of a current process, these answers provide a solid foundation for further discussion with technical experts.
Q: What are the primary advantages of this new synthesis route compared to conventional methods?
A: Unlike conventional multi-step syntheses that suffer from low yields and difficult raw material sourcing, this patented one-pot method utilizes readily available starting materials to achieve high atom economy and single product selectivity with yields up to 98%.
Q: Which catalyst system provides the optimal yield for this transformation?
A: Experimental data indicates that cuprous iodide (CuI) combined with the ligand tris[(1-benzyl-1H-1,2,3-triazol-4-yl)methyl]amine (TBTA) in acetonitrile provides the highest catalytic efficiency and product purity.
Q: Can this process be scaled for industrial production of drug intermediates?
A: Yes, the process operates under relatively mild conditions (25-120°C) and uses standard organic solvents, making it highly suitable for commercial scale-up while maintaining high purity specifications required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Acyl Dihydroquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN113234015B for the development of next-generation therapeutics. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. We are committed to delivering stringent purity specifications through our rigorous QC labs, which are equipped with state-of-the-art analytical instrumentation to verify identity and potency. Our capability to handle complex multicomponent reactions allows us to offer flexible manufacturing solutions tailored to your specific timeline and budget requirements. By partnering with us, you gain access to a supply chain that prioritizes quality, consistency, and regulatory compliance.
We invite you to contact our technical procurement team to discuss your specific needs for 3-acyl dihydroquinoline derivatives. We can provide a Customized Cost-Saving Analysis that details how implementing this copper-catalyzed route can optimize your overall production budget. Please reach out to request specific COA data for our reference standards or to schedule a consultation regarding route feasibility assessments for your proprietary analogs. Let us help you accelerate your drug development timeline with our reliable and scalable manufacturing capabilities.
