Scalable Catalyst-Free Synthesis of Bioactive Ether-Substituted 2-Pyrrolidone Intermediates
Scalable Catalyst-Free Synthesis of Bioactive Ether-Substituted 2-Pyrrolidone Intermediates
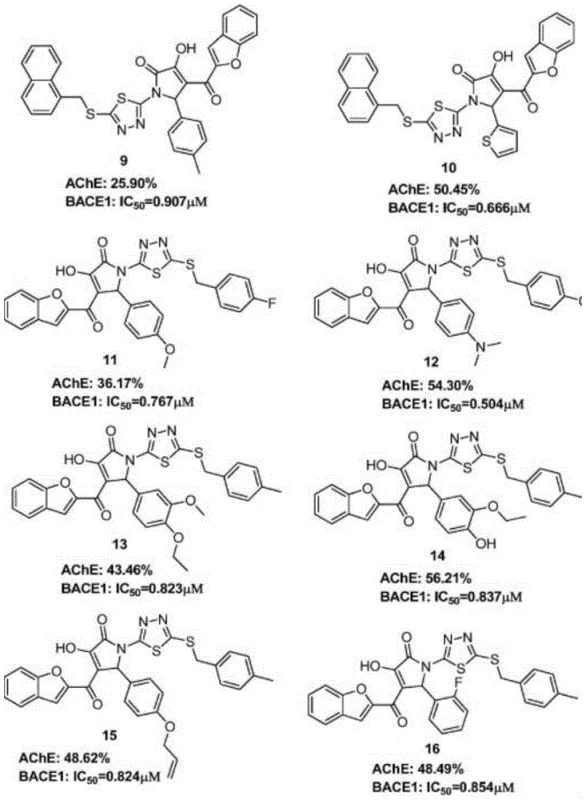
The pharmaceutical industry continuously seeks efficient pathways to construct complex heterocyclic scaffolds, particularly those exhibiting potent biological activity against neurodegenerative targets. Patent CN111393419B introduces a groundbreaking preparation method for ether-substituted 2-pyrrolidone compounds, a structural motif increasingly recognized for its potential as a bidirectional inhibitor for acetylcholinesterase (AChE) and beta-secretase (BACE1). As illustrated in the structural diversity of bioactive pyrrolidones, these compounds serve as critical building blocks in medicinal chemistry. The disclosed technology leverages a novel radical cyclization strategy that bypasses the need for transition metal catalysts, offering a streamlined route to high-value intermediates. This innovation addresses long-standing challenges in synthetic efficiency and environmental compliance, positioning it as a vital asset for the development of next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing pyrrolidone rings often rely heavily on transition metal catalysis or pre-functionalized starting materials, which introduce significant bottlenecks in process development. Conventional methods typically require expensive palladium or copper catalysts, specialized ligands, and strictly anhydrous conditions, all of which escalate manufacturing costs and complicate supply chain logistics. Furthermore, metal-catalyzed processes frequently suffer from issues related to heavy metal residue, necessitating rigorous and costly purification steps to meet pharmaceutical grade standards. The reliance on stoichiometric bases and harsh reaction conditions also limits the functional group tolerance, often leading to side reactions and reduced atom economy. These factors collectively hinder the rapid scale-up and commercial viability of pyrrolidone-based drug candidates, creating an urgent demand for more robust and economical synthetic alternatives.
The Novel Approach
In stark contrast to legacy technologies, the method described in patent CN111393419B utilizes a catalyst-free radical cyclization of 1,6-diene compounds with ether solvents. This approach fundamentally shifts the paradigm by employing the ether solvent not merely as a medium but as an active reactant, thereby enhancing atom economy and reducing waste. The reaction proceeds under an air atmosphere at elevated temperatures using tert-butyl peroxide as a benign oxidant, eliminating the need for inert gas protection or sensitive catalysts. As shown in the general reaction scheme, this strategy enables the direct construction of two carbon-carbon bonds in a single step, significantly shortening the synthetic sequence. The simplicity of the reaction setup, combined with the use of commercially available and stable reagents, offers a distinct advantage for large-scale manufacturing, ensuring consistent quality and reduced operational complexity.
Mechanistic Insights into TBHP-Mediated Radical Cyclization
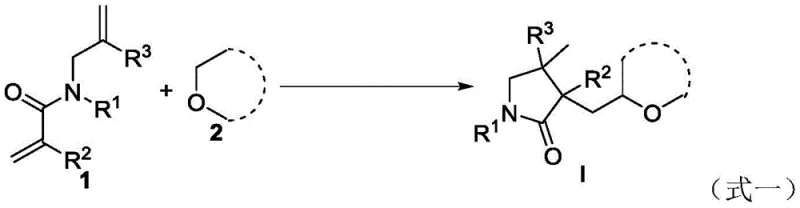
The core of this innovative synthesis lies in its unique radical mechanism, which is initiated by the homolytic cleavage of the oxidant to generate reactive radical species. Detailed mechanistic studies, including control experiments with radical scavengers like TEMPO and BHT, confirm that the reaction proceeds through a free-radical pathway. In the absence of these scavengers, the reaction yields the target pyrrolidone efficiently, whereas their presence drastically suppresses product formation, validating the radical nature of the transformation. This mechanistic understanding is crucial for R&D teams aiming to optimize reaction parameters and expand the substrate scope. The ability to generate radicals selectively at the alpha-position of the ether oxygen allows for precise functionalization without affecting other sensitive groups on the diene substrate, showcasing high chemoselectivity.
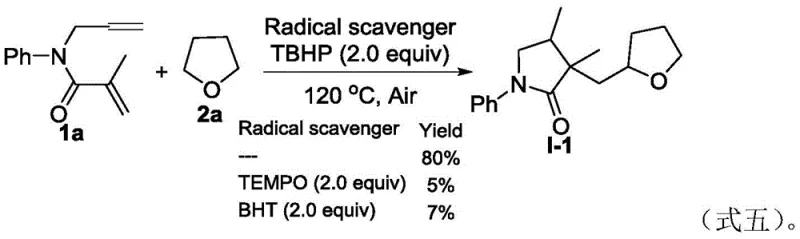
Further elucidation of the catalytic cycle reveals a sophisticated sequence of radical addition and intramolecular cyclization events. Initially, the tert-butoxy radical abstracts a hydrogen atom from the alpha-carbon of the ether solvent, generating an alpha-oxy radical intermediate. This nucleophilic radical then adds selectively to the electron-deficient double bond of the 1,6-diene substrate, forming a new carbon-carbon bond. Subsequent intramolecular radical cyclization closes the five-membered lactam ring, followed by hydrogen atom abstraction from the solvent to terminate the cycle and release the final product. This elegant cascade, depicted in the proposed mechanism, highlights the efficiency of the process in constructing complex cyclic skeletons from simple linear precursors. The absence of external catalysts ensures that the reaction profile is governed solely by the intrinsic reactivity of the radicals, minimizing side reactions and maximizing yield.
How to Synthesize Ether-Substituted 2-Pyrrolidone Efficiently
Optimization studies detailed in the patent demonstrate that reaction conditions can be finely tuned to achieve superior yields and selectivity. Experimental data indicates that using tert-butyl peroxide (TBHP) as the oxidant at a temperature of 120°C provides the optimal balance between reaction rate and product stability. The molar ratio of the diene substrate to the oxidant is a critical parameter, with a 1:2 ratio proving most effective for driving the reaction to completion while minimizing decomposition. Various ether solvents, including tetrahydrofuran, tetrahydropyran, and 1,4-dioxane, have been successfully employed, showcasing the versatility of the method. For process chemists looking to implement this technology, the standardized protocol involves simple mixing of reagents in a Schlenk flask, heating under air, and standard aqueous workup. The detailed standardized synthesis steps are provided in the guide below.
- Combine 1,6-diene substrate, ether solvent (acting as reactant), and tert-butyl peroxide oxidant in a reaction vessel.
- Heat the mixture to 110-130°C under an air atmosphere to initiate radical generation and cyclization.
- Upon completion, perform standard extraction with ethyl acetate and purify via column chromatography to isolate the target pyrrolidone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this catalyst-free methodology offers transformative benefits that directly impact the bottom line and operational resilience. By eliminating the dependency on precious metal catalysts and specialized ligands, manufacturers can significantly reduce raw material costs and mitigate supply risks associated with scarce resources. The dual role of the ether solvent as both reactant and medium further simplifies the bill of materials, reducing the volume of chemicals required and lowering waste disposal costs. Additionally, the operation under air atmosphere removes the need for expensive inert gas infrastructure, making the process adaptable to existing manufacturing facilities without major capital investment. These factors collectively contribute to a more agile and cost-effective supply chain capable of responding rapidly to market demands.
- Cost Reduction in Manufacturing: The exclusion of transition metal catalysts and bases eliminates the need for costly metal scavenging steps and complex purification protocols, leading to substantial savings in downstream processing. The use of bulk commodity chemicals like tert-butyl peroxide and common ether solvents ensures stable pricing and easy sourcing, avoiding the volatility associated with specialty reagents. Furthermore, the high atom economy of the reaction minimizes raw material waste, aligning with green chemistry principles and reducing the overall cost of goods sold. This economic efficiency makes the production of high-purity pharmaceutical intermediates more viable and competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, specifically the tolerance to air and moisture, enhances process reliability and reduces the risk of batch failures due to environmental fluctuations. Sourcing of key reagents is straightforward, as they are widely available from multiple suppliers, preventing single-source bottlenecks. The simplified workflow, characterized by fewer unit operations and shorter reaction times, accelerates production cycles and improves throughput. This reliability is paramount for maintaining continuous supply to downstream drug formulation units, ensuring that critical timelines for clinical and commercial programs are met without disruption.
- Scalability and Environmental Compliance: The inherent safety and simplicity of the radical cyclization process facilitate seamless scale-up from laboratory to pilot and commercial scales. The absence of toxic heavy metals simplifies regulatory compliance and environmental permitting, reducing the administrative burden on EHS teams. Waste generation is minimized due to the high selectivity and efficiency of the reaction, supporting sustainability goals and reducing the carbon footprint of manufacturing operations. This scalability ensures that the technology can meet the growing demand for ether-substituted 2-pyrrolidone derivatives as they advance through the drug development pipeline.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this radical cyclization technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation. Understanding these aspects is essential for stakeholders evaluating the feasibility of adopting this method for their specific production needs. The answers provide clarity on reaction mechanisms, scalability potential, and the strategic advantages offered by this novel synthetic route.
Q: What is the primary advantage of this synthesis method over traditional metal-catalyzed routes?
A: The primary advantage is the elimination of expensive transition metal catalysts and bases. This method utilizes a radical mechanism initiated by tert-butyl peroxide in an ether solvent, which serves a dual role as both reactant and medium, significantly simplifying purification and reducing heavy metal contamination risks.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process is highly suitable for industrial scale-up. It operates under air atmosphere without stringent inert gas requirements, uses commercially available and stable oxidants, and features a simple workup procedure involving extraction and chromatography, ensuring high operational stability and safety.
Q: What specific biological activities are associated with the resulting pyrrolidone compounds?
A: The resulting ether-substituted 2-pyrrolidone derivatives are structurally related to compounds known for inhibiting acetylcholinesterase (AChE) and beta-secretase (BACE1). These targets are critical in the development of therapeutics for neurodegenerative disorders such as Alzheimer's disease.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ether-Substituted 2-Pyrrolidone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN111393419B for accelerating drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are translated into reliable commercial supplies. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ether-substituted 2-pyrrolidone meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of radical chemistry safely and efficiently, delivering high-quality intermediates that support your critical R&D and manufacturing objectives.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can enhance your supply chain efficiency. Let us be your partner in bringing innovative therapies to market faster and more cost-effectively.
