Advanced Metal-Free Synthesis of Ortho-Halogenated Arylamines for Pharmaceutical Intermediates
The landscape of aromatic halogenation is undergoing a significant transformation, driven by the urgent need for more sustainable and cost-effective manufacturing processes in the fine chemical industry. Patent CN113200873B introduces a groundbreaking methodology for the synthesis of ortho-position halogenated arylamine compounds, specifically targeting the structural motif represented by formula (III). This innovation addresses critical bottlenecks in the production of key pharmaceutical intermediates and agrochemical building blocks by utilizing cheap and easily obtained dihalogen sulfoxides to react with aryl hydroxylamine compounds. Unlike traditional methods that often struggle with regioselectivity or require harsh conditions, this novel approach realizes the high-efficiency synthesis of o-haloarylamines under remarkably mild conditions. The technology boasts good universality and can well tolerate various functional groups, making it an invaluable tool for the commercial scale-up of complex pharmaceutical intermediates. By shifting away from precious metal dependency, this process offers a robust pathway for reducing lead time for high-purity intermediates while maintaining stringent quality standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of halogen atoms onto aromatic rings has relied heavily on electrophilic aromatic halogenation or transition metal-catalyzed directed C-H activation. As illustrated in the general reaction schemes for electrophilic halogenation, these classical strategies typically utilize liquid bromine, chlorine, or N-halosuccinimides, which are often highly toxic, hazardous, and difficult to handle on a large industrial scale.
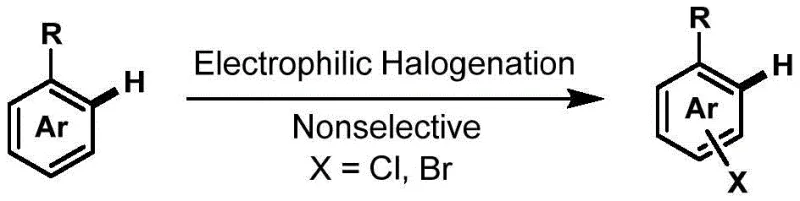
Furthermore, these conventional electrophilic methods generally suffer from poor regioselectivity, frequently producing undesirable by-products such as polyhalogenated species or various regioisomers that are notoriously difficult to separate, thereby driving up purification costs and reducing overall yield. Alternatively, transition metal-catalyzed approaches, while offering better selectivity, typically require the use of expensive catalysts such as palladium or rhodium, along with stoichiometric oxidants and high-temperature heating conditions. These factors not only inflate the raw material expenditure but also introduce complex downstream purification challenges regarding residual metal limits, which is a critical concern for reliable pharmaceutical intermediate suppliers aiming to meet strict GMP standards.
The Novel Approach
In stark contrast to these legacy technologies, the invention disclosed in CN113200873B utilizes a transition metal-free rearrangement strategy that leverages the unique reactivity of aryl hydroxylamines with dihalo sulfoxides (SOCl₂ or SOBr₂). This method achieves exceptional ortho-selectivity without the need for external directing groups or expensive metal catalysts. The core reaction involves the interaction of compound (I), an aryl hydroxylamine derivative, with compound (II), a dihalo sulfoxide, to directly yield the target ortho-halogenated product (III).
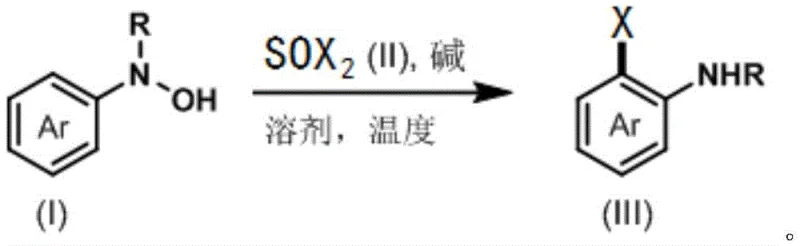
This novel approach operates under mild conditions, typically ranging from -40°C to 30°C, with a preference for 0°C, which significantly enhances safety profiles and energy efficiency compared to high-temperature alternatives. The process demonstrates remarkable versatility, successfully tolerating a wide array of substituents including fluorine, chlorine, bromine, alkyl, alkenyl, ester groups, nitro groups, and even complex heteroaryl systems. This level of functional group compatibility ensures that the synthesis of high-purity OLED materials or drug candidates containing sensitive moieties can be achieved without compromising the integrity of the molecular scaffold, representing a substantial advancement in cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Metal-Free Ortho-Halogenation
The mechanistic pathway of this transformation represents a fascinating departure from standard electrophilic substitution. The reaction initiates with the activation of the aryl hydroxylamine by the dihalo sulfoxide, generating a highly reactive intermediate that facilitates an intramolecular rearrangement. This rearrangement effectively transfers the halogen atom from the sulfur center to the ortho-position of the aromatic ring relative to the nitrogen substituent. The absence of transition metals simplifies the catalytic cycle, removing the need for oxidative addition and reductive elimination steps that often limit turnover numbers in palladium chemistry. Instead, the driving force is derived from the thermodynamic stability of the resulting sulfur-oxygen bonds and the restoration of aromaticity in the final product. This mechanism allows for the efficient conversion of diverse substrates, including substituted naphthyl, phenyl, and heteroaryl compounds, into their corresponding ortho-halogenated derivatives with excellent yields.
From an impurity control perspective, this mechanism offers distinct advantages. Because the reaction is highly regioselective for the ortho-position, the formation of para- or meta-isomers is minimized, simplifying the impurity profile of the crude reaction mixture. Additionally, the by-products generated are primarily sulfur-based species which are generally easier to remove via aqueous workup or standard chromatography compared to heavy metal residues. The patent data highlights successful applications on complex molecules, such as steroid derivatives and sugar-protected phenols, demonstrating that the steric and electronic demands of the substrate do not hinder the rearrangement. This robustness is crucial for ensuring batch-to-batch consistency, a key metric for any procurement manager evaluating a new supplier for critical API intermediates.
How to Synthesize Ortho-Halogenated Arylamines Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible roadmap for laboratory and pilot-scale production. The process begins with the preparation of the aryl hydroxylamine precursor, which can be readily obtained by reducing the corresponding nitro compound followed by acylation. Once the hydroxylamine is secured, the halogenation step is straightforward: the substrate is dissolved in a suitable solvent, cooled to 0°C, and treated with the dihalo sulfoxide reagent. The reaction progress is easily monitored by TLC, and upon completion, the product is isolated via concentration and column chromatography. For detailed operational parameters, stoichiometry, and specific workup procedures tailored to your specific substrate, please refer to the standardized synthesis guide below.
- Preparation of Aryl Hydroxylamine: Reduce the corresponding nitro compound using hydrazine hydrate and Rh/C catalyst, followed by acylation with acid chloride to form the N-substituted aryl hydroxylamine.
- Halogenation Reaction: Dissolve the aryl hydroxylamine in an appropriate solvent (THF for chlorination, DCM for bromination), cool to 0°C, and add dihalo sulfoxide (SOCl2 or SOBr2) dropwise.
- Workup and Purification: Stir the reaction mixture, monitor by TLC, concentrate under reduced pressure, and purify the crude product via column chromatography to obtain the target ortho-haloarylamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free halogenation technology translates into tangible strategic benefits beyond mere chemical curiosity. The elimination of precious metal catalysts fundamentally alters the cost structure of the synthesis, removing one of the most volatile and expensive line items from the bill of materials. Furthermore, the simplified purification process reduces the consumption of silica gel and solvents associated with removing metal scavengers, leading to significant waste reduction and lower disposal costs. This streamlined workflow enhances the overall agility of the supply chain, allowing for faster response times to market demands for key intermediates.
- Cost Reduction in Manufacturing: The most immediate impact is the drastic reduction in raw material costs associated with catalysts. By replacing palladium or rhodium systems with inexpensive dihalo sulfoxides and avoiding the need for specialized ligands, the direct material cost is significantly lowered. Moreover, the absence of heavy metals eliminates the need for expensive metal scavenging resins and the rigorous analytical testing required to certify low ppm levels of residual metals, which further drives down the cost of goods sold (COGS) and improves margin potential for high-volume production.
- Enhanced Supply Chain Reliability: Relying on transition metal catalysts often exposes supply chains to geopolitical risks and availability fluctuations, as seen with palladium markets. This new method utilizes commodity chemicals like thionyl chloride or sulfuryl chloride derivatives, which are produced globally in massive quantities, ensuring a stable and continuous supply of reagents. The robustness of the reaction conditions also means that production is less susceptible to minor variations in utility supplies or equipment performance, guaranteeing consistent delivery schedules for downstream customers relying on just-in-time inventory models.
- Scalability and Environmental Compliance: The mild reaction temperatures (0°C to room temperature) and the use of common solvents like DCM or THF make this process highly amenable to scale-up in standard glass-lined or stainless steel reactors without requiring specialized high-pressure or high-temperature equipment. From an environmental standpoint, the reduction in heavy metal waste aligns perfectly with increasingly stringent global environmental regulations, facilitating easier permitting and reducing the carbon footprint of the manufacturing process, which is a growing priority for sustainability-focused corporate buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ortho-halogenation technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing a realistic overview of the method's capabilities and limitations for potential partners considering technology transfer or contract manufacturing services.
Q: What are the primary advantages of this metal-free halogenation method over traditional palladium-catalyzed routes?
A: This method eliminates the need for expensive transition metal catalysts like palladium or rhodium, significantly reducing raw material costs and removing the regulatory burden of residual heavy metal testing in pharmaceutical intermediates.
Q: Does this synthesis strategy tolerate sensitive functional groups?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully accommodating esters, silyl groups, nitro groups, and complex natural product scaffolds like steroids without degradation.
Q: What are the optimal reaction conditions for chlorination versus bromination?
A: For chlorination, the optimal conditions involve using THF as the solvent with sodium carbonate as a base at 0°C. For bromination, the reaction proceeds best in DCM without the addition of a base, warming from 0°C to room temperature.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ortho-Halogenated Arylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for the next generation of pharmaceutical and agrochemical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace impurities and residual solvents, guaranteeing that every batch meets the highest international standards for quality and safety.
We invite you to collaborate with us to leverage this advanced chemistry for your specific project needs. Whether you require custom synthesis of complex heterocyclic intermediates or large-scale production of halogenated building blocks, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your molecule. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can optimize your supply chain and accelerate your time to market.
