Advanced Organocatalytic Synthesis of Chiral Thiopyranoindolobenzothiophene Sulfones for Drug Discovery
The pharmaceutical industry is constantly seeking novel scaffolds that can serve as privileged structures for drug discovery, particularly those capable of interacting with complex biological targets. Patent CN111606924A introduces a groundbreaking methodology for the synthesis of chiral thiopyranoindolobenzothiophene sulfone derivatives, a class of compounds that had previously remained unexplored in scientific literature. This innovation addresses a critical gap in organic synthesis by providing a robust, enantioselective route to construct a highly functionalized polycyclic system containing three contiguous chiral centers. The significance of this development lies not only in the novelty of the chemical structure but also in the efficiency of the synthetic protocol, which utilizes mild organocatalytic conditions to achieve exceptional stereocontrol. For research and development teams focused on expanding their compound libraries for high-throughput screening, access to such diverse and stereochemically defined heterocycles is invaluable. The ability to introduce multiple functional groups onto this rigid framework allows for extensive derivatization, making it a versatile platform for generating candidate molecules for new drug development.
Furthermore, the integration of the sulfone moiety within this fused ring system is strategically important, as sulfones are well-known pharmacophores found in numerous bioactive agents, including beta-lactamase inhibitors. By combining the indole and benzothiophene sulfone units into a single, rigid architecture, this technology offers a unique spatial arrangement of atoms that may interact with biological targets in ways that simpler, flexible molecules cannot. This structural complexity, achieved through a relatively straightforward synthetic operation, represents a significant leap forward in the construction of complex pharmaceutical intermediates. The method described eliminates the need for harsh reaction conditions or expensive transition metal catalysts, thereby aligning with modern green chemistry principles while delivering high-purity products suitable for sensitive biological assays.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the disclosure of this patent, the synthesis of thiopyranoindolobenzothiophene sulfone derivatives was virtually non-existent, representing a significant void in the chemical space available for medicinal chemistry exploration. Conventional methods for constructing similar polycyclic sulfur-containing heterocycles often rely on multi-step sequences involving harsh reagents, high temperatures, or stoichiometric amounts of toxic heavy metals, which can complicate purification and introduce hazardous impurities into the final product. Traditional approaches to establishing multiple chiral centers typically require chiral pool starting materials or resolution of racemic mixtures, both of which suffer from inherent limitations such as limited structural diversity and a maximum theoretical yield of 50% for the desired enantiomer. These inefficiencies create substantial bottlenecks in the supply chain for high-purity pharmaceutical intermediates, increasing both the cost and the lead time required to produce material for preclinical studies. Additionally, the lack of reported methods for this specific scaffold meant that researchers were unable to explore the potential biological activity of this unique fusion of indole and sulfone chemistries, limiting the scope of drug discovery efforts in this area.
The Novel Approach
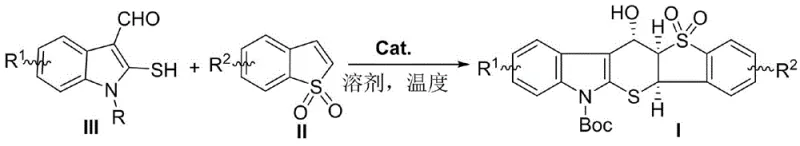
The novel approach detailed in the patent overcomes these historical challenges by employing a direct, one-pot organocatalytic cascade reaction that efficiently assembles the complex tricyclic core from simple, commercially available starting materials. By utilizing 1,1-benzothiophene dioxide and 2-mercaptoindole-3-carbaldehyde reagents, the method constructs the thiopyranoindole framework with remarkable atom economy and step efficiency. The use of chiral polyhydrogen bond tertiary amine-thiourea catalysts enables precise control over the stereochemical outcome of the reaction, generating products with three contiguous stereocenters in a single operation. This strategy bypasses the need for protecting group manipulations or intermediate isolations that typically plague multi-step syntheses, thereby drastically simplifying the overall process. The reaction proceeds under mild conditions, typically between -10°C and 25°C, which minimizes energy consumption and reduces the risk of thermal degradation of sensitive functional groups. This streamlined methodology not only accelerates the generation of novel chemical entities but also ensures that the resulting intermediates are produced with the high levels of purity and stereochemical integrity required for rigorous pharmaceutical testing.
Mechanistic Insights into Chiral Thiourea-Catalyzed Cyclization
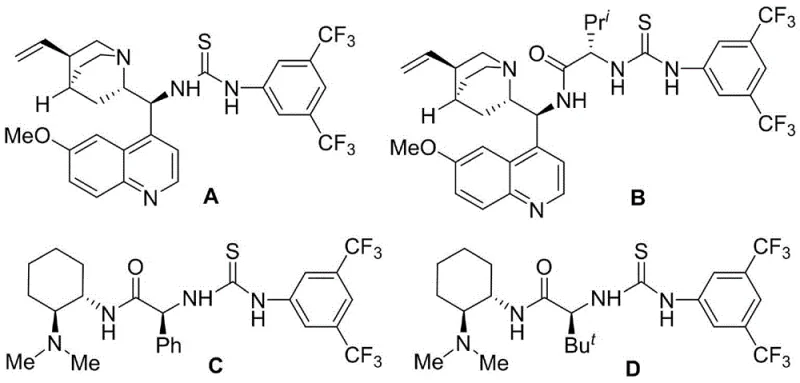
The success of this synthetic transformation hinges on the sophisticated design of the chiral organocatalysts employed, specifically the chiral polyhydrogen bond tertiary amine-thiourea derivatives. These catalysts function by simultaneously activating both the electrophilic and nucleophilic components of the reaction through a network of hydrogen bonding interactions. The thiourea moiety typically acts as a dual hydrogen bond donor, coordinating with the electron-rich oxygen atoms of the sulfone or the carbonyl group of the aldehyde, thereby lowering the energy barrier for nucleophilic attack. Concurrently, the tertiary amine portion of the catalyst can interact with the thiol group of the indole reagent, enhancing its nucleophilicity and directing its approach to the electrophile. This bifunctional activation creates a highly organized transition state within the chiral environment of the catalyst, which effectively shields one face of the reacting species while exposing the other. This steric differentiation is the key driver for the observed high enantioselectivity and diastereoselectivity, ensuring that the new carbon-carbon and carbon-sulfur bonds are formed with the correct spatial orientation.
Impurity control in this process is inherently managed by the high specificity of the organocatalytic cycle. Unlike transition metal catalysis, which can sometimes lead to side reactions such as over-reduction or metal-mediated decomposition, organocatalysis tends to be highly chemoselective. The mild reaction conditions further suppress the formation of thermal byproducts or polymerization of the reactive aldehyde intermediates. The patent data indicates that the reaction tolerates a wide range of substituents on both the indole and benzothiophene rings, including electron-withdrawing halogens and electron-donating alkoxy groups, without significant loss in yield or selectivity. This robustness suggests that the catalytic cycle is resilient to electronic variations in the substrate, likely due to the strength and directionality of the hydrogen bonding interactions that dominate the transition state. Consequently, the crude reaction mixtures are relatively clean, facilitating easier downstream purification and reducing the burden on quality control laboratories to identify and quantify complex impurity profiles.
How to Synthesize Chiral Thiopyranoindolobenzothiophene Sulfones Efficiently
The synthesis of these valuable intermediates is designed to be operationally simple, requiring standard laboratory equipment and readily available reagents. The process begins with the dissolution of the solid starting materials in a suitable organic solvent, followed by the addition of the chiral catalyst at controlled temperatures. The reaction is allowed to proceed with stirring for a defined period, after which the product is isolated through standard workup procedures such as column chromatography. This simplicity makes the method highly attractive for both small-scale discovery chemistry and larger-scale process development. For detailed procedural specifics regarding reagent ratios, solvent choices, and purification parameters, please refer to the standardized synthesis steps outlined below.
- Dissolve 1,1-benzothiophene dioxide and 2-mercaptoindole-3-carbaldehyde reagent in an organic solvent such as toluene or dichloromethane.
- Add a chiral polyhydrogen bond tertiary amine-thiourea catalyst (preferably Catalyst C or D) to the reaction mixture at a loading of approximately 1 mol%.
- Stir the reaction at temperatures between -10°C and 25°C for 48 to 96 hours, then separate and purify the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers distinct advantages that translate directly into operational efficiency and cost optimization for pharmaceutical manufacturers. The reliance on organocatalysts rather than precious metal complexes eliminates the need for expensive metal scavenging steps and reduces the regulatory burden associated with residual metal limits in active pharmaceutical ingredients. This shift significantly simplifies the manufacturing workflow, allowing for faster turnaround times from synthesis to final release testing. Furthermore, the use of commodity chemicals as starting materials ensures a stable and reliable supply chain, mitigating the risks associated with sourcing specialized or scarce reagents. The robustness of the reaction conditions means that the process is less susceptible to variability, leading to more consistent batch-to-batch quality and reducing the incidence of failed batches that can disrupt production schedules.
- Cost Reduction in Manufacturing: The implementation of this organocatalytic route drives down manufacturing costs through several mechanisms, primarily by eliminating the need for costly transition metal catalysts and the associated purification infrastructure. The low catalyst loading, which can be as low as 1 mol%, combined with the high yields reported (often exceeding 95%), ensures that raw material utilization is maximized. Additionally, the mild temperature requirements reduce energy consumption for heating or cooling, contributing to lower utility costs over the lifecycle of the product. The simplified purification process, necessitated by the clean reaction profile, further reduces the consumption of solvents and stationary phases, resulting in substantial cost savings in waste disposal and material procurement.
- Enhanced Supply Chain Reliability: The availability of starting materials and the simplicity of the synthetic protocol enhance the overall reliability of the supply chain for these complex intermediates. Since the reagents are structurally simple and widely available, the risk of supply disruption due to raw material shortages is minimized. The scalability of the process, as evidenced by successful gram-scale experiments in the patent data, suggests that transitioning from laboratory synthesis to commercial production can be achieved with minimal process re-engineering. This ease of scale-up ensures that supply can be rapidly ramped up to meet clinical trial demands or commercial launch requirements without the long lead times typically associated with developing complex asymmetric syntheses.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is favorable, aligning with increasingly stringent global regulations on chemical manufacturing. The absence of heavy metals reduces the toxicity of the waste stream, simplifying effluent treatment and disposal compliance. The high atom economy of the cascade reaction means that less waste is generated per unit of product, supporting sustainability goals. Moreover, the ability to run the reaction at near-ambient temperatures reduces the carbon footprint associated with energy-intensive heating or cryogenic cooling. These factors collectively make the process highly scalable and environmentally compliant, reducing the risk of regulatory delays and enhancing the long-term viability of the manufacturing route.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these chiral sulfone derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners and licensees. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing drug discovery pipelines or manufacturing processes.
Q: What level of stereoselectivity can be achieved with this synthesis method?
A: The method achieves excellent stereoselectivity, with diastereomeric ratios (dr) greater than 20:1 and enantiomeric excess (ee) values up to 99% when using optimized chiral thiourea catalysts.
Q: Is this synthetic route scalable for commercial production?
A: Yes, the patent demonstrates successful scale-up experiments (e.g., 2.5 mmol scale) maintaining high yields (98%) and stereoselectivity, indicating strong potential for commercial scale-up of complex pharmaceutical intermediates.
Q: Which catalysts provide the best results for this transformation?
A: Chiral polyhydrogen bond tertiary amine-thiourea catalysts, specifically those designated as Catalyst C and Catalyst D in the patent data, provide superior yields and stereocontrol compared to other variants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Thiopyranoindolobenzothiophene Sulfone Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and process development, possessing the technical expertise to bring complex molecules like chiral thiopyranoindolobenzothiophene sulfones from the laboratory bench to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of global pharmaceutical clients. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch meets the highest standards of quality and stereochemical integrity. Our commitment to excellence ensures that your supply of critical intermediates remains uninterrupted, allowing you to focus on advancing your drug candidates through the development pipeline.
We invite you to collaborate with us to leverage this innovative synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your time to market. Let us be your partner in transforming cutting-edge chemical research into commercial success.
