Advanced Synthesis of Amosulalol Hydrochloride for Commercial Pharmaceutical Manufacturing
Advanced Synthesis of Amosulalol Hydrochloride for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry constantly seeks robust, scalable, and cost-effective pathways for producing critical antihypertensive agents. Patent CN102050766B presents a groundbreaking synthetic methodology for Amosulalol Hydrochloride, a potent non-selective beta-receptor and selective alpha-1 receptor blocker used extensively in managing essential hypertension and pheochromocytoma. This technical disclosure outlines a superior route that begins with the readily available 4-methylacetophenone, navigating through a series of optimized transformations including nitration, reduction, diazotization, and sulfonation to achieve the target molecule with high purity. The structural integrity of the final product, as depicted below, is crucial for its pharmacological efficacy in reducing peripheral vascular resistance without compromising cardiac function.
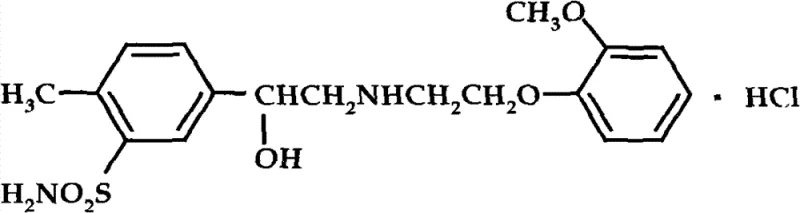
This innovative approach addresses long-standing challenges in the manufacturing of complex beta-blockers by replacing scarce starting materials with commodity chemicals and substituting labor-intensive purification techniques with efficient crystallization protocols. For R&D directors and procurement specialists, understanding the nuances of this pathway is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity API precursors. The method not only simplifies the operational workflow but also aligns with modern green chemistry principles by minimizing waste and avoiding hazardous separation technologies, thereby ensuring a sustainable supply chain for this vital cardiovascular medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Amosulalol Hydrochloride has been hindered by significant logistical and technical bottlenecks that impede large-scale industrial production. Prior art methods, such as those referenced in US patents 5063246 and 4373106, typically rely on 5-acetyl-2-methylbenzenesulfonamide as the foundational starting material. This specific intermediate is not commercially ubiquitous, creating a fragile supply chain dependency where manufacturers must either synthesize this precursor themselves through cumbersome routes or source it from limited vendors, leading to potential delays and inflated costs. Furthermore, traditional purification strategies for the final product heavily depend on column chromatography to separate impurities and isomers. While effective on a laboratory bench scale, column chromatography is notoriously difficult to scale up, requiring vast amounts of solvents, specialized equipment, and extensive labor, which drastically increases the environmental footprint and operational expenditure of the manufacturing process.
The Novel Approach
In stark contrast, the methodology disclosed in CN102050766B revolutionizes the production landscape by utilizing 4-methylacetophenone as the initial feedstock, a commodity chemical that is widely available and economically viable in domestic and international markets. This strategic shift eliminates the supply risk associated with specialized sulfonamide precursors and allows for a linear, contiguous synthesis that is far easier to manage in a reactor setting. The process ingeniously integrates the introduction of the sulfonamide group early in the sequence via a diazotization-sulfonation cascade, ensuring that the core scaffold is established efficiently before proceeding to side-chain modifications. Crucially, the purification of intermediates and the final active pharmaceutical ingredient is achieved through straightforward solvent recrystallization rather than chromatography. This modification not only streamlines the workflow but also significantly enhances the overall yield and throughput, making the process ideally suited for the commercial scale-up of complex polymer additives and pharmaceutical intermediates alike.
Mechanistic Insights into the Diazotization-Sulfonation Cascade
The heart of this synthetic innovation lies in the precise execution of the diazotization and subsequent sulfonation steps, which transform the amino group into the critical sulfonyl chloride functionality. The process begins with the reduction of the nitro group in 4-methyl-3-nitroacetophenone to yield the corresponding aniline derivative. This amine is then subjected to diazotization using sodium nitrite under strictly controlled acidic conditions, typically maintaining temperatures between -40°C and 20°C to prevent the decomposition of the unstable diazonium salt. Instead of isolating this reactive intermediate, the protocol dictates an immediate reaction with a sulfonating agent, such as sulfur dioxide gas or sodium sulfite. This one-pot transformation minimizes handling risks and maximizes atom economy, converting the diazonium species directly into 5-acetyl-2-methylbenzenesulfonyl chloride. The mechanistic efficiency here ensures that the sulfonamide group is positioned ortho to the methyl group and para to the acetyl group with high regioselectivity, which is paramount for the biological activity of the final beta-blocker.
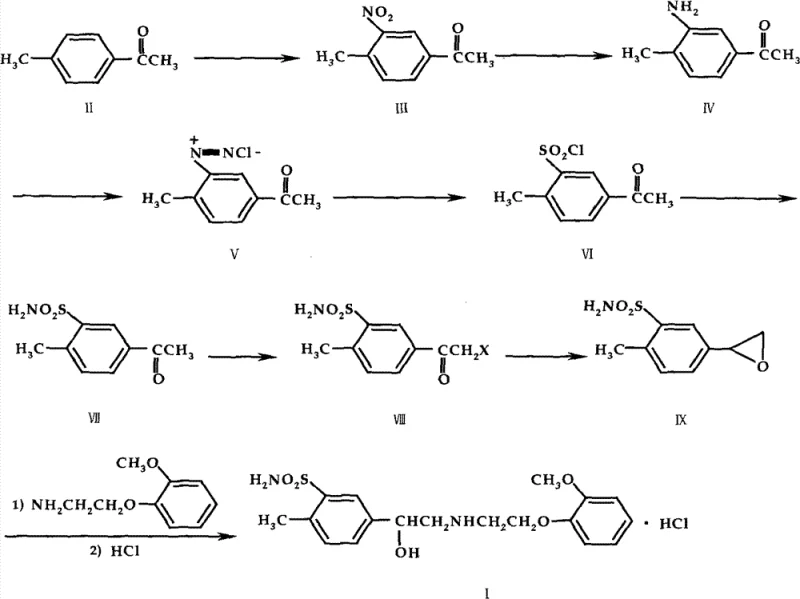
Following the establishment of the sulfonamide core, the synthesis proceeds through a halogenation-reduction sequence to construct the epoxyethyl side chain. The acetyl group is first halogenated using agents like bromine or 1,3-dichloro-5,5-dimethylhydantoin (DCDMH) to form an alpha-halo ketone. This intermediate is then selectively reduced, often using sodium borohydride or catalytic hydrogenation, which induces an intramolecular cyclization to form the epoxide ring. This epoxide serves as a highly reactive electrophile for the final coupling step. The ring is opened by nucleophilic attack from o-methoxyphenoxyethylamine, installing the requisite amino-alcohol side chain characteristic of adrenergic blockers. Finally, treatment with hydrochloric acid yields the stable hydrochloride salt. Each step is designed to operate under mild conditions, avoiding extreme temperatures or pressures that could degrade the sensitive sulfonamide moiety, thereby ensuring a clean impurity profile and high product quality.
How to Synthesize Amosulalol Hydrochloride Efficiently
Executing this synthesis requires careful attention to reaction parameters, particularly temperature control during the exothermic nitration and diazotization phases. The process is divided into three main stages: the construction of the sulfonamide core, the formation of the epoxide intermediate, and the final coupling and salt formation. Operators must ensure rigorous stirring and cooling capabilities to maintain the specified temperature ranges, such as keeping the nitration mixture below 0°C to prevent polynitration or oxidation side reactions. The detailed standardized synthetic steps, including specific molar ratios, solvent choices, and workup procedures for each intermediate from compound II to compound I, are outlined in the comprehensive guide below.
- Nitration of 4-methylacetophenone to form 4-methyl-3-nitroacetophenone using concentrated nitric acid and sulfuric acid at low temperatures.
- Reduction of the nitro group to an amine, followed by diazotization and sulfonation to introduce the sulfonyl chloride group.
- Conversion to sulfonamide, halogenation of the acetyl group, reduction to form the epoxide, and final ring opening with o-methoxyphenoxyethylamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic benefits that extend beyond mere technical feasibility. By shifting the starting point to 4-methylacetophenone, manufacturers can leverage a robust global supply network for raw materials, insulating production schedules from the volatility often associated with niche fine chemical intermediates. The elimination of column chromatography is perhaps the most significant cost driver; removing this step reduces solvent consumption, waste disposal costs, and processing time, leading to substantial cost savings in antihypertensive drug manufacturing. Furthermore, the reliance on recrystallization for purification simplifies the equipment requirements, allowing for the use of standard stainless steel reactors and filtration units rather than specialized chromatography columns, which enhances the flexibility and agility of the production facility.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of expensive, hard-to-source starting materials with commodity chemicals and the removal of chromatographic purification. By avoiding the use of large volumes of organic solvents required for column chromatography, the operational expenditure related to solvent recovery and waste treatment is drastically lowered. Additionally, the higher yields reported in the patent examples suggest a more efficient conversion of raw materials into finished product, reducing the cost per kilogram of the active ingredient. This efficiency allows for more competitive pricing structures without compromising margin, providing a distinct advantage in tender negotiations for generic pharmaceutical formulations.
- Enhanced Supply Chain Reliability: Dependence on a single supplier for a complex intermediate like 5-acetyl-2-methylbenzenesulfonamide poses a significant risk to business continuity. By adopting a route that starts from 4-methylacetophenone, supply chain managers can diversify their vendor base, as this raw material is produced by numerous chemical manufacturers worldwide. This redundancy ensures that production lines remain operational even if one supplier faces disruptions. Moreover, the simplified purification steps reduce the lead time for batch release, enabling faster response to market demand fluctuations and ensuring a steady flow of high-purity API intermediates to downstream formulation partners.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of complex separation technologies make this process inherently scalable from pilot plant to multi-ton commercial production. The reduced solvent load and the ability to recycle mother liquors from recrystallization steps contribute to a smaller environmental footprint, aligning with increasingly stringent global regulations on industrial emissions and waste. This compliance not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is becoming a key criterion for partnerships with major multinational pharmaceutical companies seeking green supply chain solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the practical aspects of scaling this technology. Understanding these details is vital for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of this supply source.
Q: What is the primary starting material for this Amosulalol synthesis?
A: The process utilizes 4-methylacetophenone as the primary starting material, which is a commercially available and cost-effective domestic raw material, unlike previous methods that relied on hard-to-source intermediates.
Q: How does this method improve purification compared to prior art?
A: This novel route eliminates the need for column chromatography, relying instead on simple solvent recrystallization steps which significantly enhances production efficiency and scalability for industrial applications.
Q: What are the key reaction conditions for the diazotization step?
A: The diazotization is performed under acidic conditions using sodium nitrite at temperatures between -40°C and 20°C, followed immediately by sulfonation using sulfur dioxide or sulfites without isolating the intermediate diazonium salt.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amosulalol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, scalable, and compliant manufacturing processes in the modern pharmaceutical landscape. Our team of expert chemists has thoroughly analyzed the synthetic route disclosed in CN102050766B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art reactors capable of handling the low-temperature diazotization and sulfonation steps safely, along with rigorous QC labs that ensure every batch meets stringent purity specifications. Our commitment to quality assurance means that we can deliver Amosulalol Hydrochloride intermediates that are free from the impurities often associated with older synthetic methods, ensuring safety and efficacy for the final drug product.
We invite pharmaceutical partners to collaborate with us to leverage this advanced technology for their supply chains. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized process can reduce your overall cost of goods sold. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your validation studies. Let us be your trusted partner in bringing high-quality, cost-effective antihypertensive medications to the global market.
