Advanced C-H Activation for High-Purity Benzothiadiazole Derivatives in Electronic Materials
Introduction to Next-Generation Benzothiadiazole Functionalization
The landscape of organic synthesis for advanced electronic materials is undergoing a significant transformation, driven by the need for more efficient and sustainable manufacturing processes. A pivotal development in this field is documented in patent CN109096226B, which discloses a highly innovative aryl hydrocarbon activation acetoxylation method utilizing 2,1,3-benzothiadiazole (BTD) as an intrinsic directing group. This technology represents a paradigm shift from traditional cross-coupling strategies, offering a direct route to functionalize the BTD core without the need for pre-installed halogen handles. For R&D directors and procurement specialists in the optoelectronic sector, this methodology promises not only enhanced synthetic efficiency but also a substantial reduction in the environmental footprint associated with producing liquid crystal materials and organic photovoltaic components. The ability to introduce acetoxy groups with high precision opens new avenues for tuning the electronic properties of these conjugated systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction and modification of 2,1,3-benzothiadiazole-based conjugated molecules have relied heavily on palladium-catalyzed cross-coupling reactions, such as the Suzuki-Miyaura coupling. While effective, these conventional pathways suffer from inherent inefficiencies that impact both cost and throughput in a commercial setting. The primary bottleneck lies in the requirement for pre-functionalized starting materials; specifically, the need to synthesize halogenated benzothiadiazole derivatives and corresponding boronic acid partners adds multiple synthetic steps prior to the actual coupling event. This multi-step sequence not only increases the consumption of raw materials and solvents but also generates significant quantities of inorganic salt waste. Furthermore, traditional electrophilic aromatic substitution often lacks the necessary regioselectivity, leading to complex mixtures of mono- and poly-substituted isomers that are difficult and expensive to separate, ultimately depressing the overall yield of the desired target molecule.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN109096226B leverages the unique electronic structure of the benzothiadiazole ring itself to drive the reaction. By employing the nitrogen atoms within the BTD heterocycle as a directing group, the process enables site-specific C-H activation at the ortho-position of the adjacent phenyl ring. This strategic utilization of the substrate's intrinsic functionality eliminates the need for pre-halogenation, effectively shortening the synthetic route by removing entire preparatory stages. The reaction system is remarkably simple, utilizing a mixture of acetic acid and acetic anhydride as both solvent and reagent source, which are inexpensive and readily available on an industrial scale. As illustrated in the general reaction scheme below, this approach allows for the direct conversion of simple biaryl precursors into valuable acetoxylated derivatives with exceptional efficiency.
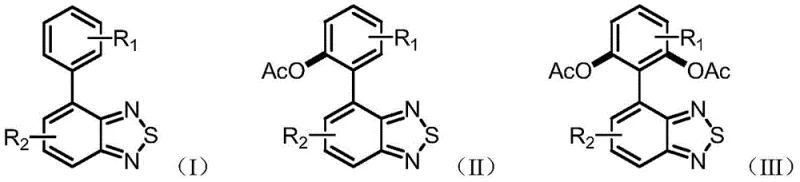
The versatility of this novel approach is further highlighted by its tunable selectivity. By simply adjusting the reaction temperature and the stoichiometry of the oxidant, chemists can selectively produce either the mono-acetoxylated product (Formula II) under mild conditions or the di-acetoxylated product (Formula III) under elevated temperatures. This level of control is critical for manufacturing diverse libraries of electronic materials where specific substitution patterns dictate the final optoelectronic performance. The reported yields are outstanding, reaching up to 99% in optimized cases, which stands as a testament to the robustness of this catalytic system compared to the often moderate yields of traditional multi-step syntheses.
Mechanistic Insights into Pd-Catalyzed Directed C-H Activation
To fully appreciate the value of this technology for high-purity intermediate manufacturing, one must understand the underlying mechanistic pathway that ensures such high regioselectivity. The reaction proceeds through a classic Pd(II)/Pd(IV) catalytic cycle initiated by the coordination of the palladium catalyst to the nitrogen lone pair of the benzothiadiazole ring. This coordination event forms a stable five-membered palladacycle intermediate, which positions the metal center in immediate proximity to the ortho-C-H bond of the pendant phenyl ring. This geometric arrangement drastically lowers the activation energy for the subsequent C-H bond cleavage, a process known as concerted metalation-deprotonation (CMD). The specificity of this interaction ensures that functionalization occurs exclusively at the ortho-position, completely bypassing the statistical distribution of isomers that plagues non-directed methods.
Following the C-H activation step, the organopalladium species undergoes oxidative addition with the hypervalent iodine oxidant, phenyliodine diacetate (PhI(OAc)2), generating a high-valent Pd(IV) intermediate. This step is crucial as it introduces the acetoxy ligands onto the metal center. The cycle concludes with a reductive elimination step, where the C-O bond is formed to release the acetoxylated product and regenerate the active Pd(II) catalyst. The use of acetic anhydride in the solvent mixture plays a dual role: it acts as a dehydrating agent to drive the equilibrium forward and serves as a source of acetate ligands, ensuring a high concentration of the nucleophile required for the final bond-forming step. This mechanistic elegance translates directly to process reliability, minimizing the formation of side products and simplifying downstream purification.
How to Synthesize Acetoxylated Benzothiadiazole Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and purity. The process is designed to be operationally simple, avoiding the need for inert atmosphere techniques or exotic reagents, which facilitates easy adoption by manufacturing teams. The key to success lies in the precise control of reaction temperature and time, which dictates the degree of substitution. For those seeking to produce the mono-acetoxylated intermediate, maintaining the reaction at ambient temperature is critical, whereas higher thermal energy is required to overcome the barrier for the second acetoxylation event. Detailed standardized operating procedures for this transformation are outlined below to guide your technical team.
- Dissolve the 2,1,3-benzothiadiazole substrate, palladium acetate catalyst, and phenyliodine diacetate oxidant in a mixed solvent system of acetic acid and acetic anhydride.
- Control the reaction temperature precisely: maintain at room temperature (20-30°C) for 36 hours to achieve mono-acetoxylation, or heat to 120°C for 16 hours for di-acetoxylation.
- Perform standard post-reaction workup involving ethyl acetate extraction, brine washing, drying over anhydrous sodium sulfate, and purification via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this C-H activation technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The most significant advantage is the drastic simplification of the supply chain for raw materials. By eliminating the need for halogenated precursors and boronic acids, manufacturers can source cheaper, more abundant starting materials, thereby reducing exposure to price volatility in the specialty chemical market. Furthermore, the reduction in synthetic steps directly correlates to a lower cost of goods sold (COGS), as fewer unit operations mean less labor, energy, and equipment time are required to produce each kilogram of the final intermediate. This streamlined process enhances the overall resilience of the supply chain against disruptions.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the principle of atom economy and step reduction. Traditional routes involve the purchase of expensive halogenated building blocks and the generation of stoichiometric amounts of boron and halide waste, which incur disposal costs. In this novel process, the solvent system consists of acetic acid and acetic anhydride, which are commodity chemicals with low procurement costs. Additionally, the catalyst loading is minimal, and the high conversion rates minimize the loss of valuable starting material. These factors combine to create a manufacturing process that is inherently more cost-effective, allowing for substantial savings in the production of complex electronic chemical intermediates without compromising on quality.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for continuous manufacturing operations in the electronics and pharmaceutical sectors. This method improves reliability by reducing dependency on complex, multi-vendor supply chains for specialized coupling partners. Since the starting materials are simpler and more generic, the risk of supply shortages is mitigated. Moreover, the robustness of the reaction conditions—tolerating various substituents on the phenyl ring as shown in the patent examples—means that the process is less sensitive to minor variations in feedstock quality. This tolerance ensures consistent output and reduces the frequency of batch failures, leading to more predictable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to commercial production often reveals hidden challenges, but this acetoxylation method is well-suited for scale-up. The use of common solvents and the absence of sensitive reagents simplify the engineering requirements for large-scale reactors. From an environmental perspective, the process aligns with green chemistry principles by reducing the number of steps and the associated waste stream. The elimination of heavy metal waste typical of stoichiometric coupling reagents and the use of recyclable solvent systems contribute to a lower environmental impact. This compliance with stringent environmental regulations reduces the regulatory burden on manufacturing sites and supports corporate sustainability goals, making it an attractive option for long-term production contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this directed C-H acetoxylation technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for decision-makers evaluating this process for their own supply chains. Understanding these nuances is essential for assessing the feasibility of integrating this method into existing production workflows.
Q: What is the primary advantage of this C-H activation method over traditional Suzuki coupling?
A: Unlike Suzuki coupling which requires pre-functionalized halogenated substrates and boronic acids, this method utilizes the intrinsic nitrogen atoms of the benzothiadiazole ring as a directing group. This eliminates the need for prior halogenation steps, significantly reducing synthetic steps, waste generation, and raw material costs while improving atom economy.
Q: How is regioselectivity controlled in this acetoxylation process?
A: Regioselectivity is strictly governed by the coordination of the benzothiadiazole nitrogen atoms to the palladium center. This directing effect ensures that C-H activation occurs exclusively at the ortho-position relative to the heterocyclic core, preventing the formation of random isomeric mixtures common in non-directed electrophilic aromatic substitutions.
Q: Can this process be scaled for commercial production of optoelectronic intermediates?
A: Yes, the process utilizes inexpensive and commercially available solvents like acetic acid and acetic anhydride, and operates under relatively mild conditions. The high yields reported (up to 99%) and the simplicity of the workup procedure indicate strong potential for scalable manufacturing of high-purity electronic chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,1,3-Benzothiadiazole Derivative Supplier
As the demand for high-performance organic electronic materials continues to surge, the ability to access high-purity intermediates through efficient synthetic routes becomes a critical competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced catalytic technologies like the one described in CN109096226B to deliver superior value to our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2,1,3-benzothiadiazole derivative meets the exacting standards required for optoelectronic applications.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific project requirements, demonstrating how this innovative acetoxylation method can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a reliable supply of high-quality intermediates that will power the next generation of electronic devices.
